Back to Journals » International Journal of General Medicine » Volume 13
Clinical Efficacy of Baloxavir Marboxil in the Treatment of Seasonal Influenza in Adult Patients: A Prospective Observational Study
Authors Yoshino Y , Kitazawa T, Ota Y
Received 22 July 2020
Accepted for publication 7 September 2020
Published 1 October 2020 Volume 2020:13 Pages 735—741
DOI https://doi.org/10.2147/IJGM.S273266
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Yusuke Yoshino,1 Takatoshi Kitazawa,1 Yasuo Ota2
1Department of Internal Medicine, Teikyo University School of Medicine, Itabashi, Tokyo, Japan; 2Department of Internal Medicine, National Hospital Organization Higashi Saitama Hospital, Hasuda, Saitama, Japan
Correspondence: Yusuke Yoshino Department of Internal Medicine
Teikyo University School of Medicine, 2-11-1 Kaga, Itabashi-ku 173-8605, Tokyo, Japan
Tel +81 3 3964 1211 (Ext 7731)
Fax +81 3 3579 6310
Email [email protected]
Purpose: Baloxavir marboxil, a recently developed antiviral drug, has been used to treat influenza in some countries including Japan. The aim of this study was to determine the clinical efficacy of the drug, which currently remains unclear.
Patients and Methods: Overall, 43 adult patients with seasonal influenza who visited the outpatient clinic of Teikyo University Hospital in Tokyo during the winter of 2018– 2019 were enrolled. Of them, 14, 13, and 16 were prescribed baloxavir marboxil (40 or 80 mg once), oseltamivir (75 mg twice daily for 5 days), and laninamivir (40 mg once), respectively. A questionnaire was used to collect data about symptoms, and the Medical Outcome Study 8-Items Short Form Health Survey was employed to examine health-related quality-of-life (HRQOL) before and 7 days after admission. The main study endpoints included time to defervescence and the extent of improvement in HRQOL after treatment initiation. The data were analyzed with Welch’s t-test and Fisher’s exact test using StatFlex version 6.
Results: No significant differences in clinical background characteristics were observed among the patients. The mean time to defervescence in the baloxavir group (median [interquartile range]; 1.0 [1.0– 2.0] days) was significantly shorter than that in the laninamivir group (2.0 [1.5– 3.5] days; p=0.0322). No significant differences in mean time to defervescence, change in HRQOL, and time for resolution of other symptoms were observed between the groups.
Conclusion: The results suggest that baloxavir marboxil has a better antipyretic effect than oseltamivir and laninamivir. Moreover, baloxavir marboxil might be clinically more valuable than the other two drugs owing to higher medication adherence among patients.
Keywords: seasonal influenza, baloxavir marboxil, clinical efficacies, health-related quality-of-life
Introduction
Influenza patients present with signs and symptoms of upper and/or lower respiratory tract infection, accompanied by systemic illness symptoms such as fever, headache, myalgia, and weakness. Apart from being a self-limiting disease, influenza can also occur as a pandemic. The influenza pandemic often leads to severe complications, death, and social and economic disruption. For example, the 1918 influenza pandemic led to a high number of deaths.1 In the last 40 years, the 2009 H1N1 influenza virus, also known as the swine flu virus, has been the first new influenza pandemic. The highly pathogenic influenza A virus subtype H7N9, which is an emerging avian influenza virus, has been globally considered a potential pandemic threat.2 In addition, the emergence of novel pandemic strains remains a continuous threat. Therefore, influenza prevention and control strategies including vaccination and new antiviral agents have been the topic of research worldwide.
In Japan, influenza is usually treated with neuraminidase inhibitors such as oseltamivir, zanamivir, laninamivir, and peramivir. A novel antiviral drug, called baloxavir marboxil, has recently been approved in Japan and the United States.3 This drug has become commercially available in Japan in March 2018. It reduces viral proliferation through a new mechanism of action wherein it inhibits cap-dependent endonucleases. The treatment of influenza is accomplished with a single oral dose of this drug. In Phase 3 clinical trials, baloxavir marboxil significantly shortened the time to alleviation of influenza symptoms when compared to that achieved with placebo. Moreover, it reduced the infectious viral titer and the duration of virus shedding more rapidly than oseltamivir in otherwise healthy patients (CAPSTONE-1) and high-risk patients (CAPSTONE-2).4,5 However, this latest anti-flu agent is currently available in a limited number of countries, and its clinical efficacy in clinical practice remains undetermined.
In addition, seasonal flu is usually a self-limiting condition and lasts for 3–7 days in healthy individuals, although it is severe in some cases. However, influenza can lead to other indirect consequences such as school absenteeism, loss of workplace productivity, and low health‐related quality-of-life (HRQOL). Indeed, influenza was reported to contribute to a substantial loss of HRQOL at the population level.6,7 However, the extent of change in HRQOL among flu patients treated with baloxavir marboxil has never been evaluated.
In this study, we evaluated and compared the clinical course and changes in HRQOL among adult patients with seasonal influenza treated with oseltamivir, laninamivir, or baloxavir marboxil to examine the clinical efficacy of baloxavir marboxil.
Patients and Methods
This prospective, observational, single-center trial compared the clinical efficacies of baloxavir marboxil, oseltamivir, and laninamivir. The study protocol was reviewed and approved by the Ethics Committee of Teikyo University (No. TUIC-COI 18–0760).
Patients
In this trial, we recruited influenza patients who visited the outpatient clinic of Teikyo University Hospital, Tokyo, Japan, between November 2018 and April 2019. Written informed consent was obtained from all study participants. A patient was diagnosed with influenza depending on a positive result for the influenza rapid antigen test (Fujirebio Espline Influenza A&B-N, Tokyo, Japan) and presentation of signs and symptoms of influenza-like illness, such as fever, muscle pain, chills, headache, dry cough, fatigue, nasal congestion, disorientation, and respiratory failure, without other focal signs of infection.8 Fever was defined as an axillary temperature of ≥37.0°C, and any patient febrile for ≥48 hours on the first hospital visit was excluded from this trial. The patients had been prescribed either oseltamivir (75 mg twice daily for 5 days), laninamivir (40 mg once), or baloxavir marboxil (40 or 80 mg once) at their physicians’ discretion.
Study Variables
Demographic and clinical data, including age, sex, maximum body temperature after disease onset, and time course of fever, were collected on the first visit. Moreover, we recorded data of symptoms such as cough, sore throat, nasal discharge, headache, muscle pain, joint pain, nausea/vomiting, and diarrhea, as well as body temperature, pulse rate, and type of influenza (influenza A or B). We also assessed the general HRQOL after 7 days of treatment based on the Japanese version of the Medical Outcome Study 8-Items Short Form Health Survey (SF-8).9,10 The SF-8 is an HRQOL assessment that consists of eight scales and generates two summary measures: a physical component summary (PCS) and a mental component summary (MCS). After influenza treatment, the clinical course of the symptoms was evaluated using a questionnaire. Patients who did not submit or respond to the questionnaires were excluded from this study.
The main endpoints of this study were time to defervescence after treatment and the extent of improvement in HRQOL. Resolution of other symptoms after treatment was also evaluated. If the body temperature fell below 37°C and did not rise to normal for 2 days, then the first day that the body temperature fell below 37°C was defined as the day of alleviation of fever. The extent of improvement in HRQOL was based on changes in the PCS and MCS and was defined as the difference between HRQOL after treatment initiation and that before treatment initiation. The results were compared among the three anti-flu agents.
Statistical Analysis
The results were expressed as median [interquartile range] unless otherwise stated. For multiple comparisons of independent groups, Wilcoxon signed-rank test, Fisher’s exact test, or Mann–Whitney U-test were used to analyze continuous and categorical data, as appropriate. All p-values were two-sided, and p < 0.05 was considered to indicate statistical significance. StatFlex version 6 (Artech Co., Osaka, Japan) was used for all statistical analyses.
Results
Seventy patients were enrolled in the study; however, only 43 patients could be followed up: 13 patients in the oseltamivir group, 16 in the laninamivir group, and 14 in the baloxavir group. The clinical background characteristics of the study population are summarized in Table 1. The duration of fever on the first visit was similar among the three groups. The other clinical characteristics and accompanying symptoms were not significantly different among the three groups without the MCS. A significant difference in MCS on the first day was observed between the oseltamivir group and the laninamivir group and between the baloxavir group and the laninamivir group.
 |
Table 1 Clinical Background Characteristics of the Study Population |
The extent of improvement in HRQOL in the three groups is shown in Figure 1. Regarding the efficacies of the anti-flu agents, the PCS improved significantly in each group, whereas the MCS significantly worsened only in the laninamivir group. However, the extent of change in either the PCS or the MCS was not significantly different across the groups.
As shown in Figure 2, the time to defervescence in the baloxavir group (1.0 [1.0–2.0] days) was significantly shorter than that in the laninamivir group (2.0 [1.5–3.5] days; p=0.0322). The median time to defervescence in the baloxavir group was also shorter than that in the oseltamivir group (3.0 [1.0–3.0] days). However, there was no significant difference between them (p=0.0670). As shown in Figure 3, the percentage of defervescence in the baloxavir group was significantly higher than that in the other groups on the second day of treatment.
The data of the mean time of the disappearance of symptoms other than fever are listed in Table 2. Table 3 shows the data on the number of patients who continued to have cough even after 7 days of treatment in each group. Digestive symptoms, ie, nausea, vomiting, and diarrhea, occurred in some cases after the patients started receiving anti-flu treatment (Table 4). However, no significant difference was found across groups.
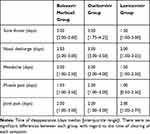 |
Table 2 Average Time of Disappearance of Symptoms |
 |
Table 3 Number of Patients Who Continued to Have Cough 7 Days After Treatment |
 |
Table 4 Digestive Symptoms That Occurred After Treatment Initiation |
Discussion
This study showed that the PCS in patients with seasonal flu was significantly improved after 7 days of treatment, while the MCS significantly worsened after 7 days of treatment. The differences in terms of PCS and MCS changes among groups were not significant. The type of the anti-flu agent had no association with the change in HRQOL after treatment initiation. During the infection, HRQOL worsened significantly. After the patient’s full recovery from symptoms, the HRQOL also fully recovered. However, there are no reports on the short-time change in each side of the MCS and PCS of HRQOL in seasonal flu patients after treatment. Worsening of the MCS of the HRQOL only after treatment seems to be one of the specific features of seasonal flu. Seasonal flu is well known as a highly contagious disease and can easily appear as an outbreak. An infected person should undergo self-quarantine to inhibit the spread of the virus. These facts might let mental QOL of flu patients worsen. In addition, low MCS of the HRQOL can lower labor productivity. It can also cause sleep disturbances and depression. We should pay attention to the changes in the MCS of HRQOL of the patients with seasonal flu during treatment.
Regarding the antipyretic effect, baloxavir marboxil might be superior to the other two anti-flu drugs. The CAPSTONE 1 study showed that baloxavir marboxil was not significantly different from oseltamivir in terms of clinical efficacy in relieving clinical symptoms, eg, fever.4 The CAPSTONE 2 study demonstrated that baloxavir marboxil could reduce fever in type B influenza patients at a significantly faster rate than oseltamivir.5 In addition, both clinical studies showed that baloxavir marboxil reduced viral loads more significantly than oseltamivir. In line with our study, the previous studies also suggested the clinical superiority of baloxavir marboxil over oseltamivir, although factors such as race, generation, and phenotypes might confound the clinical effects of these drugs. However, baloxavir marboxil is clinically valuable owing to high medication adherence among patients.
Nevertheless, baloxavir marboxil resistance has been observed in some clinical trials. The influenza strain became resistant in 2.2% and nearly 10% of baloxavir recipients in Phase 2 and 3 trials, respectively.4 The resistance might be due to the presence of polymerase acidic protein variants with I38T/M/F substitutions. The rate of acquiring resistance to baloxavir marboxil is relatively high; however, viruses harboring the I38T substitution showed severely impaired replicative fitness in cells.11 Indeed, the effect of the emergence of these resistant strains in the clinical field has not been fully evaluated. However, I38T mutant influenza A (H3N2) viruses were isolated from baloxavir-treated children in December 2018.12 In the future, criteria regarding which patients should receive baloxavir marboxil need to be developed. This issue was not discussed in the present study because we used the rapid antigen test to diagnose influenza and evaluated only the clinical course of our patients using a questionnaire.
This study had several limitations that should be considered while interpreting the results. First, this study was conducted for a short period and at a single center. Second, the sample size was relatively small. However, this study makes an important contribution to the existing literature given that very few reports on the clinical efficacy of baloxavir marboxil for its use in the treatment of seasonal influenza have been published.
Conclusion
In summary, our study results suggest that baloxavir marboxil might have a better antipyretic effect than other drugs. Moreover, baloxavir marboxil might be clinically more valuable than the other two drugs owing to higher medication adherence among patients. Indeed, only a single oral dose of baloxavir marboxil is needed for influenza treatment. Nevertheless, a large prospective randomized controlled study is required to fully demonstrate the efficacy of baloxavir marboxil in the real world.
Ethical Approval
This study was approved by the Ethics Committee of the Teikyo University School of Medicine (No. TUIC-COI 18-0760), and written informed consent was obtained from all study participants. Routine data for clinical purposes were used, and all patient information remained confidential in the study. All humans research procedures were followed in accordance with the standards set forth in the Declaration of Helsinki principles of 1975, as revised in 2013 (http://ethics.iit.edu/ecodes/node/3931).
Acknowledgments
We would like to thank Editage (www.editage.com) for English language editing.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
None. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Taubenberger JK, Morens DM. 1918 Influenza: the mother of all pandemics. Emerg Infect Dis. 2006;12(1):15–22. doi:10.3201/eid1209.05-0979
2. Su S, Gu M, Liu D, et al. Epidemiology, evolution, and pathogenesis of H7N9 influenza viruses in five epidemic waves since 2013 in China. Trends Microbiol. 2017;25(9):713–728. doi:10.1016/j.tim.2017.06.008
3. Locke SC, Splawn LM, Cho JC. Baloxavir marboxil: a novel cap-dependent endonuclease (CEN) inhibitor for the treatment of acute uncomplicated influenza. Drug Today. 2019;55(6):359–366. doi:10.1358/dot.2019.55.6.2999889
4. Hayden FG, Sugaya N, Hirotsu N, et al. Baloxavir marboxil for uncomplicated influenza in adults and adolescents. N Engl J Med. 2018;379(10):913–923. doi:10.1056/NEJMoa1716197
5. Ison MG, Portsmouth S, Yoshida Y, Shishido T, Hayden F, Uehara T. LB16. Phase 3 trial of baloxavir marboxil in high-risk influenza patients (CAPSTONE-2 study). Open Forum Infect Dis. 2018;5(suppl_1):S764–S5. doi:10.1093/ofid/ofy229.2190
6. Fragaszy EB, Warren-Gash C, White PJ, et al. Effects of seasonal and pandemic influenza on health-related quality of life, work and school absence in England: results from the Flu Watch cohort study. Influenza Other Respir Viruses. 2018;12(1):171–182. doi:10.1111/irv.12506
7. Yang J, Jit M, Zheng Y, et al. The impact of influenza on the health related quality of life in China: an EQ-5D survey. BMC Infect Dis. 2017;17(1):686. doi:10.1186/s12879-017-2801-2
8. Centers for Disease Control and Prevention NCfIaRDN. Clinical signs and symptoms of influenza. Available from: https://www.cdc.gov/flu/professionals/acip/clinical.htm2019.
9. Tokuda Y, Okubo T, Ohde S, et al. Assessing items on the SF-8 Japanese version for health-related quality of life: a psychometric analysis based on the nominal categories model of item response theory. Value Health. 2009;12(4):568–573. doi:10.1111/j.1524-4733.2008.00449.x
10. Fukuhara S, Suzukamo Y. Manual of the SF-8 Japanese version: institute for health outcomes & process evaluation research, Kyoto. 2004.
11. Omoto S, Speranzini V, Hashimoto T, et al. Characterization of influenza virus variants induced by treatment with the endonuclease inhibitor baloxavir marboxil. Sci Rep. 2018;8(1):9633.
12. Takashita E, Kawakami C, Morita H, et al. Detection of influenza A (H3N2) viruses exhibiting reduced susceptibility to the novel cap-dependent endonuclease inhibitor baloxavir in Japan, December 2018. Euro Surveill. 2019;24(3). doi:10.2807/1560-7917.ES.2019.24.3.1800698.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



