Back to Journals » Cancer Management and Research » Volume 11
Clinical features, treatment and outcomes of colorectal mucosa-associated lymphoid tissue (MALT) lymphoma: literature reviews published in English between 1993 and 2017
Authors Won JH, Kim SM, Kim JW, Park JH, Kim JY
Received 2 May 2019
Accepted for publication 13 August 2019
Published 20 September 2019 Volume 2019:11 Pages 8577—8587
DOI https://doi.org/10.2147/CMAR.S214197
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Rudolph Navari
Jae Hee Won,1 Sung Min Kim,1 Jong Wan Kim,1 Jun Ho Park,2 Jeong Yeon Kim1
1Department of Surgery, Dongtan Sacred Hospital, Hallym University College of Medicine, KyungKee, Korea; 2Department of Surgery, Kangdong Sacred Hospital, Hallym University College of Medicine, KyungKee, Korea
Correspondence: Jeong Yeon Kim
Department of Surgery, Dongtan Sacred Hospital, Hallym University College of Medicine, Seokwoodong 42, kyungkee 18450, Republic of Korea
Tel +82 031 8 086 2430
Fax +82 031 8 086 2029
Email [email protected]
Background: Colorectal mucosa-associated lymphoid tissue (MALT) lymphoma (cMALToma) is rare and comprises only 2.5% of the MALT lymphomas. Its etiology and treatment have not been well established. The aim of this systematic literature review was to try to characterize cMALToma and analyze the risk factors for treatment failure with various therapeutic strategies.
Methods: We retrospectively reviewed 50 case reports from 1993 to 2017 through a PubMed search of English medical literature, describing cMALToma. We included 67 patients from previous case reports and we added 6 patients treated for cMALToma in our multicenter institutes. Risk factor analysis was done for treatment failure, defined as remission failure and recurrence.
Results: Of 73 patients diagnosed with cMALToma, tumors were located in 54 patients (74.0%) in the rectum, in 10 patients (13.6%) in the right colon, in 3 patients (4.1%) in the transverse colon, and in 6 patients (8.2%) in the sigmoid colon. In first-line treatment, patients achieved complete remission (CR) with surgery (18/19 cases), local resection (18/19 cases), chemotherapy (12/13 cases), radiation therapy (4/5 cases), antibiotics therapy including Helicobacter pylori eradication (12/15 cases), and no treatment (1/2 cases). Among these, eight cases (10.9%) needed second-line treatment, and there was overall remission failure in 3 cases (4.1%). Of the remaining 70 patients with CR, the tumor recurred in 5 patients (6.8%). The multivariable analysis showed that male sex, multiple tumors, and first-line treatment failure were significantly related to treatment failure (p=0.03, p=0.05, p=0.03, respectively).
Conclusion: CR of primary cMALToma was achieved using various therapeutic strategies. First-line treatment failure and multiple tumors were associated with treatment failure, although the numbers of cases that failed are too small to draw definitive conclusions.
Keywords: MALT-associated lymphoma, colon, rectum, prognosis
Introduction
Mucosa-associated lymphoid tissue (MALT) lymphoma is a subtype of non-Hodgkin’s lymphoma (NHL), accounting for approximately 5% of all NHL.1 MALT lymphoma can occur anywhere external to outside of the lymph nodes, such as in the gastrointestinal (GI) tract, thyroid gland, salivary glands, lungs, and breast. The most common site of occurrence of MALT lymphoma is the stomach in 60% to 75% of cases, followed by small intestine, ileum, cecum, colon, and rectum. Involvement of the esophagus has also been reported, as well as the extra-intestinal sites including lungs, ocular adnexa, lung, breast, and skin.2 Primary malignant lymphoma in the large intestine is rare, accounting for about 1% of GI MALT lymphomas and less than 1% of all colon malignancies.3 MALT lymphoma is known to have a good prognosis, but it may exhibit less response to chemotherapy.4 Thus, treatment methods for MALT lymphoma are being studied, different to those for conventional lymphoma.5
Although colorectal MALT lymphoma is the third most common in GI MALT lymphoma, because of the rarity, its management has not been standardized and interpretation of outcomes following various therapeutic approaches is difficult. This manuscript reviews the English literature on primary colon and rectum MALT lymphoma, describes the treatment failure cases, and analyzes the risk factors.
Methods
We reviewed case reports on colorectal lymphoma from 1993 to 2017. A PubMed search of the English medical literature was conducted using the search words “colon”, “rectum”, and “Maltoma” or “MALT lymphoma” as filters. Through the review, 65 case reports were found, of which 15 case reports were excluded, due to being written in languages other than English, poorly documented cases, and combined colonic disease such as adenocarcinoma or inflammatory bowel disease. Subsequently, 67 patients reported in 50 studies were included from PubMed search (supplementary data). In addition, we retrospectively reviewed the medical records of 6 patients who were pathologically diagnosed and treated (surgery or chemotherapy or endoscopic mucosal dissection; EMR) with colon or rectal MALT lymphoma at OO University-affiliated hospitals (Kangnam Sacred Heart Hospital, Kangdong Sacred Heart Hospital, Chuncheon Sacred Heart Hospital, Dongtan Sacred Heart Hospital, Hangang Sacred Heart Hospital, and Hallym Sacred Heart Hospital) between January 2005 and February 2015 via PubMed. Approval for all research-related activities was obtained from the Institutional Review Board of the Hallym University (IRB 2019–07-016). As all data were accessed through publicly available sources, patient consent was not required. The research followed ethical considerations involved and adhered to the principles enunciated in the Declaration of Helsinki.
Assessment of clinical characteristics and recurrence
Patients' demographic information and characteristics of the disease and its treatment were tabulated. Patient factors included age, sex, tumor location, tumor size, number of tumors, other GI tract involvement, symptoms, size, and status of Helicobacter pylori infection, comorbidity, and diagnosis method. The H. pylori testing modalities included individual or combination evaluation of urea breath test, H. pylori culture, histologic biopsies (gastric and/or rectal), stool antigen, serum antibody, and rapid urease test (Figure 1). Tumor size was based on the longest diameter. Tumor location was categorized as rectum, colon (ascending colon, transverse colon, and sigmoid colon). Symptoms included fecal occult blood, hematochezia or melena, abdominal pain or discomfort, defecation problem such as prolapsed mass, constipation, and others with diagnosed as screening colonoscopy or intussusceptions etc. We categorized tumors into two groups, solitary and multiple. Other GI tract involvement was defined as tumor found in stomach, duodenum, ileum or different colonic site. We divided colon into four quadrants (ascending, transverse, descending/sigmoid, rectum). The different colonic sites were defined as location in a quadrant of colon other than quadrant of the origin of tumor. The diagnosis was done by endoscopic biopsy, excisional biopsy (EMR or transanal excision), and surgery. The treatment methods were also retrieved from case reports or medical records. Treatment variables included radical surgery, local excision (EMR or transanal excision), chemotherapy, radiation, H. pylori eradication (antibiotics therapy) or combination.
 |
Figure 1 The endoscopic finding of H. pylori infection in the stomach. |
The chemotherapy regimens were diverse, including CHOP (cyclophosphamide, doxorubicin, vincristine, prednisolone), MACOPB (methotrexate with leucovorin rescue, doxorubicin, cyclophosphamide, vincristine, prednisone, bleomycin), and R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, prednisolone). And rituximab alone as a single agent had also been used. In patients treated with radiation, the radiation range varied, from 30 to 45 Gy. H. pylori eradiation regimens administered included 7-day course of omeprazole, amoxicillin, and metronidazole, 7 days of rabenprazole, amoxicillin, and clarithromycin, a 10-day course of amoxicillin and clarithromycin, 10 days of levofloxacin repeated three times, a 14-day course of omeprazole, amoxicillin, and clarithromycin, 14 days of amoxicillin and clarithromycin, and undocumented days of amoxicillin and clarithromycin. Dose of medications and times of administration per day varied widely, drug doses were: lansoprazole 30 and 60 mg, rabenprazole 20 mg, amoxicillin 1000 and 1500 mg, clarithromycin 400, 500, 600, and 800 mg, and levofloxacin 300mg.
Complete remission (CR) was defined as the disappearance of the lesion(s) on endoscopic evaluation and histologic diagnosis of MALT with biopsy. Remission failure was defined as no response or partial response after second-line treatment, defined as the additional treatment due to failure of the first-line treatment. Recurrence was defined as new appearance of the tumor in the same lesion or other GI or another organ during the follow-up after CR. Treatment failure comprised remission failure and tumor recurrence.
Statistical analysis
Summary statistics were represented by counts (percentage) or median (range), where appropriate. Chi-squared test was used to compare recurrence among treatment methods (radical surgery vs local excision vs chemotherapy vs radiation vs antibiotics vs combination therapy). Univariable analysis was performed for risk factor analysis of treatment failure using chi-squared test. Comparisons between population medians were performed using a Kruskal Wallis test for non-normally distributed variables for normally distributed variables. Logistic regression model was used to further investigate the association between treatment failure and clinical factors for multivariable analysis. All statistical analyses were performed using SPSS for Windows, version 17.0 (SPSS Inc., Chicago, IL, USA). All reported p-values were two-sided, with statistical significance set at p<0.05.
Results
Patient characteristics
Here, we newly report 6 colorectal MALT lymphoma cases. Tumors were found in rectum in two cases and in four cases in colon (ascending 3, sigmoid 1). Four cases were incidentally found by screening colonoscopy, one case was related to occult blood positive and the other had abdominal discomfort. None of them had B symptoms and any history of inflammatory bowel disease. Surgery was performed in four cases, while there were two other cases where EMR and chemotherapy was done respectively. In more detail, local excision at rectum was performed in two cases and right hemicolectomy at ascending colon in the other two cases. One case showed remission failure with first-line treatment but, CR was achieved after adjuvant chemotherapy. None of them had recurrence (Table 1).
 |
Table 1 Clinical characteristics of colorectal MALT lymphoma cases in six Hallym medical institutes |
A total of 73 patients with colorectal MALT lymphoma were included in our study, data of 6 were retrieved from our multicenter hospital and 67 were from 50 case reports. The median age of disease was 62 (range 26–87). 48 patients (65.8%) were women and the rest (25) were men (34.2%), indicating that women’s incidence rate was two times as high as men. Most patients were diagnosed with MALT lymphoma in rectum (54, 74.0%), 10 patients (13.6%) in right colon, 3 patients (4.1%) in the transverse colon, and 6 (8.2%) patients in the sigmoid colon. Most patients had solitary type (70.0%), while 22 cases showed multiple tumors (30.0%). These colorectal MALT lymphomata developed simultaneously in the colorectum and other intestines such as stomach or duodenum (2), other quadrant of colon (7) in nine cases (12.3%). The most common symptom was hematochezia or melena (38.4%), though, there were cases with no symptoms before the disease was detected through colonoscopy (16.4%). The median tumor size was 20 mm (2~90 mm). The test for H. pylori infection was done in 44 cases and it was positive in 14 cases (19.2%). Pathological confirmation was done by colonoscopic biopsy (46.6%), 20 patients were diagnosed by endoscopic excisional biopsy (27.4%), and 19 patients (26%) were diagnosed surgically without a biopsy. The mean follow-up period was 18.5 months (2–120 months) after treatment (Table 2).
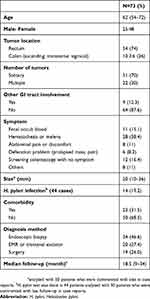 |
Table 2 Demographic and clinical characteristics and diagnostic information |
Therapeutic modality
The therapeutic modalities and recurrence rates are described in Table 3. The following strategies were used for 73 patients as the first-line treatment: surgery (19 cases), local resection (19 cases), chemotherapy (13 cases), radiation therapy (5 cases), antibiotics therapy including H. pylori eradication (15 cases), and no treatment (2 cases). The overall remission rate was 95.8%, not different among patients treated by tumor resection, chemotherapy, radiation therapy, and antibiotics (92.1% vs 92.3% vs 80% vs 80%, p=0.29). Tumor recurrence occurred in 5 (6.8%), treatment variables did not significantly affect the recurrence rate (p=0.20).
 |
Table 3 Therapeutic modality with treatment response |
Among the 73 patients, 8 patients showed no response or partial response and needed second-line treatment (Table 4). There were 2 cases of remission after second-line treatment, following additional tumor resection after the first-line chemotherapy or radiation therapy. Two patients who did not respond to H. pylori treatment had additional radiation therapy, but only one of them achieved remission. One patient who had been treated with H. pylori eradication achieved remission after additional antibiotic treatment. There was one case of residual tumor in the resection margin after a tumor excision, which showed remission after adjuvant chemotherapy. There were two cases in whom no treatment was implemented at all. Tumor disappeared in one of them, while it remained uncured in the other. Overall, remission failure occurred in three cases (4.1%). Tumor recurred in 5 patients (6.8%). Three patients showed local recurrence and two patients showed systemic recurrence. In one case of local recurrence, chemotherapy was implemented with multiple rectal MALT lymphomas, but the tumor recurred in the same site four months thereafter. Another case of local recurrence, protruding masses through sigmoid colon had been removed by excision and subsequent chemotherapy was implemented. In the follow-up colonoscopy, this patient showed an abnormal lesion and underwent EMR, and was then diagnosed with MALT lymphoma. In the last case of local recurrence, a rectal single polypoid mass was treated with H. pylori eradication, but 18 months later, the tumor recurred in the same site. In one case of systemic recurrence, gastric, duodenal, and rectal lesions occurred at the time of initial diagnosis, where chemotherapy was initially performed but the tumor recurred in the same sites. In the other case of systemic recurrence, the tumor recurred on the gingiva of a patient who received chemotherapy for solitary rectal MALT lymphoma.
 |
Table 4 Description of treatment failure cases |
Risk factor analysis of treatment failure
In univariate analysis with chi-squared test, male sex, multiple tumors, and first-line treatment failure were significant high-risk factors for treatment failure (p=0.02, p=0.04, p=0.04, respectively) (Table 5). In multivariate analysis, male sex had a 7.69-fold increased risk, first-line treatment failure 2.50-fold increased risk, and multiple tumors 6.80-fold increased risk for treatment failure (p=0.04, p=0.03, p=0.02, respectively) (Table 6).
 |
Table 5 Univariable analysis of risk factors for treatment failure (second-line treatment failure cases and tumor recurrence) |
 |
Table 6 Multivariable analysis of risk factors for treatment failure (second-line treatment failure cases and tumor recurrence) |
Discussion
In this study, we found that the failure of first-line treatment, multiple tumors, and male sex were associated with a risk of treatment failure. As primary colonic MALT lymphoma is most commonly associated with GI bleeding, surgical resection is a good option for symptomatic treatment of colorectal MALT lymphoma, although this treatment option did not show a significant difference in remission of first-line treatment.
MALT lymphoma was first described by Isaacson and Wright in 1983.2 Although the pathogenesis of MALT lymphoma has not been clearly elucidated, several etiological factors have been postulated to explain this rare disease. There are two types of MALT lymphoma, one that originates from normally present lymphoid tissue like Peyer patches in the gut, and the second that arises from acquired lymphoid tissue that develops in response to inflammation in the setting of infection/autoimmune disease/inflammatory bowel disease/immunosuppression.6
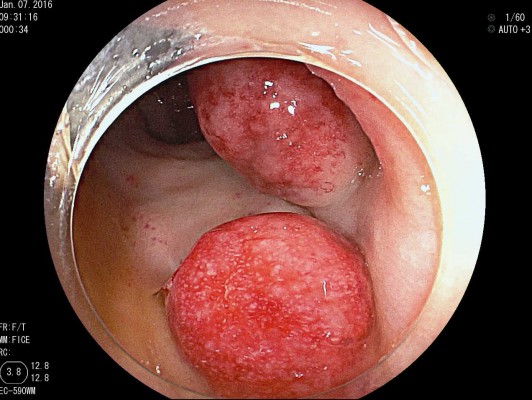
The colonic MALT lymphomas are predominantly seen in the sixth decade of life. The median age of presentation is 62. Although there is no difference among the sexes according to one review,7 female predominance is seen in a 2:1 ratio compared to males. According to previous reports, the majority of colorectal lymphomas are known to develop in the cecum or ascending colon, and >70% are proximal to the hepatic flexure.8 However, based on an analysis of 73 cases, the most common site of colonic MALT lymphoma is the rectum (74.0%) and second site was proximal to the hepatic flexure (13.7%). Clinical presentation ranges from asymptomatic and discovered only through screening colonoscopy to presentation with non-specific abdominal discomfort to more dramatic presentation that includes GI bleeding, defecation problem or pain sometimes combined with intussusception or obstruction. MALT lymphoma of the colon sometimes occurs simultaneously at other sites in other GI tracts such as stomach, duodenum or other quadrant of colon (12.3%); therefore, at the time of diagnosis, synchronous lesions should also be ruled out. So, staging for MALT lymphomas should include gastroscopy, colonoscopy, bone marrow biopsy, and CT of thorax and abdomen. When grossly visible in colonoscopy, most MALT lymphomas are observed as a single mass with protrusion, ulceration or sessile or infiltrated.9 Most commonly, a single polypoid lesion (70%) is seen, but multiple polypoid lesions are also reported (30%) and should be distinguished during colonoscopy (Figure 2). The median tumor size was 20 mm (2–90 mm), so it could easily be treated by using EMR.10,11 In this analysis, colonic MALT lymphoma was diagnosed during surgery (26%) or endoscopic biopsy (46.6%). In the case of endoscopic biopsy, the submucosa must be collected, so, sometimes EMR was preferred for biopsy (27.4%).
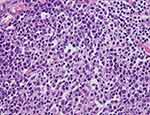 |
Figure 2 The feature of multiple polypoid MALT manifestations in the rectum. |
NHL, including MALT-type can be evaluated using the Ann-Arbor Staging Classification,12 which focuses on the number of tumor sites (nodal vs extranodal), location, and the presence or absence of systemic B symptoms (weight loss, night sweating, unexplained persistent or recurrent fever). Colonic MALT lymphoma is mostly localized and systemic B symptoms are rare.13 Furthermore, because of limited data with cases reports, we only classified as single or multiple and distant organ involvement or not, instead of using this classification. Our data showed, 30% of colonic MALT lymphomas were multiple tumors, and 12.3% involved another GI tract organ. Most cases did not show systemic B symptoms. Diagnosis uses tissue immunohistochemistry and can be used to help distinguish MALT lymphomas from other small B-cell NHLs (Figure 3). B-cell-associated antigens such as CD19, CD20, CD22, and CD79a are usually expressed in such MALT lymphoma specimens.14
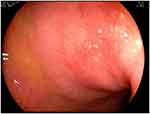 |
Figure 3 Polymorphous immunoblots exhibiting prominent nucleoli within irregular and vesicular nuclei (H&E ×400). |
For colonic MALT lymphoma, there is no standardized therapy and the best treatment modality has long been debated. In most cases, tumor resection or chemotherapy is used as the primary treatment. Remission rates of resection and chemotherapy were higher than 90%. Radical surgery or local excision showed 5.3% treatment failure at first-line treatment. We think surgery can be a good remedy for melena and hematochezia, which are the most common symptoms in colonic MALT lymphoma. Most of the cases treated with surgery had combined symptoms such as bleeding or obstruction related with huge mass. Also, it is known that for other locations, radiation is a very effective treatment modality for MALT lymphoma. However, only five cases with colonic MALT lymphoma were treated with radiation therapy. For the colonic MALT lymphoma, radiation therapy is considered to be limited in view of reduced bowel motility due to problems such as radiation colitis.
The results of the analysis of treatment failure including 3 patients who did not achieve CR and 5 patients who had recurrence after treatment were reported to be comparatively inferior to men and even in the cases of multiple lesions. On multivariate analysis for treatment failure, multiple lesions were analyzed as a risk factor. Kojima et al15 reported that in MALT lymphoma of the rectum in which multiple lesions were found, EMR was done and radiation therapy was added, but remission could not be achieved. Patients treated with chemotherapy for multiple rectal MALT lymphomas also showed recurrence in the same site.16 The reason for treatment failure of multiple tumor type is believed to be the possibility of malignancy hidden in the surrounding lymphoid tissue which manifests as multiple tumors. Therefore, some treatment methods for multiple tumors seemed to have limited abililty to remove all the hidden cancer cells. Interestingly, our data showed that patients without remission following first-line treatment had a high rate of treatment failure. Among 8 patients with failed primary treatment, only 5 patients (62.5%) achieved remission after the second treatment. Therefore, the choice of first-line treatment is very important for treatment of colonic MALT lymphoma. In the case of patients with first-line remission failure, second-line treatment should be considered to select the method for complete removal of primary tumor. When we reviewed final remission failure cases, two patients selected radiation treatment for second-line treatment, but it seemed to be meaningless.15,17 Regarding two risk factors, we should select the best treatment method to achieve complete eradication of primary tumor at first-line treatment. Interestingly, one of the recent articles introduced a new treatment strategy, curative endoscopic resection of a large polypoid MALT lymphoma after downsizing tumors with radiotherapy or chemotherapy. This treatment reduces the size or extent of tumor and increases the possibility of complete removal of the tumor, and can broaden options for treating colonic MALT lymphoma.18,19
While surgery or chemotherapy was chosen as the first-line treatment in most cases, there are rare cases where complete resolution with H. pylori therapy was achieved. Nosaka et al20 reported a case of a patient with rectal MALT lymphoma treated with H. pylori eradication, but the case ended up with local recurrence in the same area within 18 months. Our data showed that antibiotic treatment for H. pylori eradication is not inferior to other treatments such as surgery, radiation, or chemotherapy, even though it has shown a lower remission rate than others; 80% compared to 90%. Further antibiotics prescription was helpful for CR of some failure cases that had previously been treated for H. pylori. This convinced us that colonic MALT lymphoma is also associated with viral infection. Although the pathogenesis of MALT lymphoma in the colorectum has not been elucidated yet, antiviral therapy seems to be effective for the treatment of colorectal MALT lymphoma. However, there have been no previous studies demonstrating the existence of certain mutations as described previously, or virus strains. Although the question of why a certain proportion of colorectal MALT lymphomas may respond to antibiotics remains unanswered, several crucial findings may support the following speculations: 1) HPE prescriptions can also eradicate other bacteria or bacteria similar to H. pylori associated with the development of human colon MALT lymphoma.21,22 For example, Morgner et al23 found that five gastric MALT lymphoma patients with documented Helicobacter heilmannii infection achieved CR after 14 days of omeprazole and amoxicillin therapy. 2) HPE regimens can also eradicate intestinal microorganisms associated with the development of colorectal MALT lymphoma. Lecuit et al24 found that small intestinal MALT lymphoma patients with documented Campylobacter jejuni infection achieved CR with triple antimicrobial therapy (amoxicillin, metronidazole, and clarithromycin) with omeprazole. 3) In addition to eradicating HP and HP-like bacteria, clarithromycin has direct anti-neoplastic or immunomodulatory effects.25,26 In a B-cell lymphoma cell line derived from a BALB/c mouse model, O’Hara et al27 showed that clarithromycin inhibited cell viability and induced apoptosis through down-regulating BCL-2 expression. Mizunoe et al28 showed that macrolides, either clarithromycin or azithromycin, caused apoptosis of activated lymphocytes through attenuation of BCL-XL expression.
This study has some limitations: first, the number of patients was insufficient due to the very rare disease, and was only reported as case reports. Secondly, the data lack consistency as it was collected through multicenter case reports, thus, the accuracy of the data is not guaranteed. Lastly, long-term follow-up results may not be included although we added the analysis of treatment failure. However, our study may still be meaningful since there are no previous studies that have involved separate reviews of treatment failure for colonic MALT lymphoma.
Conclusion
There is a standard established treatment for gastric MALT lymphoma, but this is not the case for colorectal MALT lymphoma. Although colonic MALT lymphoma is an easily curable disease with a relatively low recurrence rate, success of treatment cannot be guaranteed without success of first-line treatment. A careful approach is needed in designing the first-line treatment strategies for patients with multiple tumors or for males. This is because the prognosis for multiple tumors and males is likely to be poor although the number of failure cases is too small to draw definitive conclusions. This finding deserves further validations by investigators in their research.
Acknowledgment
We really appreciate Dong Woo Shin for supervising our article.
Disclosure
All authors declare that they have no conflicts of interest in this work.
References
1. Kahl BS. Update: gastric MALT lymphoma. Curr Opin Oncol. 2003;15(5):347–352.
2. Abbas H, Niazi M, Makker J. Mucosa-Associated Lymphoid Tissue (MALT) lymphoma of the colon: a case report and a literature review. Am J Case Rep. 2017;18:491–497. doi:10.12659/AJCR.902843
3. Shepherd NA, Hall PA, Coates PJ, Levision DA. Primary malignant lymphoma of the colon and rectum. A histopathological and immunohistochemical analysis of 45 cases with clinicopathological correlations. Histopathology. 1988;12(3):235–252. doi:10.1111/j.1365-2559.1988.tb01939.x
4. Hasegawa N, Kato K, Yamada K, et al. Extranodal marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue (MALT) of the sigmoid colon. Gastrointest Endosc. 2000;52(6):802–804. doi:10.1067/mge.2000.110737
5. Chen PH, Lin YM, Yen HH. Primary mucosa-associated lymphoid tissue lymphoma of the colon. Clin Gastroenterol Hepatol. 2011;9(8):e74–5. doi:10.1016/j.cgh.2011.02.018
6. Romaguera J, Hagemeister FB. Lymphoma of the colon. Curr Opin Gastroenterol. 2005;21(1):80–84.
7. Inoue F, Chiba T. Regression of MALT lymphoma of the rectum after anti-H. pylori therapy in a patient negative for H. pylori. Gastroenterology. 1999;117(2):514–515. doi:10.1053/gast.1999.0029900514b
8. Seo SW, Lee SH, Lee DJ, et al. Colonic mucosa-associated lymphoid tissue lymphoma identified by chromoendoscopy. World J Gastroenterol. 2014;20(48):18487–18494.
9. Kim MH, Jung JT, Kim EJ, et al. A case of mucosa-associated lymphoid tissue lymphoma of the sigmoid colon presenting as a semipedunculated polyp. Clin Endosc. 2014;47(2):192–196. doi:10.5946/ce.2014.47.2.192
10. Hasegawa D, Yoshida N, Ishii M, et al. A case of colonic mucosa-associated lymphoid tissue lymphoma observed under endoscopy with narrow-band imaging. Nihon Shokakibyo Gakkai Zasshi. 2013;110(12):2100–2106.
11. Akasaka R, Chiba T, Dutta AK, et al. Colonic mucosa-associated lymphoid tissue lymphoma. Case Rep Gastroenterol. 2012;6(2):569–575. doi:10.1159/000342726
12. Carbone PP, Kaplan HS, Musshoff K, mithers DW, Tubiana M. Report of the committee on hodgkin’s disease staging classification. Cancer Res. 1971;31(11):1860–1861.
13. Zucca E, Conconi A, Pedrinis E, et al. Nongastric marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue. Blood. 2003;101(7):2489–2495. doi:10.1182/blood-2002-04-1279
14. Wotherspoon AC, Doglioni C, Diss TC, et al. Regression of primary low-grade B-cell gastric lymphoma of mucosa-associated lymphoid tissue type after eradication of Helicobacter pylori. Lancet. 1993;342:575–577. doi:10.1016/0140-6736(93)91409-f
15. Kojima M, Itoh H, Motegi A, Sakata N, Masawa N. Localized lymphoid hyperplasia of the rectum resembling polypoid mucosa-associated lymphoid tissue lymphoma: a report of three cases. Pathol Res Pract. 2005;201(11):757–761. doi:10.1016/j.prp.2005.07.007
16. Gavioli M, Bagni A, Santacroce G, Piccagli I, Natalini G. Endorectal sonographic appearances of rectal MALT lymphoma, its response to therapy, and local recurrence. J Clin Ultrasound. 2001;29(7):401–405.
17. Amouri A, Chtourou L, Mnif L, et al. MALT lymphoma of the rectum: a case report treated by radiotherapy. Cancer Radiother. 2009;13(1):61–64. doi:10.1016/j.canrad.2008.11.001
18. Piotrowski R, Kramer R, Kamal A. Image of the month. Extranodal marginal zone B-cell (mucosa-associated lymphoid tissue) lymphoma of the colon presenting as an obstructing mass. Clin Gastroenterol Hepatol. 2008;6(4):e18–19. doi:10.1016/j.cgh.2007.12.033
19. Navarra G, Adani GL, Bardella E, et al. Primary rectal lymphoma as a cause of bowel obstruction. Report of a case.. Tumori. 2003;89(1):88–90.
20. Nosaka K, Shono T, Yonemura Y, et al. Regression of primary rectal MALT lymphoma after Helicobacter pylori eradication. Rinsho Ketsueki. 2014;55(8):948–952.
21. Nakamura S, Yao T, Aoyagi K, Iida M, Fujishima M, Tsuneyoshi M. Helicobacter pylori and primary gastric lymphoma: a histopathologic and immunohisotochemical analysis of 237 patients. Cancer. 1997;79:3–11.
22. Kuo SH, Yeh KH, Wu MS, et al. First-line antibiotic therapy in Helicobacter pylori-negative low-grade gastric mucosa-associated lymphoid tissue lymphoma. Sci Rep. 2017;7(1):14333. doi:10.1038/s41598-017-14102-8
23. Morgner A, Lehn N, Andersen LP, et al. Helicobacter heilmannii-associated primary gastric low-grade MALT lymphoma: complete remission after curing the infection. Gastroenterology. 2000;118:821–828. doi:10.1016/S0016-5085(00)70167-3
24. Lecuit M, Abachin E, Martin A, et al. Immunoproliferative small intestinal disease associated with campylobacter jejuni. N Engl J Med. 2004;350(3):239–248. doi:10.1056/NEJMoa031887
25. Raderer M, Streubel B, Wöhrer S, Häfner M, Chott A. Successful antibiotic treatment of Helicobacter pylori negative gastric mucosa associated lymphoid tissue lymphomas. Gut. 2006;55:616–618. doi:10.1136/gut.2005.083022
26. Govi S, Dognini GP, Licata G, et al. Six-month oral clarithromycin regimen is safe and active in extranodal marginal zone B-cell lymphomas: nal results of a single-centre phase II trial. Br J Haematol. 2010;150:226–229.
27. Ohara T, Morishita T, Suzuki H, Masaoka T, Ishii H, Hibi T. Antibiotics directly induce apoptosis in B cell lymphoma cells derived from BALB/c mice. Anticancer Res. 2004;24:3723–3730.
28. Mizunoe S, Kadota J, Tokimatsu I, Kishi K, Nagai H, Nasu M. Clarithromycin and azithromycin induce apoptosis of activated lymphocytes via down-regulation of Bcl-xL. Int Immunopharmacol. 2004;4:1201–1207. doi:10.1016/j.intimp.2004.05.011
29. Matsumoto T, Shimizu M, Iida M, Amano K, Nakamura S, Fujishima M. Primary low-grade, B-cell, mucosa-associated lymphoid tissue lymphoma of the colorectum: clinical and colonoscopic features in six cases. Gastrointest Endosc. 1998;48(5):501–508. doi:10.1016/s0016-5107(98)70092-6
30. Chahil N, Bloom P, Tyson J, Jazwari S, Robilotti J, Gaultieri N. Novel approach to treatment of rectal mucosa-associated lymphoid tissue lymphoma. BMJ Case Rep. 2011;2011:2969. doi:10.1136/bcr.05.2010.2969
31. Matsumoto T, Inokuma T, Imai Y. Education and imaging. Gastrointestinal: colonic mucosa-associated lymphoid tissue lymphoma regressed by levofloxacin. J Gastroenterol Hepatol. 2013;28(4):750. doi:10.1111/jgh.2013.28.issue-4
32. Bschorer R, Lingenfelser T, Kaiserling E, Schwenzer N. Malignant lymphoma of the mucosa-associated lymphoid tissue (MALT)–consecutive unusual manifestation in the rectum and gingiva. J Oral Pathol Med. 1993;22(4):190–192.
33. Gianni L, Tassinari D, Sartori S, Rinaldi P, Ravaioli A. Gastric, duodenal and rectal multifocal MALT lymphoma: the possible co-existence of two different cell populations. Eur J Cancer. 1998;34(10):1640–1641. doi:10.1016/s0959-8049(98)00145-2
34. Tanaka S, Ohta T, Kaji E, Kosaka T, Murakami I. EMR of mucosa-associated lymphoid tissue lymphoma of the rectum. Gastrointest Endosc. 2003;57(7):956–959. doi:10.1016/s0016-5107(03)70039-x
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
