Back to Journals » Neuropsychiatric Disease and Treatment » Volume 17
Clinical Evaluation of the Efficacy and Safety of Co-Administration of Wuzhi Capsule and Tacrolimus in Adult Chinese Patients with Myasthenia Gravis
Authors Peng Y, Jiang F, Zhou R, Jin W, Li Y, Duan W, Xu L, Yang H
Received 10 May 2021
Accepted for publication 24 June 2021
Published 12 July 2021 Volume 2021:17 Pages 2281—2289
DOI https://doi.org/10.2147/NDT.S319500
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yuping Ning
Yuyao Peng, Fei Jiang, Ran Zhou, Wanlin Jin, Yi Li, Weiwei Duan, Liqun Xu, Huan Yang
Department of Neurology, Xiangya Hospital, Central South University, Changsha, 410008, People’s Republic of China
Correspondence: Liqun Xu; Huan Yang
Department of Neurology, Xiangya Hospital, Central South University, Changsha, 410008, People’s Republic of China
Tel +86 731-8432-7236
Fax +86 731-8432-7401
Email [email protected]; [email protected]
Background: Tacrolimus has been recommended as an effective immunosuppressant for patients with myasthenia gravis (MG), while the high price, variable bioavailability, and narrow therapeutic window restrict its clinical application. Wuzhi capsule (WZC) could improve tacrolimus blood concentration by inhibiting the metabolism of cytochrome P450 3A (CYP3A) and P-glycoprotein (P-gp). There are few studies focused on the coadministration of WZC and tacrolimus in autoimmune diseases. This study was aimed at quantifying the efficacy and safety of coadministration of WZC and tacrolimus in adult Chinese patients with MG.
Methods: In this retrospective study, 122 patients with MG on tacrolimus were enrolled. The initial tacrolimus dose was 2 mg/d. Patients with standard initial tacrolimus concentration were classified into group A (standard-dose group). Those failed to reach target concentration were divided into group B (high-dose group) and group C (co-administering WZC group), according to treatment adjustment of increasing tacrolimus dose and co-administration of WZC, respectively. A logistic analysis was used to identify factors associated with clinical outcome. Adverse drug reactions (ADRs) were recorded for safety analysis.
Results: The tacrolimus concentration after coadministration of WZC was remarkably increased. It was higher compared with simply increasing the tacrolimus dose (p< 0.001). The multivariate logistic analysis indicated that the baseline quantitative MG score was a predictive factor for clinical outcomes (OR=0.189; 95% CI 0.082– 0.436; p< 0.001). Fourteen patients (11.5%) reported ADRs after tacrolimus therapy. ADRs incidence was not related to WZC coadministration.
Conclusion: The coadministration of WZC and tacrolimus can substantially elevate the tacrolimus concentration. It is a safe and economic treatment for adult Chinese patients with MG. Patients with a worse disease condition tend to present a better clinical outcome after tacrolimus therapy.
Keywords: myasthenia gravis, tacrolimus, Wuzhi capsule, clinical efficacy
Introduction
Tacrolimus (FK506) is a calcineurin inhibitor that was first found in the fermentation broth of a strain of Streptomyces1 and is widely used in organ transplantation and autoimmune diseases. Since 2002, a low dose of tacrolimus (3 mg/d) has been allowed as an immunosuppressant in the treatment of MG in Japan.2,3 Emerging clinical studies have shown that tacrolimus can effectively and safely improve the clinical condition of patients with MG.4,5 Besides suppressing interleukin-2 (IL-2) production associated with T-cell activation and inhibiting differentiation and proliferation of T-cells, tacrolimus also has been reported to have the ability to enhance ryanodine receptor function and increase muscle strength.6 Compared with azathioprine and mycophenolate mofetil, tacrolimus displayed better clinical efficacy and tolerance in Chinese patients with MG.7 Recently, tacrolimus monotherapy has been confirmed to be a promising option to rapidly alleviate all symptoms of MG, especially for ptosis and bulbar symptoms.8
The efficacy of tacrolimus in MG treatment has been considered to be concentration dependent. Patients with an adequate tacrolimus concentration have a better MG prognosis than those with a low tacrolimus concentration.9 However, tacrolimus has a narrow therapeutic index and a variable rate of absorption and oral bioavailability; approximately 25% of the oral dose is bioavailable.10 Lots of food and medications may affect tacrolimus metabolism, such as grape juice, magnesium oxide, sodium bicarbonate and sucralfate. The adverse events of tacrolimus include diarrhea, rash, respiratory infection, nephrotoxicity, and hepatotoxicity; the occurrence of nephrotoxicity is concentration dependent. Previous studies have shown that the risk of early nephrotoxicity increases by 22% with a 1-ng/mL increase in FK506.11 The incidence of adverse events related to tacrolimus treatment was 37.5–80.0% in Chinese patients with MG.8,12,13 Several prospective studies with tacrolimus doses ranging from 2.0 to 4.5 mg/d and most of them set the tacrolimus dose as 3 mg/d.4,14,15 During clinical observation and practice, we found that some patients administered 2 mg/d tacrolimus achieved the target blood concentration and experienced remarkable improvement. The expense associated with long-term tacrolimus therapy has always been a challenge for patients. Tacrolimus at low doses reduces the occurrence of adverse events and is economical, but it is difficult to achieve the target therapeutic blood concentration.
Wuzhi capsule (WZC) is a formulation containing the ethanol extract from the ripe fruits of Nan-Wuweizi (Schisandra sphenanthera). The main active ingredients include schisandrin, schisantherin A, schizandrol B, schisanhenol, and deoxychizandrin. It is a cheap traditional Chinese medicine that has been extensively used to treat chronic hepatitis and drug-induced liver injury in organ transplant recipients.16,17 It can increase the blood concentration and bioavailability of tacrolimus owing to its inhibitory effect on cytochrome P450 3A (CYP3A) and P-glycoprotein (P-gp) with no obvious adverse reactions.18,19 Most studies on coadministration of WZC and tacrolimus have been performed in organ transplant patients and focused on pharmacokinetics and pharmacogenetic characteristics. There are few studies conducted in patients with autoimmune diseases. Thus, in this study, we aimed to quantify the efficacy and safety in co-administration of Wuzhi capsule and tacrolimus in adult Chinese patients with MG. The findings will promote the coadministration of WZC and tacrolimus in clinical practice.
Materials and Methods
Patients and Data Collection
Patients who visited the Neurology Department of Xiangya Hospital of Central South University, China, from November 2016 to November 2019, diagnosed with MG, and received tacrolimus as an immunosuppressant were selected. The study was approved by the Ethics Committee of Xiangya Hospital (approval number 201703107) and conducted in accordance with the Declaration of Helsinki. All patients signed an informed consent form. The inclusion criteria were as follows:
(1) patients with confirmed diagnosis of MG according to the typical clinical manifestations and the results of the neostigmine test, repetitive nerve stimulation test, and MG-related autoantibody test;
(2) patients over the age of 18 years;
(3) patients receiving low-dose tacrolimus therapy with tacrolimus concentration detection; and
(4) patients followed-up for at least 3 months after tacrolimus administration.
The following demographic and clinical data were collected: age, sex, disease duration, baseline disease severity, and quantitative myasthenia gravis (QMG) scores evaluated before tacrolimus administration and 3 months after tacrolimus therapy. Baseline disease severity was determined according to the Myasthenia Gravis Foundation of America (MGFA) classification at the start of tacrolimus therapy and categorized as ocular MG (MGFA class 1) and generalized MG (MGFA classes 2A and 2B, MGFA classes 3A and 3B, and MGFA classes 4A, 4B, and 5).20 Disease duration was defined as the time from disease onset to the time of tacrolimus therapy initiation. Clinical efficacy was estimated by the changes in the QMG scores, with ≥3 (2) point changes indicating clinically significant treatment effects when the baseline QMG score >16 (≤16).21,22 The results of the autoantibody test (performed at Guangzhou Daan Clinical Laboratory Center) were also collected; anti-AChR antibody (Ab) was detected using an AChR Ab ELISA Kit (RSR Ltd., Cardiff, UK), with a concentration of ≥0.45 nmol/L defined as positive. We also investigated adverse drug reactions (ADRs) including nephrotoxicity, increased liver enzymes, headache, dizziness, and tremors. The incidence of ADRs after tacrolimus administration was described with a number for each ADR and the number of affected patients.
Treatment Methods and Tacrolimus Concentration Detection
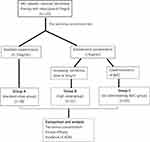
In this retrospective study, all patients received tacrolimus as an immunosuppressant. Besides the prednisolone, no additional immunosuppressive agents were used at the same time. The initial dose of tacrolimus was 2 mg/d. Patients were treated with tacrolimus capsules twice a day (Huadong Medicine Co., Ltd, national medicine permission number: H20094027) and were strictly required to take tacrolimus on an empty stomach. Other food and medicines should not be taken within two hours of taking tacrolimus. FK506 concentration was detected between the 7th to 14th day of tacrolimus therapy by liquid chromatography-mass spectrometry in the Clinical Laboratory of Xiangya Hospital, with a target tacrolimus concentration of 5–10 ng/mL. The dose of tacrolimus was adjusted considering the individual’s disease condition and tacrolimus concentration. The patients who reached the target tacrolimus concentration were included in Group A (standard-dose group). The tacrolimus dose was increased to 3 mg/d in those who failed to reach the target, and the patients were placed in Group B (high-dose group). Some patients chose other immunosuppressants instead of tacrolimus because the cost of tacrolimus treatment increased with the increase of tacrolimus dose, and they were not included into this retrospective study. As WZC was administered together with tacrolimus from 2019, some patients with sub-optimal tacrolimus concentrations were required to administer two tablets of WZC (Sichuan HYGIEN Pharmaceutical Co., Ltd, national medicine permission number: Z10983013) together with tacrolimus every time and were included in Group C (co-administering WZC group). The second tacrolimus concentration was determined 1 month after treatment adjustment. The flowchart of this retrospective study is presented in Figure 1.
Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics, version 25.0 (IBM Corp., Armonk, NY, USA). Quantitative data with a normal distribution are reported as the mean ± standard deviation (SD), and those with a non-normal distribution are reported as the median (interquartile range values (IQRs) p25, p75). Categorical data are presented as count and proportion. When the quantitative data followed a normal distribution, Student’s t-test or one-way analysis of variance was conducted. Otherwise, non-parametric Mann–Whitney U-test or Kruskal–Wallis H-test was conducted, as appropriate. Categorical data were compared using the Chi-square test or Fisher’s exact test. Multivariate logistic regression analyses were carried out using Back logistic regression method. Covariates were selected according to clinical meaningful aspects and the results of bivariate analysis. Variables with a p-value of <0.2 in bivariate analysis were then included in the final model to estimate the odds ratio (OR) and 95% confidence interval (CI). Results with a p-value of <0.05 were considered significant.
Results
Demographic and Clinical Characteristics
In this study, 122 patients were enrolled, with 40 men and 82 women (Table 1). The age of patients ranged from 18 to 80 years, and the median age was 40 years. The median disease duration was 32 months. Thymoma was observed in 26 patients (21.3%). Before receiving tacrolimus therapy, 17 patients were classified under ocular MG (OMG) and 105 under generalized MG (GMG). A total of 107 patients underwent the AChR Ab test and the positive rate was 86.9% (96/107). The median AChR Ab titer was 13.86 ng/mL. 63 patients took prednisolone in combination with tacrolimus. The median baseline QMG score was 10.00. The median change in the QMG score was −3.00. Tacrolimus therapy in 70 patients was clinically effective, whereas in 52 patients, it was clinically ineffective.
 |
Table 1 Demographic and Clinical Characteristics |
There were 38 patients in group A, 31 in group B, and 53 in group C. The median disease duration in group A was 43 months, whereas that in group B was 27 months and in group C was 21 months. There were significant differences among the three groups (p=0.044). Other generic data, such as sex and age, were not significantly different among the three groups (p>0.05). The ACHR Ab positivity rate was statistically significant among the three groups (p=0.033): 94.1% in group A, 96.0% in group B, and 77.1% in group C. However, there was no significant difference in the remaining clinical data, including thymus, MGFA classification, ACHR Ab titer, co-administration of prednisolone, baseline QMG score, QMG change, and clinical efficacy among the three groups (all p>0.05).
FK506 in Different Subgroups
The FK506 concentration in group A was 7.30 ± 2.48 ng/mL. It was 2.69±0.98 ng/mL in group B, whereas the final FK506 concentration turned to be 5.48±2.99 ng/mL after increasing the tacrolimus dose to 3 mg/d. In group C, the FK506 concentration before co-administering WZC was 2.51±1.13 ng/mL, which increased to 8.19±2.91 ng/mL after co-administering WZC. The results summarized in Table 2 suggest that the initial FK506 concentration between group A, group B and group C was significant (p<0.001), although it was not significant between groups B and C (p=0.356). The final FK506 concentration was higher after co-administering WZC than after increasing the tacrolimus dose (p<0.001). The FK506 concentration after increasing the tacrolimus dose in group B was still lower than the initial FK506 concentration in group A (p=0.001). The FK506 concentration after co-administering WZC in group C was higher than the initial FK506 concentration in group A (p=0.039). The final FK506 concentration between group A, group B and group C was significant (p<0.001).
 |
Table 2 Comparisons of Tacrolimus Concentration in Different Subgroups |
Factors Associated with Clinical Effectiveness
To investigate probable factors associated with clinical effectiveness, we compared the clinical characteristics among MG patients according clinical outcome (Table 3). There were 70 patients classified into effective group, the other 52 patients were classified into ineffective group. The thymus histology and baseline QMG score were significantly different (p<0.05). Variables with p-value of <0.2 were entered into multivariate logistic regression model for final analysis, including thymus histology, final tacrolimus concentration, coadministration of WZC and baseline QMG score. The results of multivariate logistic regression suggested that the baseline QMG score (OR=0.189; 95% CI 0.082–0.436; p=0.000) was a predictive factor for the clinical outcome (Table 4).
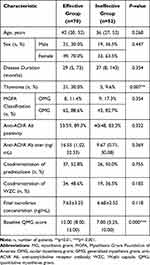 |
Table 3 Clinical Characteristic Among MG Patients According to Clinical Outcome for Tacrolimus Therapy |
 |
Table 4 Logistic Regression Analysis of Factors Associated with Clinical Effectiveness |
Adverse Effects of Tacrolimus Therapy
Overall, 14 patients (14/122, 11.5%) experienced ADRs after starting tacrolimus treatment, and the total number of ADRs observed was 27 (Table 5). In Group A, 6 (6/38, 15.8%) patients reported ADRS. In Group C, 8 (8/53, 15.1%) patients reported ADRs. The incidence of ADRs between these two groups was not significant (p=0.928). No patient in Group B reported ADRs. The most frequent ADRs were gastrointestinal disturbances (5/27, 18.5%), abnormal dreams (4/27, 14.8%), increased blood glucose concentration (3/27, 11.1%), and increased blood lipid concentration (3/27, 11.1%). Other less frequent ADRs, such as nephrotoxicity, muscle pain, upper respiratory infection, and dizziness, were also recorded. Most ADRs resolved spontaneously or with a reduction in the tacrolimus dose or by treating the symptoms. Patients reporting serious ADRs were not included in this study because of discontinuation of tacrolimus treatment.
 |
Table 5 List of Adverse Drug Reactions (ADRs) After Tacrolimus Therapy |
Discussion
In this study, 122 patients with MG were enrolled and 53 patients took WZC together with tacrolimus. The FK506 concentration in group A was 7.30±2.48 ng/mL. It was 2.69±0.98 ng/mL in group B, whereas the final FK506 concentration turned to be 5.48±2.99 ng/mL after increasing the tacrolimus dose to 3 mg/d. In group C, the FK506 concentration before co-administering WZC was 2.51±1.13 ng/mL, whereas it increased to 8.19±2.91 ng/mL after co-administering WZC. A comparison of the initial FK506 concentration among different groups revealed that the initial FK506 between groups B and C was not different (p=0.356). However, the final FK506 concentration was higher after co-administering WZC than after increasing the tacrolimus dose (p<0.001). The FK506 concentration after co-administering WZC in group C was even higher than the initial FK506 concentration in group A (p=0.039), indicating that administering WZC together with tacrolimus could effectively increase the tacrolimus concentration, consistent with the findings of a previous study.23 The main active ingredients of WZC such as schisandrin A, schisandrol A and schisandrol B are the substrate of CYP3A4 and CYP3A5. When co-administering WZC with tacrolimus, metabolism of these compounds might be occurred in a competitive manner. Schisandrin A, schisandrol A and schisandrol B with a higher affinity to CYP3A could result in a stronger metabolism by CYP3A, which means a lower metabolism of tacrolimus and a higher tacrolimus concentration.17,24 Although some patients took prednisolone in combination with tacrolimus and there was no strict consistent regimen for prednisolone dose, it has been confirmed that prednisolone has no effect on tacrolimus concentration in MG patients.25 And the proportion of co-administration of prednisolone among different subgroups was similar (p>0.05). Thus, the increase of tacrolimus concentration in group C was caused by co-administration of WZC. This finding is meaningful for patients who fail to reach the target concentration range with an initial tacrolimus dose of 2 mg/d. In clinical practice, the high price of tacrolimus is considered a limitation for long-term therapy; WZC is relatively less expensive. The price of a tablet of tacrolimus was approximately 13 times of a tablet of WZC. Compared with increasing the tacrolimus dose, co-administering WZC with tacrolimus remarkably improves the tacrolimus concentration, which could help relieve financial burdens and increase patient compliance at the same time.
Additionally, we found that the initial FK506 concentration between different groups was significantly different (p<0.001). The FK506 concentration after increasing the tacrolimus dose in group B was still lower than the initial FK506 concentration in group A (p=0.001), and this is consistent with previous study findings. There were many factors associated with the FK506 concentration, such as patients’ heterogeneity, gene polymorphism, and initial tacrolimus dose. Several pharmacokinetic and pharmacodynamic studies have shown that genetic factors play an important role in responses to tacrolimus treatment, particularly those related to CYP3A4 and CYP3A5 polymorphisms. Patients with different CYP3A4 and CYP3A5 genotypes require different doses of tacrolimus to reach the same therapeutic window.26–28 A previous study in our laboratory also confirmed this.29
Furthermore, we focused on the clinical efficacy of co-administration of WZC and tacrolimus. By comparing the clinical characteristics among MG patients according the clinical outcome, we found that there were more patients with thymoma in the effective group (p=0.007). This might because tacrolimus also has effects on the ryanodine receptor related sarcoplasmic calcium release to potentiate excitation-contraction coupling in skeletal muscles. Ryanodine receptor antibodies occur in some MG patients, especially those with thymoma.30 Tacrolimus might exert extra therapeutic benefit in thymoma-associated MG patients.31,32 The multivariate logistic regression results showed that FK506 concentration was not associated with clinical efficacy (p>0.05), consistent with the findings of previous studies in our laboratory.29,33 We also found that the clinical outcome between group A, group B and group C was not significantly different (p=0.278). The proportion of co-administration of WZC between effective group and ineffective group did not significantly differ (p=0.185). Although the co-administration of WZC significantly improved the tacrolimus concentration, it seemed to have no effect on the clinical efficacy. The target therapeutic range is not the only key to clinical outcomes. Tacrolimus treatment regimens and responses vary greatly from one individual to another. The relationship between pharmacokinetic and pharmacodynamic factors and clinical efficacy requires further research.
In addition, the results of the multivariate logistic analysis indicated that baseline the QMG score was a predictive factor of clinical outcome (OR=0.189; 95% CI 0.082–0.436; p<0.001). Patients with a higher QMG score were more likely present clinical efficacy. In previous studies, the QMG scores of 10–16 and >16 indicated mild and severe diseases, respectively.22 The higher the QMG score, the worse the disease condition. This suggests that patients with worse disease conditions might be more easily treated with tacrolimus therapy.
Indeed, we found that the incidence of ADRs was not related to the co-administration of WZC. Fourteen patients (11.5%) reported ADRs after tacrolimus therapy; this number was relatively lower than that reported in previous studies, where the incidence of adverse events in patients treated with tacrolimus varied from 0% to 87.5%.34–36 This might be because these studies were carried out using different designs. It should be noted that the adverse events in the early periods of tacrolimus therapy partially account for the discontinuation of tacrolimus although they are not very serious. In this study, patients who were affected by insufferable ADRs and discontinued tacrolimus treatment were excluded, which might result in a low incidence of ADRs. The incidence of ADRs between groups A and C was similar (p=0.928), indicating that the co-administration of tacrolimus and WZC did not increase the risk of tacrolimus therapy. However, none of the patients reported ADRs in group B. This might be because the FK506 concentration in group B was lower than in the other two groups even after increasing the tacrolimus dose.
Limitations
This study has several limitations, especially due to the retrospective observational study design. There was no placebo-controlled group and the sample size was relatively small. As tacrolimus has been gradually widely recommended and applicated in patients with MG, the time from the onset of the disease to the beginning of tacrolimus therapy tends to be shortened. This might account for the statistical analysis results that there was a significant difference in the disease duration among group A, group B and group C (p=0.044). We only evaluated the clinical efficacy by comparing the changes in the QMG scores at the time of tacrolimus therapy as 3 months. Further studies are required to evaluate the long-term efficacy of tacrolimus treatment.
Conclusions
In conclusion, our study proved that the coadministration of WZC and tacrolimus can substantially elevate the tacrolimus concentration. It is a safe and affordable treatment for adult Chinese patients with MG. Patients with a worse disease condition tend to present a better clinical outcome after tacrolimus therapy. These results provide valuable information for individualized, precise treatment with tacrolimus.
Data Sharing Statement
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Consent for Publication
All patients involved in the study signed an informed consent form.
Acknowledgments
We would like to thank Editage for English language editing.
Funding
This work was supported by a grant from the National Natural Science Foundation of China (Grant number: 81771364).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Kino T, Hatanaka H, Miyata S, et al. FK-506, a novel immunosuppressant isolated from a Streptomyces. II. Immunosuppressive effect of FK-506 in vitro. J Antibiot (Tokyo). 1987;40(9):1256–1265. doi:10.7164/antibiotics.40.1256
2. Nagane Y, Utsugisawa K, Obara D, Kondoh R, Terayama Y. Efficacy of low-dose FK506 in the treatment of Myasthenia gravis--a randomized pilot study. Eur Neurol. 2005;53(3):146–150. doi:10.1159/000085833
3. Yoshikawa H, Mabuchi K, Yasukawa Y, Takamori M, Yamada M. Low-dose tacrolimus for intractable myasthenia gravis. J Clin Neurosci. 2002;9(6):627–628. doi:10.1054/jocn.2001.0907
4. Cruz JL, Wolff ML, Vanderman AJ, Brown JN. The emerging role of tacrolimus in myasthenia gravis. Ther Adv Neurol Disord. 2015;8(2):92–103. doi:10.1177/1756285615571873
5. Wang L, Zhang S, Xi J, et al. Efficacy and safety of tacrolimus for myasthenia gravis: a systematic review and meta-analysis. J Neurol. 2017;264(11):2191–2200. doi:10.1007/s00415-017-8616-7
6. Imai T, Tsuda E, Hozuki T, et al. Early effect of tacrolimus in improving excitation-contraction coupling in myasthenia gravis. Cin Neurophysiol. 2012;123(9):1886–1890. doi:10.1016/j.clinph.2012.01.017
7. Zhang C, Bu B, Yang H, et al. Immunotherapy choice and maintenance for generalized myasthenia gravis in China. CNS Neurosci Ther. 2020;26(12):1241–1254. doi:10.1111/cns.13468
8. Fan Z, Li Z, Shen F, et al. Favorable effects of tacrolimus monotherapy on myasthenia gravis patients. Front Neurol. 2020;11:594152. doi:10.3389/fneur.2020.594152
9. Kanai T, Uzawa A, Kawaguchi N, et al. Adequate tacrolimus concentration for myasthenia gravis treatment. Eur J Neurol. 2017;24(2):270–275. doi:10.1111/ene.13189
10. Venkataramanan R, Swaminathan A, Prasad T, et al. Clinical pharmacokinetics of tacrolimus. Clin Pharmacokinet. 1995;29(6):404–430. doi:10.2165/00003088-199529060-00003
11. Jacobson PA, Schladt D, Israni A, et al. Genetic and clinical determinants of early, acute calcineurin inhibitor-related nephrotoxicity: results from a kidney transplant consortium. Transplantation. 2012;93(6):624–631. doi:10.1097/TP.0b013e3182461288
12. Zhao CB, Zhang X, Zhang H, et al. Clinical efficacy and immunological impact of tacrolimus in Chinese patients with generalized myasthenia gravis. Int Immunopharmacol. 2011;11(4):519–524. doi:10.1016/j.intimp.2010.12.012
13. Zhou L, Liu W, Li W, et al. Tacrolimus in the treatment of myasthenia gravis in patients with an inadequate response to glucocorticoid therapy: randomized, double-blind, placebo-controlled study conducted in China. Ther Adv Neurol Disord. 2017;10(9):315–325. doi:10.1177/1756285617721092
14. Konishi T, Yoshiyama Y, Takamori M, Saida T. Long-term treatment of generalised myasthenia gravis with FK506 (tacrolimus). J Neurol Neurosurg Psychiatry. 2005;76(3):448–450. doi:10.1136/jnnp.2004.042176
15. Ahn SW, Joo IS, Kim BJ, et al. A multicenter prospective observational study on the safety and efficacy of tacrolimus in patients with myasthenia gravis. J Neurol Sci. 2017;379:271–275. doi:10.1016/j.jns.2017.05.060
16. Loo WT, Cheung MN, Chow LW. Fructus schisandrae (Wuweizi)-containing compound inhibits secretion of HBsAg and HBeAg in hepatocellular carcinoma cell line. Biomed Pharmcother. 2007;61(9):606–610. doi:10.1016/j.biopha.2007.08.023
17. Qin XL, Chen X, Zhong GP, et al. Effect of Tacrolimus on the pharmacokinetics of bioactive lignans of Wuzhi tablet (Schisandra sphenanthera extract) and the potential roles of CYP3A and P-gp. Phytomedicine. 2014;21(5):766–772. doi:10.1016/j.phymed.2013.12.006
18. Wei H, Tao X, Di P, et al. Effects of traditional chinese medicine Wuzhi capsule on pharmacokinetics of tacrolimus in rats. Drug Metab Dispos. 2013;41(7):1398–1403. doi:10.1124/dmd.112.050302
19. Xie SP, Yan Q, Chen HZ. [Study on the clinical application of wuzhi capsule after renal transplantation]. Zhongguo Zhong Xi Yi Jie He Za Zhi Zhongguo Zhongxiyi Jiehe Zazhi. 2011;31(9):1213–1215. [Chinese]
20. Andersen JB, Gilhus NE, Sanders DB. Factors affecting outcome in myasthenia gravis. Muscle Nerve. 2016;54(6):1041–1049. doi:10.1002/mus.25205
21. Benatar M, Sanders DB, Burns TM, et al. Recommendations for myasthenia gravis clinical trials. Muscle Nerve. 2012;45(6):909–917. doi:10.1002/mus.23330
22. Katzberg HD, Barnett C, Merkies IS, Bril V. Minimal clinically important difference in myasthenia gravis: outcomes from a randomized trial. Muscle Nerve. 2014;49(5):661–665. doi:10.1002/mus.23988
23. Sun Z, Ren M, Wu Q, Du X. Co-administration of Wuzhi capsules and tacrolimus in patients with idiopathic membranous nephropathy: clinical efficacy and pharmacoeconomics. Int Urol Nephrol. 2014;46(10)):1977–1982. doi:10.1007/s11255-014-0801-3
24. Qin XL, Yu T, Li LJ, et al. Effect of long-term co-administration of Wuzhi tablet (Schisandra sphenanthera extract) and prednisone on the pharmacokinetics of tacrolimus. Phytomedicine. 2013;20(3–4):375–379. doi:10.1016/j.phymed.2012.11.008
25. Fan Z, Zheng D, Wen X, et al. CYP3A5*3 polymorphism and age affect tacrolimus blood trough concentration in myasthenia gravis patients. J Neuroimmunol. 2021;355:577571. doi:10.1016/j.jneuroim.2021.577571
26. Chen D, Hou S, Zhao M, Sun X, Zhang H, Yang L. Dose optimization of tacrolimus with therapeutic drug monitoring and CYP3A5 polymorphism in patients with myasthenia gravis. Eur J Neurol. 2018;25(8):1049–e1080. doi:10.1111/ene.13652
27. Hesselink DA, van Schaik RH, van der Heiden IP, et al. Genetic polymorphisms of the CYP3A4, CYP3A5, and MDR-1 genes and pharmacokinetics of the calcineurin inhibitors cyclosporine and tacrolimus. Clin Pharmacol Ther. 2003;74(3):245–254. doi:10.1016/S0009-9236(03)00168-1
28. Chen L, Prasad GVR. CYP3A5 polymorphisms in renal transplant recipients: influence on tacrolimus treatment. Pharmgenomics Pers Med. 2018;11:23–33.
29. Meng HY, Li X, Jin WL, et al. Multiple genetic factors affecting the pharmacokinetic and pharmacodynamic processes of tacrolimus in Chinese myasthenia gravis patients. Eur J Clin Pharmacol. 2020;76(5):659–671. doi:10.1007/s00228-019-02803-0
30. Romi F, Aarli JA, Gilhus NE. Myasthenia gravis patients with ryanodine receptor antibodies have distinctive clinical features. Eur J Neurol. 2007;14(6):617–620. doi:10.1111/j.1468-1331.2007.01785.x
31. Takamori M, Motomura M, Kawaguchi N, et al. Anti-ryanodine receptor antibodies and FK506 in myasthenia gravis. Neurology. 2004;62(10):1894–1896. doi:10.1212/01.WNL.0000125254.99397.68
32. Mitsui T, Kunishige M, Ichimiya M, Shichijo K, Endo I, Matsumoto T. Beneficial effect of tacrolimus on myasthenia gravis with thymoma. Neurologist. 2007;13(2):83–86. doi:10.1097/01.nrl.0000256352.77668.ef
33. Shumei Y, Yi L, Huanyu M, et al. IL-2 gene polymorphisms affect tacrolimus response in myasthenia gravis. Eur J Clin Pharmacol. 2019;75(6):795–800. doi:10.1007/s00228-019-02642-z
34. Yoshikawa H, Kiuchi T, Saida T, Takamori M. Randomised, double-blind, placebo-controlled study of tacrolimus in myasthenia gravis. J Neurol Neurosurg Psychiatry. 2011;82(9):970–977. doi:10.1136/jnnp-2011-300148
35. Shimojima Y, Matsuda M, Gono T, Ishii W, Tokuda T, Ikeda S. Tacrolimus in refractory patients with myasthenia gravis: coadministration and tapering of oral prednisolone. J Clin Neurosci. 2006;13(1):39–44. doi:10.1016/j.jocn.2004.12.008
36. Tada M, Shimohata T, Tada M, et al. Long-term therapeutic efficacy and safety of low-dose tacrolimus (FK506) for myasthenia gravis. J Neurol Sci. 2006;247(1):17–20. doi:10.1016/j.jns.2006.03.010
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.