Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 16
Clinical Control Criteria to Determine Disease Control in Patients with Severe COPD: The CLAVE Study
Authors Soler-Cataluña JJ , Almagro P , Huerta A , González-Segura D , Cosío BG
Received 7 October 2020
Accepted for publication 7 January 2021
Published 25 January 2021 Volume 2021:16 Pages 137—146
DOI https://doi.org/10.2147/COPD.S285385
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Juan José Soler-Cataluña,1 Pere Almagro,2 Arturo Huerta,3 Diego González-Segura,4 Borja G Cosío5 On behalf of the CLAVE study Investigators
1Department of Pneumology, Hospital Universitari Arnau de Vilanova and CIBERES, Valencia, Spain; 2Multimorbidity Patients Unit. Internal Medicine Department, H. Mutua Terrassa University Hospital, Terrassa, Barcelona, Spain; 3Emergency Department – Medicine and Pulmonary Section, H. Clínic de Barcelona, Barcelona, Spain; 4Medical Department, Chiesi SAU, Barcelona, Spain; 5Department of Pneumology, H. Universitari Son Espases Hospital-IdISBa and CIBERES, Palma de Mallorca, Balearic Islands, Spain
Correspondence: Juan José Soler-Cataluña
Department of Pneumology, H. Universitari Arnau de Vilanova, C/San Clemente 12, Valencia 46015, Spain
Tel + 973 24 81 00
Email [email protected]
Background: Clinical control in chronic obstructive pulmonary disease (COPD) has not been completely characterized. A proposal of clinical control criteria (CCC) has been recently defined and validated as a tool for determining control, but there is scarce information on patients with severe COPD.
Objective: To evaluate clinical control in severe COPD using the CCC.
Patients and Methods: The study design was observational, multicenter, cross-sectional study involving 4801 patients with severe COPD in Spain. Clinical control was defined according to clinical impact (dyspnea grade, use of rescue treatment in last week, sputum color, and daily physical activity) and stability (exacerbations in last 3 months and patient’s perception about health status). Clinical control of COPD was alternatively evaluated with the COPD assessment test (CAT) and the presence of exacerbations in the last 3 months.
Results: According to CCC, 61.0% of patients had low clinical impact, and 41.4% showed clinical stability. Overall, 29.9% of patients had both low clinical impact and stability (controlled), whereas 70.1% showed high clinical impact and/or no clinical stability (non-controlled). COPD control was also assessed by using only the definition of CAT≤ 16 and no exacerbations in the last 3 months. Results obtained with this definition were similar to those obtained by CCC, and the concordance between both definitions was high (Kappa index = 0.698).
Conclusion: By using the CCC, approximately only one third of patients with severe COPD were considered as controlled. Physical activity, adherence to inhalers, age, post-bronchodilator FEV1, age-adjusted Charlson comorbidity index, and healthcare level were independent factors associated with COPD control.
Keywords: COPD, control, clinical, exacerbations, criteria, CAT
Introduction
Management of chronic obstructive pulmonary disease (COPD) aims at reducing the impact of the disease, minimizing symptoms, and preventing exacerbations.1,2 Current treatment strategies are based on the use of symptoms and exacerbations.3 However, several studies have raised doubts about the effectiveness of this model for determining if the patient is well controlled.4,5 Contrary to what happens in asthma the concept of control in COPD has not been completely characterized. A number of patients with COPD under treatment still experience symptoms or even exacerbations and show limitations in daily activities. Based on this, a novel dynamic definition of control in COPD has been recently proposed, combining the clinical impact or repercussion of the disease on the patient, and the clinical stability (symptomatology, exacerbations) over time. Soler-Cataluña et al,6 validated a proposal of clinical control criteria (CCC) as a tool for determining the clinical control of COPD in 265 patients. Clinical control status was associated with better outcomes (time to exacerbation, hazard rate, HR, 2.0 95% confidence interval, 95% CI, 1.5–2.7; time to hospitalization, HR 2.2, 95% CI 1.1–4.3; and survival, HR 2.7, 0.8–8.9). Control status is a dynamic concept that can be used at every clinical visit and provides complementary information that may have therapeutic implications.7 Available studies about the clinical control in patients with severe COPD (post-bronchodilator forced expiratory volume in 1 second, FEV1, <50% of predicted) are limited.3 The CLAVE (`Estudio observacional transversal para Caracterizar La EPOC grAVe en España´; Severe COPD categorization in Spain) study was designed to characterize severe COPD, and determine the level of clinical control in a large cohort of patients from Spain. Therefore, the main objective of the present study was to evaluate the control of severe COPD in Spain with CCC. The secondary objective was to identify factors potentially associated with COPD control.
Patients and Methods
Study Design and Patients
The study design was observational, cross-sectional and multicenter, involving patients with severe COPD. Inclusion criteria to participate in the CLAVE study were as follows: males and females aged ≥40 years old; active smokers or ex-smokers with a smoking history of ≥10 pack-year; diagnosis of COPD (at least 12 months before study recruitment); post-bronchodilator FEV1 < 50% of predicted (in 12 months before study recruitment); and receiving maintenance treatment (bronchodilators and/or inhaled corticosteroids and/or phosphodiesterase-4 inhibitors and/or non-drug therapies, including smoking cessation interventions, nutrition therapy, continuous positive airways pressure, pulmonary rehabilitation, home oxygen therapy, psychological support and education, and home mechanical ventilation). Exclusion criteria included: patients receiving oral corticosteroids or antibiotics due to a COPD exacerbation; or patients with a mental or other disorder who are unable to understand or adequately perform the study procedures. Investigators were responsible for the enrolment of patients on the study, and the collection of information from their medical records of routine clinical visits. Procedures were approved by the Research Ethics Committee of the Hospital Clinic of Barcelona (Spain). All patients signed the written informed consent to participate in the study. Procedures were in accordance with the Declaration of Helsinki.
Outcomes and Variables
The primary efficacy outcome was to identify the proportion of clinical control of COPD in patients with severe COPD, determined by using the previous validated CCC.6 The CCC was defined according to clinical impact and stability. Clinical impact was considered low when patients met 3 out of the 4 clinical variables: low dyspnea grade [Modified Medical Research Council (mMRC) score] ≤2,8 use of rescue treatment in last week less than 3 times, clear sputum color, and daily physical activity ≥30 min each day. Additional details about these clinical variables were described in the original validation study.6 All other patients were classified as high impact. Sputum color was considered dark (if green, yellow, brown), or clear (if white or absent of color). Physical activity performed in the last week was measured by using the Spanish version of the International Physical Activity Questionnaire (IPAQ).9 IPAQ categorizes the physical activity into 3 groups: high, moderate and low. For considering clinical stability, patients had to meet 2 variables: absence of exacerbations in the last 3 months and patient’s positive perception about their health status. The patient’s perception was measured with a 5-point Likert scale, with values ranging from −2 (very dissatisfied) to 2 (very satisfied). A positive perception included: satisfactory (value 0), satisfied (value 1), and very satisfied (value 2) results. Depending on CCC, patients were categorized into 2 groups: controlled (with low clinical impact and stability), and non-controlled (with high clinical impact and/or no clinical stability). Clinical control of COPD was alternatively evaluated with the Spanish version of the COPD assessment test (CAT) and the presence of exacerbations in the last 3 months. The CAT is an 8-item questionnaire used to determine the impact of COPD on health status.10 Each item can be scored from 0 (no limitation) to 5 (very limited). Higher total scores are indicative of worse health status. In this occasion, for considering clinical control, patients had to show a CAT score ≤16 and no exacerbations in the last 3 months. Secondary objectives included the identification of factors significantly associated with clinical control. Ex-smokers were defined as those individuals who stopped smoking at least in the last 6 months. Pack-year of smoking was calculated as the number of pack of cigarettes per day (cigarettes/20) multiplied by the number of years. Adherence to inhalers was quantified with the 12-item test of the adherence to inhalers (TAI).11 Items 1 to 10 are completed by patients and are scored between 1 and 5. Higher score are indicative of high adherence. TAI categorizes the adherence to inhalers into 3 groups: good, intermediate and poor. Comorbidities were measured with Charlson index.12 All information was collected during a unique routine visit to the physician.
Statistical Analyses
Continuous variables were expressed as mean and standard deviation (SD); while categorical ones as absolute and relative frequencies. Comparisons between clinical control groups were performed with the non-parametric Mann–Whitney U-test (for continuous variables) or Chi-square/Fisher Exact test (for categorical variables), when appropriate. Data were not normally distributed, thus non-parametric test were used. The concordance between the total number of patients classified as controlled and non-controlled compared to the total number of patients with low CAT and exacerbations was determined with the kappa coefficient. Values from kappa coefficient range from <0.2 (poor concordance), 0.21–0.40 (fair), 0.41–0.60 (moderate), 0.61–0.80 (good), and 0.81–1.0 (very good concordance).13 A backward binary logistic regression was performed to identify independent factors associated with clinical control (Odd ratio, OR; 95% CI). Factors with a significance <0.1 in the univariate analysis were introduced in the multivariate analysis. Statistical significance was established with P-value ≤0.05. All the statistical analyses were performed with SAS version 9.4 software.
Results
Between September 2017 and May 2018, a total of 4916 patients were recruited in 406 centers throughout Spain; however, 115 patients were excluded from the analysis due to meeting an exclusion criteria or lack of information/data (Figure 1).
 |
Figure 1 Flowchart of patients. FEV1, forced expiratory volume in 1 second; CAT, COPD assessment test. *Some patients could have been excluded by more than one exclusion criterion. |
Therefore, the total number of patients included in the study was 4801. Patients were predominantly male (82.2%), ex-smokers (75.7%), with a mean age of 69.6 years (SD 9.3; Table 1). The mean age-adjusted Charlson comorbidity index was 2.2 (SD 1.5). Mean post-bronchodilator FEV1 was 39.0% of predicted (SD 8.3). Main comorbidities included: diabetes (in 21.3% of patients), congestive heart failure (11.6%), peripheral vascular disease (10.4%), and myocardial infarction (10.1%).
 |
Table 1 Demographic and Clinical Characteristics of Patients, Considering Clinical Impact and Stability of COPD |
Controlled versus Uncontrolled Patients According to Criteria
Considering clinical variables defining CCC, 61.0% of patients had low clinical impact, and 41.4% showed clinical stability (Figure 2A). Overall, 70.1% of patients showed high clinical impact and/or no clinical stability (non-controlled); whereas 29.9% had lower clinical impact and stability (controlled; Figure 2B).
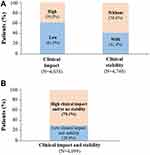 |
Figure 2 Clinical control of COPD assessed by CCC. Percentage of recruited patients considering: (A) clinical impact or clinical stability; (B) clinical impact and stability. |
Alternatively, control of COPD was evaluated with CAT and exacerbations in the last 3 months. Following this criterion, 49.4% of patients showed a CAT score ≤16, and 44.8% had no exacerbations (Figure 3A). Thus, COPD was non-controlled in 72.5% of patients and controlled in 27.5% (Figure 3B). The concordance between both definitions was good (Kappa index = 0.698).
 |
Figure 3 Clinical control of COPD assessed by CAT score and exacerbations in the last 3 months. Percentage of recruited patients considering: (A) clinical variables; (B) COPD control. |
Factors Associated with Clinical Control
Factors independently associated with clinical control were as follows (Table 2): Post-bronchodilator FEV1 (≥30% vs <30%, OR 2.0, 95% CI 1.6–2.5; P-value<0.001), age (<70 versus ≥70 years, OR 1.2, 95% CI 1.0–1.4; P-value=0.022), smoking habits (active smoker versus ex-smoker, OR 1.2, 95% CI 1.0–1.4; P-value=0.020), physical activity (high versus low, OR 5.6, 95% CI 4.6–6.9; and moderate versus low, OR 4.1, 95% CI 4 3.5–4.9; P-value<0.001 both), level of care in the hospital where attended (specialist versus primary care, OR 1.5, 95% CI 1.1–2.0; P-value=0.011), adherence to inhalers (good versus poor, OR 1.8, 95% CI 1.5–2.3; P-value<0.001; and intermediate versus poor, OR 1.7, 95% CI 1.4–2.2; P-value=0.011), and age-adjusted Charlson comorbidity index (1 vs ≥2, OR 1.3, 95% CI 1.1–1.5; P-value<0.001).
 |
Table 2 Factors Independently Associated with Clinical Control of COPD |
The strength of the association between independent factors and COPD control is graphically shown in Figure 4. Prevalence rates of categorical factors were as follows: 84.0% for FEV1 ≥30%, 48.3% for age <70 years; 24.1% for active smoker; 16.5% for high physical activity; 43.3% for moderate physical activity; 93.1% for specialist healthcare; 61.0% for good adherence to inhalers; 19.5% for intermediate adherence to inhalers; and 44.8% for age-adjusted Charlson comorbidity index <2.
Discussion
In this study, we have found that the proportion of patients with COPD and severe airflow limitation who are controlled as measured by the CCC was only 29.9%, and factors associated with this control were post-bronchodilator FEV1, age, smoking habits, physical activity, healthcare level, adherence to inhaled treatment, and age-adjusted Charlson comorbidity index. Moreover, to our knowledge this is the first time that an alternative and simplified concept of COPD control using CAT and exacerbations has been evaluated. By following these criteria, the proportion of controlled patients is 27.5%. The concordance between both definitions was high (Kappa index = 0.698). Available studies about the clinical control in patients with severe COPD are scarce. Nibber et al,14 performed a real-world study aimed at validating the new concept of control (implicating clinical impact and stability). The authors reported that 4.5% of patients with mild/moderate COPD were controlled by using the original control criteria, following clinical features; however, 21.5% by using the novel one with the CAT questionnaire. Considering clinical features, none of the patients with severe COPD were controlled; whereas the percentage of controlled patients was 8.3% using the CAT. Similarly, an international study involving 314 patients with COPD and evaluating the novel concept of control, Miravitlles et al,15 evaluated the novel concept of COPD control in terms of stability (no exacerbations or impairment in CAT scores) and low impact (low level of symptoms). Of 314 total patients, 21% were considered controlled by using these pre-specified criteria, all of them experiencing mild or moderate disease. When authors analyzed more restrictive criteria for COPD control (low impact by both clinical criteria and low CAT or clinical COPD questionnaire scores) 13% of patients were considered as controlled. By contrast, up to 32% of patients were considered as controlled when only used CAT score. It is necessary to indicate the studies by Nibber et al,14 and Miravitlles et al,15 were carried out by the same working group, using the original criteria of COPD control, previous to the validation study in 2018.6 They also incorporated patients from different origin.
Results from our present study, indicate that 29.9% of patients with severe COPD in Spain are controlled, with the proportion dropping to 27.5% if following the simplified criteria (CAT score and exacerbations). In other words, 70.1% (or 72.5%) of patients were considered non-controlled according to these criteria, suggesting significant improvement is needed in the control of patients with severe COPD. The percentage of patients who are controlled (FEV1<50%) is lower than shown in previous studies using the same validated CCC. In the validation study, Soler-Cataluña et al,6 showed that up to 50% of patients with FEV1<50% were controlled. Moreover, the proportion of controlled patients among mild/moderate cases (FEV1>50) was 64.2%. Miravitlles et al,16 in a prospective, international, multicenter study aimed at determining the 6-month prognostic value of control status in 267 patients with COPD, revealed a proportion of controlled patients of 59.3% among severe cases, and 68.5% among mild/moderate ones. Soler-Cataluña et al,7 in a prospective, multicenter, observational study designed to compare changes in control over a 3-month period with changes in risk level, GOLD stage, and clinical phenotype in 354 patients with COPD, the proportion of controlled patients among severe cases was 44.7% versus 56.1% in mild/moderate ones. Authors also showed the distribution of controlled patients at 3 months according to the GOLD A-D category. In the category A, 40 patients (90.9%) were considered as controlled by clinical evaluating, and 37 patients (84.1%) if using CAT assessment. One possible explanation for the low COPD control found in our study may derive from the type of departments involved in the study. While our present study involved diverse departments (primary medicine, internal medicine, general pneumology, and specialized COPD units), the previously cited studies were mainly carried out in specialized COPD units.6,7,16
Factors significantly associated with clinical control of COPD were in concordance with those implicated in the disease severity. Low levels of physical activity are correlated with higher decline in lung function, risk of all-cause mortality, and incidence of comorbidities, such as diabetes, cardiovascular diseases or depression.17–19 For this reason, increasing levels of physical activity should be part of any effective management and control of the disease, and improved long-term clinical outcomes.20 Indeed, physical activity has become an important outcome to healthcare professionals and COPD patients.21 There are different tools to assess the physical activity in patients, either objectively (such as pedometers, accelerometers), subjectively (questionnaires, logs, diaries), or hybrid (objective and subjective measures).22 Despite its importance, the ability to perform physical activity in patients with COPD is frequently limited, and patients often require rehabilitation programs, non-invasive ventilatory support and supplemental oxygen.23 In our study, the likelihood of achieving a low clinical impact and stability was 5.4 times higher in patients with a high level of physical activity, respectively, than those with lower. It is necessary to note that physical activity is part of the CCC definition. Nevertheless, in the definition, physical activity was only evaluated as “daily physical activity ≥ 30 min each day”; whereas in the regression model it was determined according to its level (high, moderate, or low/inactive), and using the IPAQ questionnaire (implicating more aspects than minutes).
Smoking cessation is one of the most important factors influencing the natural history of the disease.24 A number of studies have demonstrated that smoking cessation is beneficial for COPD patients, slowing the accelerated rate of lung function decline and improving survival, compared with those with active smokers.25,26 Furthermore, active smokers with COPD have poorer health-related quality of life, greater healthcare costs, impaired productivity than ex-smokers.27 It is interesting to note that, in our study, smokers had 1.2 times higher likelihood of achieving clinical control than ex-smokers. The explanation of such a result is difficult to determine. This might be associated with the cross-sectional design of the study. Active smoking could be related to a better controlled, milder disease, and can be considered a consequence, and not a cause, of good control. Another explanation might be related to a selection bias, ie severe patients that experienced fewer complications in the past or were less symptomatic, which does not force them to smoke cessation.
Bronchodilators, mainly inhaled formulations, are the mainstay for COPD control.1 They improve lung function while reducing symptoms and exacerbations.28 Nevertheless, adherence to treatments among patients with COPD is especially poor, with rates of non-adherence ranging between 50% and 80%.29,30 Non-adherence to treatment is associated with high mortality and morbidity, number of hospitalizations, and reduced quality of life.31 Patients with good adherence to inhalers showed 1.9 times higher likelihood of achieving low clinical impact and stability than those with poor adherence rates. Age is also another important factor that impacts on psychological and clinical outcomes in COPD.32 The prevalence of COPD increases with age, from 3.1% in individuals aged <40 years to 14.2% in those older than 65 years.33 Moreover, there is evidence of a strong association between aging, disease, and comorbidities (or multimorbidity in older patients).34,35 Aged patients, with advanced disease, usually show lower lung function and worse tolerance to physical activity. In our study, clinical control was inversely associated with age (1.2 times higher in patients aged<70 years than older) and comorbidities (age-adjusted Charlson comorbidity index; 0.9 times higher per greater index value). Airway obstruction, measured by FEV1 during the spirometry, is correlated with disease progression, and lung function decline may be an indicative of poor disease control.5 Among our patients, a higher value of post-bronchodilator FEV1 was significantly associated with a higher likelihood of low clinical impact and stability. Finally, healthcare level also has an influence over the clinical control of the disease. Our results indicated a higher likelihood (1.6 times) of clinical control in patients followed by a specialist than a primary care physician. This can be presumably because the study is focused on patients with severe COPD who are frequently managed by specialists in hospital setting. Overall, some of the factors associated with clinical control depend on patients (physical activity, adherence to inhalers), which become them as principal players in the management and evolution of their disease.
One of the main limitations of the study derives from the non-standardized definition of disease control, by using the CCC. Confounding bias may also be present at the time of identifying factors potentially associated with clinical control. Another limitation from the study is that data were collected from a single visit, ie the study did not have a longitudinal design. Considering all these limitations, conclusions should be made with caution. Furthermore, it is also important to take into account the concept of control in a chronic disease. In severe cases, COPD symptoms (such as dyspnea) can be controlled but they cannot disappear. Based on this, disease control should be considered as stability of the disease, rather than disappearance of symptoms, especially in severe cases.
Conclusions
By using the novel CCC, approximately only one third of patients from this Spanish cohort with severe COPD are controlled. Physical activity, age, adherence to inhalers, post-bronchodilator FEV1, age-adjusted Charlson comorbidity index, and healthcare level were independent factors associated with clinical control. Further prospective and specifically designed studies should be performed to corroborate the present observations.
Abbreviations
COPD, chronic obstructive pulmonary disease; CCC, clinical control criteria; FEV1, forced expiratory volume in 1 second; IPAQ, International Physical Activity Questionnaire; CAT, COPD assessment test; TAI, test of the adherence to inhalers; SD, standard deviation; OR, odds ratio; 95% CI, 95% confidence interval.
Data Sharing Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval and Informed Consent
Procedures were approved by the Research Ethics Committee of the Hospital Clinic of Barcelona (Spain). All patients signed the written informed consent to participle in the study. Procedures were in accordance with the Declaration of Helsinki.
Consent for Publication
Not applicable.
Acknowledgments
The authors of this study would like to thank ADKNOMA for the editorial support. Investigators in the CLAVE study are listed in the Appendix.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
The study described within the paper was sponsored by Chiesi España SAU.
Disclosure
Pere Almagro declares he has received speaking or advisory fees, or economic aid to attend congresses from Astra-Zeneca, GSK, Novartis, Chiesi, Menarini, Boehringer-Ingelheim, Ferrer, and Rovi. Borja G Cosío declares he has received speaking or advisory fees, or economic aid to attend congresses from Astra-Zeneca, GSK, Novartis, Chiesi, Mundipharma, Menarini, Sanofi, TEVA, Boehringer-Ingelheim, and Rovi. He also reports non-financial support from Separ. Juan José Soler-Cataluña has received speaker fees from AstraZeneca, Bial, Boehringer Ingelheim, Chiesi, Esteve, Ferrer, GSK, Menarini, Mundipharma, Novartis, Rovi, and consulting fees from AstraZeneca, Boehringer Ingelheim, Chiesi, GSK, Mundipharma and Novartis. Diego Gonzalez-Segura is an employee of Chiesi España SAU, the sponsor of the study. The authors report no other conflicts of interest in this work.
References
1. Vogelmeier CF, Criner GJ, Martínez FJ, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease 2017 report: GOLD executive summary. Arch Bronconeumol. 2017;53(3):128–149. doi:10.1016/j.arbres.2017.02.001
2. Wedzicha JA, Calverley PMA, Albert RK, et al. Prevention of COPD exacerbations: a european respiratory society/american thoracic society guideline. Eur Respir J. 2017;50(3):1602265. doi:10.1183/13993003.02265-2016
3. Lopez-Campos JL, Bustamante V, Muñoz X, Barreiro E. Moving towards patient-centered medicine for COPD management: multidimensional approaches versus phenotype-based medicine – A critical view. COPD. 2014;11:591–602. doi:10.3109/15412555.2014.898035
4. Schlecht NF, Schwartzman K, Bourbeau J. Dyspnea as clinical indicator in patients with chronic obstructive pulmonary disease. Chron Respir Dis. 2005;2(4):183–191. doi:10.1191/1479972305cd079oa
5. Guimarães M, Bugalho A, Oliveira AS, Moita J, Marques A. COPD control: can a consensus be found? Revista Portuguesa De Pneumologia. 2009;18(114):167–176. doi:10.1016/j.rppnen.2016.01.004
6. Soler-Cataluña JJ, Marzo M, Catalán P, Miralles C, Alcazar B, Miravitlles M. Validation of clinical control in COPD as a new tool for optimizing treatment. Int J Chron Obstruct Pulmon Dis. 2018;13:3719–3731. doi:10.2147/COPD.S178149
7. Soler-Cataluña JJ, Alcazar B, Marzo M, Pérez J, Miravitlles M. Evaluation of changes in control status in COPD: an opportunity for early intervention. Chest. 2020;157:1138–1146. doi:10.1016/j.chest.2019.11.004
8. Bestall JC, Paul EA, Garrod R, Garnham R, Jones PW, Wedzicha JA. Usefulness of the medical research council (MRC) dyspnoea scale as a measure of disability in patients with chronic obstructive pulmonary disease. Thorax. 1999;54(7):581–586. doi:10.1136/thx.54.7.581
9. Hagströmer M, Oja P, Sjöström M. The International Physical Activity Questionnaire (IPAQ): a study of concurrent and construct validity. Public Health Nutr. 2006;9(6):755–762. doi:10.1079/PHN2005898
10. Jones PW, Harding G, Berry P, Wiklund I, Chen WH, Kline Leidy N. Development and first validation of the COPD assessment test. Eur Respir J. 2009;34(3):648–654. doi:10.1183/09031936.00102509
11. Plaza V, Fernández-Rodríguez C, Melero C, et al. Validation of the ‘Test of the Adherence to Inhalers’ (TAI) for asthma and COPD patients. J Aerosol Med Pulm Drug Deliv. 2016;29(2):142–152. doi:10.1089/jamp.2015.1212
12. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–383. doi:10.1016/0021-9681(87)90171-8
13. Altman DG. Practical Statistics for Medical Research. London: Chapman and Hall; 1991.
14. Nibber A, Chisholm A, Soler-Cataluña JJ, Alcazar B, Price D, Miravitlles M. Validating the concept of COPD control: A real-world cohort study from the United Kingdom. COPD. 2017;14(5):504–512. doi:10.1080/15412555.2017.1350154
15. Miravitlles M, Sliwinski P, Rhee CK, et al. Evaluation of criteria for clinical control in a prospective, international, multicenter study of patients with COPD. Respir Med. 2018;136:8–14. doi:10.1016/j.rmed.2018.01.019
16. Miravitlles M, Sliwinski P, Rhee CK, et al. Changes in control status of COPD over time and their consequences: a prospective international study. Arch Bronconeumol. 2020;
17. Garcia-Aymerich J, Lange P, Benet M, Schnohr P, Antó JM. Regular physical activity modifies smoking-related lung function decline and reduces risk of chronic obstructive pulmonary disease: a population-based cohort study. Am J Respir Crit Care Med. 2007;175(5):458–463. doi:10.1164/rccm.200607-896OC
18. Waschki B, Kirsten A, Holz O, et al. Physical activity is the strongest predictor of all-cause mortality in patients with COPD: a prospective cohort study. Chest. 2011;140(2):331–342. doi:10.1378/chest.10-2521
19. Watz H, Pitta F, Rochester CL, et al. An official European Respiratory Society statement on physical activity in COPD. Eur Respir J. 2014;44(6):1521–1537. doi:10.1183/09031936.00046814
20. Troosters T, van der Molen T, Polkey M, et al. Improving physical activity in COPD: towards a new paradigm. Respir Res. 2013;14:115. doi:10.1186/1465-9921-14-115
21. Gimeno-Santos E, Raste Y, Demeyer H, et al. The PROactive instruments to measure physical activity in patients with chronic obstructive pulmonary disease. Eur Respir J. 2015;46(4):988–1000. doi:10.1183/09031936.00183014
22. Gore S, Chindam T, Goldberg A, Huang MH, Shoemaker M, Blackwood J. Reliability and validity of patient-reported, rater-based, and hybrid physical activity assessments in COPD: A systematic review. COPD. 2020;1–11.
23. Moga AM, de Marchie M, Saey D, Spahija J. Mechanisms of non-pharmacologic adjunct therapies used during exercise in COPD. Respir Med. 2012;106(5):614–626. doi:10.1016/j.rmed.2012.01.006
24. Bai JW, Chen XX, Liu S, Yu L. Xu JF1. Smoking cessation affects the natural history of COPD. Int J Chron Obstruct Pulmon Dis. 2017;12:3323–3328. doi:10.2147/COPD.S150243
25. Godtfredsen NS, Lam TH, Hansel TT, et al. COPD-related morbidity and mortality after smoking cessation: status of the evidence. Eur Respir J. 2008;32(4):844–853. doi:10.1183/09031936.00160007
26. Zamarro García C, Bernabé Barrios MJ, Santamaría Rodríguez B, Rodríguez Hermosa JL. Smoking in COPD. Arch Bronconeumol. 2011;47(Suppl 8):3–9. doi:10.1016/S0300-2896(11)70059-X
27. Galaznik A, Chapnick J, Vietri J, Tripathi S, Zou KH, Makinson G. Burden of smoking on quality of life in patients with chronic obstructive pulmonary disease. Expert Rev Pharmacoecon Outcomes Res. 2013;13(6):853–860. doi:10.1586/14737167.2013.842128
28. Miravitlles M, Baek S, Vithlani V, Lad R. Optimal bronchodilation for COPD patients: are all long-acting β₂-agonist/long-acting muscarinic antagonists the same? Tuberc Respir Dis (Seoul). 2018;81(3):198–215. doi:10.4046/trd.2018.0040
29. Humenberger M, Horner A, Labek A, et al. Adherence to inhaled therapy and its impact on chronic obstructive pulmonary disease (COPD). BMC Pulm Med. 2018;18(1):163. doi:10.1186/s12890-018-0724-3
30. George M. Adherence in asthma and COPD: new strategies for an old problem. Respir Care. 2018;63(6):818–831. doi:10.4187/respcare.05905
31. Vestbo J, Anderson JA, Calverley PM, et al. Adherence to inhaled therapy, mortality and hospital admission in COPD. Thorax. 2009;64(11):939–943. doi:10.1136/thx.2009.113662
32. Holm KE, Plaufcan MR, Ford DW, et al. The impact of age on outcomes in chronic obstructive pulmonary disease differs by relationship status. J Behav Med. 2014;37(4):654–663. doi:10.1007/s10865-013-9516-7
33. Raherison C, Girodet PO. Epidemiology of COPD. Eur Respir Rev. 2009;18(114):213–221. doi:10.1183/09059180.00003609
34. Vaz Fragoso CA, Concato J, McAvay G, et al. Chronic obstructive pulmonary disease in older persons: A comparison of two spirometric definitions. Respir Med. 2010;104(8):1189–1196. doi:10.1016/j.rmed.2009.10.030
35. Parulekar AD, Martinez C, Tsai CL, et al. Examining the effects of age on health outcomes of chronic obstructive pulmonary disease: results from the genetic epidemiology of chronic obstructive pulmonary disease study and evaluation of chronic obstructive pulmonary disease longitudinally to identify predictive surrogate endpoints cohorts. J Am Med Dir Assoc. 2017;18(12):1063–1068. doi:10.1016/j.jamda.2017.09.028
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

