Back to Journals » Clinical Ophthalmology » Volume 8
Clinical course of focal choroidal excavation in Vogt–Koyanagi–Harada disease
Authors Nishikawa Y, Fujinami K, Watanabe K, Noda T, Tsunoda K, Akiyama K
Received 8 October 2014
Accepted for publication 30 October 2014
Published 4 December 2014 Volume 2014:8 Pages 2461—2465
DOI https://doi.org/10.2147/OPTH.S75558
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Yuko Nishikawa,1–3,* Kaoru Fujinami,1,2,4,5,* Ken Watanabe,1,2 Toru Noda,1,2 Kazushige Tsunoda,1,2 Kunihiko Akiyama1,2
1Department of Ophthalmology, National Hospital Organization, Tokyo Medical Center, Tokyo, Japan; 2Laboratory of Visual Physiology, National Institute of Sensory Organs, National Tokyo Medical Center, Tokyo, Japan; 3Department of Ophthalmology, Osaka Medical College, Takatsuki, Osaka, Japan; 4Department of Ophthalmology, Keio University School of Medicine, Tokyo, Japan; 5UCL Institute of Ophthalmology, London, UK
*These authors contributed equally to this work
Abstract: We describe focal choroidal excavation (FCE) in a case of Vogt–Koyanagi–Harada (VKH) disease and compare the findings with different chorioretinal conditions. A 55-year-old man was diagnosed with VKH based on panuveitis and exudative retinal detachments. Spectral-domain optical coherence tomography demonstrated a dome-shaped protrusion with a nonconforming pattern at the fovea, which had been detected as a conforming pattern 1 year before the onset. The FCE pattern returned into a conforming pattern following corticosteroid therapy. These findings suggest that the natively existent FCE could be affected by pathophysiological changes of VKH as well as other chorioretinal conditions.
Keywords: choroidal excavation, focal choroidal excavation, Vogt–Koyanagi–Harada disease, optical coherence tomography
Introduction
Focal choroidal excavation (FCE) was first described by Jampol et al1 as an anomalous excavation of the choroid; it is typically observed by optical coherence tomography (OCT).1,2 This condition has been associated with several chorioretinal conditions and asymptomatic status, including central serous chorioretinopathy, choroidal neovascularization, and best vitelliform macular dystrophy.1–12 However, the mechanisms underlying the formation of FCE remain uncertain.
Wakabayashi et al2 described two patterns of FCE: 1) conforming pattern – excavations that involved the outer retinal layers up to and including the external limiting membrane; and 2) nonconforming pattern – excavations that involved only the retinal pigment epithelium (RPE).
Recently, a case report of FCE associated with Vogt–Koyanagi–Harada (VKH) disease has been published,13 which documented cross-sectional observation. Here we describe a clinical course of FCE associated with VKH before and after treatment and compare the morphological findings of FCE in four subjects with different chorioretinal conditions.
Case report
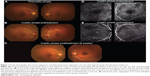
A 55-year-old man (subject 1) presented with bilateral metamorphopsia, with decimal visual acuity being 0.8 in the right eye and 1.2 in the left. The clinical diagnosis of VKH was made based on bilateral panuveitis and multifocal exudative retinal detachments at the posterior poles (Figure 1). High-dose corticosteroid therapy with gradual tapering was initiated 3 days after presentation. The serous detachments entirely resolved within 9 weeks and visual acuity in the right eye improved to 1.2.
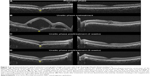
Serial spectral-domain OCT (SD-OCT) images obtained at the preuveitic, uveitic, and posttreatment phase are shown in Figure 2. SD-OCT obtained 1 year before the onset of VKH due to symptoms of a floater and visual disturbance demonstrated a dome-shaped posterior protrusion of the RPE and outer retinal layers into the choroidal cavity (ie, conforming FCE) at the fovea in the right eye. SD-OCT at the uveitic phase identified multiple bilateral sensory retinal detachments, with the FCE now involving only the RPE (ie, nonconforming FCE). Nine weeks after the treatment, the nonconforming FCE returned into a conforming pattern.
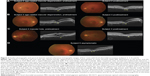
The FCE pattern and its alteration during treatment of the case with VKH were compared to those of four subjects with FCE associated with other chorioretinal conditions (Figure 3). This comparison group consisted of two patients with age-related macular degeneration (AMD; subjects 2 and 3), one subject with a macular hole (MH; subject 4), and one asymptomatic individual (subject 5). Treatment with intravitreal ranibizumab injections and vitrectomy had been performed in the patients with AMD and MH, respectively. A conforming FCE was observed in three patients (subjects 2, 3, and 5) and pattern alteration from nonconforming to conforming FCE was found after treatment in one subject (subject 4). In the two subjects with AMD, a hyperreflective material around the FCE was absorbed, resulting in a well-demarcated conforming FCE after treatment.
Discussion
A detailed clinical course of FCE at the fovea was documented in a case with VKH disease. SD-OCT images obtained at the preuveitic, uveitic, and posttreatment phase suggested the preexistence of FCE before the onset of VKH, and the FCE pattern change (from conforming to nonconforming pattern during the period of choroidal inflammation) was observed during the follow-up.
The FCE identified at the preuveitic phase in our case supported the congenital/acquired posterior pole malformation, as previously suggested.1,4,9,11,12 On the other hand, Hashida et al13 speculated that a direct pressure effect on the choroidal layer by subretinal fibrin disrupted the choroidal integrity and focal choroidal atrophy/thinning following inflammation and resulted in a formation of FCE in a case with VKH.4,7,13 Although such a remarkable subretinal fibrin was not detected, the association between the FCE formation and choroidal inflammation could not be excluded entirely in our case, considering other possible subclinical inflammatory events prior to this disease history.
An alteration of the FCE pattern observed in VKH was also found in the comparison group with other chorioretinal conditions. In subject 4 with an MH, the nonconforming FCE at presentation associated with vitreomacular traction changed to conforming FCE after treatment. In two subjects with AMD, a hyperreflective material around the FCE was observed to be absorbed, resulting in a well-demarcated conforming FCE via treatment (subjects 2 and 3). These findings suggest that the cavity within FCE can be filled with subretinal fluid associated with inflammation and exudative material generated by choroidal neovascularization. The decreased adhesion within the FCE between RPE and photoreceptor could complicate MHs. Given the proximity between the FCE and the active lesions, the involved retina around the active lesion may well show disorganization of retinal layers in the pathological process; then the conforming pattern is hard to maintain, unsurprisingly. In addition, it is possible that the cavity within FCE may potentially accelerate the pathophysiological changes of chorioretinal disorders.
Acknowledgments
This research was supported in part by research grants from the Ministry of Health, Labor and Welfare, Japan and Grant-in-Aid for Scientific Research from Japan Society for the Promotion of Science. KF is supported by a Grant-in-Aid for Young Scientists (B) of the Ministry of Education, Culture, Sports, Science and Technology (Japan). The sponsor or funding organization had no role in the design or conduct of this research.
Disclosure
The authors have no proprietary or commercial interest in any materials discussed in this article. The authors report no other conflicts of interest in this work.
References
Jampol LM, Shankle J, Schroeder R, Tornambe P, Spaide RF, Hee MR. Diagnostic and therapeutic challenges. Retina. 2006;26(9):1072–1076. | ||
Wakabayashi Y, Nishimura A, Higashide T, Ijiri S, Sugiyama K. Unilateral choroidal excavation in the macula detected by spectral-domain optical coherence tomography. Acta Ophthalmol. 2010;88(3):e87–e91. | ||
Margolis R, Mukkamala SK, Jampol LM, et al. The expanded spectrum of focal choroidal excavation. Arch Ophthalmol. 2011;129(10):1320–1325. | ||
Ellabban AA, Tsujikawa A, Ooto S, et al. Focal choroidal excavation in eyes with central serous chorioretinopathy. Am J Ophthalmol. 2013;156(4):673–683. | ||
Obata R, Takahashi H, Ueta T, Yuda K, Kure K, Yanagi Y. Tomographic and angiographic characteristics of eyes with macular focal choroidal excavation. Retina. 2013;33(6):1201–1210. | ||
Say EA, Jani PD, Appenzeller MF, Houghton OM. Focal choroidal excavation associated with polypoidal choroidal vasculopathy. Ophthalmic Surg Lasers Imaging Retina. 2013;44(4):409–411. | ||
Lee CS, Woo SJ, Kim YK, et al. Clinical and spectral-domain optical coherence tomography findings in patients with focal choroidal excavation. Ophthalmology. 2014;121(5):1029–1035. | ||
Or C, Forooghian F. Vitelliform focal choroidal excavation. Ophthalmic Surg Lasers Imaging Retina. 2014;45 Online:e26–e28. | ||
Xu H, Zeng F, Shi D, Sun X, Chen X, Bai Y. Focal choroidal excavation complicated by choroidal neovascularization. Ophthalmology. 2014;121(1):246–250. | ||
Parodi MB, Zucchiatti I, Fasce F, Bandello F. Bilateral choroidal excavation in best vitelliform macular dystrophy. Ophthalmic Surg Lasers Imaging Retina. 2014;45 Online:e8–e10. | ||
Suzuki M, Gomi F, Hara C, Sawa M, Nishida K. Characteristics of central serous chorioretinopathy complicated by focal choroidal excavation. Retina. 2014;34(6):1216–1222. | ||
Lee JH, Lee WK. Choroidal neovascularization associated with focal choroidal excavation. Am J Ophthalmol. 2014;157(3):710–718.e1. | ||
Hashida N, Fok A, Nishida K. Choroidal excavation in vogt-koyanagi-harada disease. Case Rep Ophthalmol. 2014;5(2):222–225. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.