Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
Clinical Characteristics and Outcomes of Patients with Asthma–COPD Overlap in Japanese Patients with COPD
Authors Kobayashi S , Hanagama M, Ishida M, Ono M, Sato H, Yamanda S , Yanai M
Received 15 August 2020
Accepted for publication 26 October 2020
Published 12 November 2020 Volume 2020:15 Pages 2923—2929
DOI https://doi.org/10.2147/COPD.S276314
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Russell
Seiichi Kobayashi,1 Masakazu Hanagama,1 Masatsugu Ishida,1 Manabu Ono,1 Hikari Sato,1 Shinsuke Yamanda,1,2 Masaru Yanai1
1Department of Respiratory Medicine, Japanese Red Cross Ishinomaki Hospital, Ishinomaki, Miyagi, Japan; 2Department of Pulmonary Medicine, Sendai Kosei Hospital, Sendai, Miyagi, Japan
Correspondence: Seiichi Kobayashi
Department of Respiratory Medicine, Japanese Red Cross Ishinomaki Hospital, 71 Nishimichishita, Hebita, Ishinomaki 986-8522, Japan
Tel +81 225 21 7220
Fax +81 225 96 0122
Email [email protected]
Purpose: Asthma–COPD overlap (ACO) has been reported as an association with a lower quality of life, frequent exacerbations, and higher mortality than those with COPD alone. However, clinical characteristics and outcomes of ACO remain controversial.
Patients and Methods: We conducted a prospective observational study analyzing data of patients with stable COPD enrolled from the Ishinomaki COPD Network Registry. Patients with features of asthma who had a history of respiratory symptoms that vary over time and intensity, together with documented variable expiratory airflow limitation, were identified, and then defined as having ACO. The characteristics, frequency of exacerbations, and mortality during the 3-year follow-up were compared between patients with ACO and patients with COPD alone.
Results: Among 387 patients with COPD, 41 (10.6%) were identified as having ACO. Patients with ACO tended to be younger, have higher BMI, have a shorter smoking history, and use more respiratory medications, especially inhaled corticosteroids. Inflammatory biomarkers including fractional exhaled nitric oxide, blood eosinophil count, total immunoglobulin E (IgE) level, and presence of antigen-specific IgE were significantly higher in patients with ACO than in those with COPD alone. Lung function, mMRC score, CAT score, and comorbidity index were not different between the groups. The annual rate of all exacerbations and severe exacerbations required hospital admission were not different between ACO and COPD alone (0.20 vs 0.14, 0.12 vs 0.10, events per person, respectively). Mortality was significantly higher in patients with COPD alone compared with those with ACO during the study period (P=0.037).
Conclusion: The results of our study indicate that ACO is not associated with poor clinical features nor outcomes in an outpatient COPD cohort receiving appropriate treatment.
Keywords: asthma, asthma–COPD overlap, COPD, exacerbations, mortality
Introduction
Asthma and chronic obstructive pulmonary disease (COPD) are common and potentially serious chronic respiratory diseases that could induce substantial economic and social burden.1,2 These diseases show different features and have been regarded as two distinct entities. However, they are now recognized as heterogeneous and often overlap.3,4 The Global Initiative for Asthma (GINA) and the Global Initiative for Chronic Obstructive Lung Disease (GOLD) issued a joint document describing asthma–COPD overlap (ACO), which characterized ACO as persistent airflow limitation with several features usually associated with asthma and several features usually associated with COPD.1 ACO have been recognized to be associated with a significantly lower quality of life,5,6 frequent exacerbations,5,7–9 increased hospitalization,5,9 and higher mortality3,10 than those with asthma or COPD alone. However, clinical features and outcomes of ACO remain controversial in the real-world COPD population.
We conducted a prospective observational study to investigate the prevalence and clinical characteristics of ACO in community-dwelling patients with COPD. We also evaluated the exacerbation rates and mortality of patients affected by ACO during a 3-year follow-up.
Patients and Methods
Study Design
We analyzed prospectively collected data from the Ishinomaki COPD Network (ICON) Registry.11,12 Baseline data were obtained at consecutive scheduled patient visits or newly patient registration between May 2015 and June 2016, and the participants were followed for 3 years.
ICON is a regional medical cooperation system that aims to provide comprehensive care for patients with COPD with a multicenter interprofessional collaboration including respiratory specialists, general practitioners, nurses, therapists, pharmacists, and other healthcare providers in Ishinomaki and surrounding cities in Japan.11,12 Patients are registered in ICON, then receive the standard care in general practice clinics according to the guidelines,2,13 and undergo scheduled examinations and education at the Japanese Red Cross Ishinomaki Hospital (a 452-bed tertiary community hospital) every 6–12 months. The patient education program included early recognition of exacerbation and a written action plan for an exacerbation using a self-management diary. Patients were prescribed with short-acting bronchodilators, but not oral corticosteroids or antibiotics, for self-administration during exacerbations. Patients with an exacerbation of COPD were treated by general practitioners at first and would be referred to the Japanese Red Cross Ishinomaki Hospital if necessary.
This research is part of an ongoing COPD cohort study registered with the UMIN Clinical Trials Registry (identifier UMIN000017376). The study was conducted in accordance with the ethical principles laid out in the Declaration of Helsinki, and was approved by the Ethics Committee at the Japanese Red Cross Ishinomaki Hospital (approval number: 12–14-1). All patients provided written informed consent.
Patients
Patients were included if they were aged ≥40 years with a smoking history of at least 10 pack-years and had been diagnosed with COPD according to the GOLD criteria.2 Persistent airflow limitation, defined as post-bronchodilator forced expiratory volume in 1 s (FEV1)/forced vital capacity (FVC) <0.7, was confirmed by spirometry. Exclusion criteria included chronic bronchitis or emphysema without airflow limitation, history of lung resection, tuberculosis, and exacerbation of COPD in the 4 weeks preceding data collection.
Clinical and Physiologic Measurements
The sociodemographic characteristics and smoking history of each patient were recorded at baseline. Body mass index (BMI) was calculated as the body weight divided by the square of the body height (kg/m2). Dyspnea was evaluated using the modified Medical Research Council dyspnea questionnaire (mMRC) score.2,13 Health status impairment of COPD was measured using the COPD Assessment Test (CAT), which is an 8-item questionnaire with possible scores ranging from 0 to 40 where higher scores indicate the reduced quality of life.14,15 Comorbidities were evaluated using the Charlson comorbidity index.16
Spirometry was performed by a trained technician following published guidelines,17 while the patients were in a stable condition. The severity of airflow limitation was evaluated as the GOLD staging.2 The ABCD assessment was performed according to the 2017 GOLD report.2,12
Biomarker Measurements
Measurement of inflammatory biomarkers were conducted as described previously.11
Fractional exhaled nitric oxide (FENO) level was measured using the NIOX MINO device (Aerocrine, Morrisville, NC, USA) prior to performing spirometry according to the guidelines.18
Blood samples were obtained to determine blood eosinophil count, and total serum immunoglobulin E (IgE) levels on the same day as FENO measurement. The cut-off value of total serum IgE level was set at 173 IU/mL according to the reference range at the hospital. The presence of antigen-specific IgE was determined by using the ImmunoCAP Phadiatop test (Thermo Fisher Scientific, Uppsala, Sweden), an in vitro assay for antigen-specific IgE antibodies to common inhalant allergens.
Diagnosis of ACO
In this study participants fulfilled the features of COPD, such as age ≥40 years, confirmed persistent airflow limitation by spirometry, and exposure to cigarette smoke. COPD patients with features of asthma who had a history of respiratory symptoms together with documented variable expiratory airflow limitation were identified as ACO. A history of respiratory symptoms, such as wheezing, shortness of breath, chest tightness, and cough, that vary over time and intensity, were evaluated based on medical records. Variable expiratory airflow limitation, which defined as a post-bronchodilator increase in FEV1 of >12% and >200 mL from baseline, or an increase in FEV1 by >12% and >200 mL (or peak expiratory flow by >20%) from baseline after 4 weeks of anti-inflammatory treatment, were assessed prior to biomarker measurement.
Longitudinal Assessment
The exacerbations during the 3-year follow-up were evaluated at the scheduled annual visit by direct patient interview, self-management diaries completed by the patient or caregivers, referral letters from general practitioners, and review of medical records. Exacerbations were defined as the use of antibiotics and/or systemic corticosteroids for the worsening of respiratory symptoms without evidence that would suggest an alternate diagnosis.2 Mild exacerbations treated with short-acting bronchodilators only were excluded from the study.
All-cause mortality was assessed by telephone interview, referral letters from general practitioners, and review of medical records during the study period.
Statistical Analysis
The data are shown as the median with interquartile range or mean ± standard deviation (SD) unless otherwise specified. Differences between two groups were assessed using the Student’s t-test or Mann–Whitney U-test. Odds ratios for exacerbations were estimated by logistic regression analysis. Survival analysis was performed using the Kapkan–Meier method and Log Rank test to compare the survival curve between the groups.
All statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria).19 P-values less than 0.05 were considered statistically significant.
Results
Characteristics of the Study Patients
A total of 387 eligible patients (354 men, 33 women; median age, 75 years) were identified. Among these, 53 (13.7%) had a history of variable respiratory symptoms, 85 (22.0%) had variable expiratory airflow limitation, and 41 (10.6%) had both features and were identified as having ACO. Among the 41 patients with ACO, none had childhood respiratory symptoms, but 2 (4.9%) displayed symptoms before the age of 40 years. Four (9.8%) patients showed an airflow improvement with bronchodilator in FEV1 of >12% and >400 mL from the baseline.
Clinical Characteristics of Patients with ACO
Characteristics between patients with and without ACO are shown in Table 1. Patients with ACO tended to be younger (P = 0.003) with a higher BMI (P = 0.016) and less smoking history (P = 0.027). Rhinitis was observed more frequently in ACO compared with COPD alone. Lung function (FEV1, %FEV1, and FVC), dyspnea (mMRC score), symptoms (CAT score), and comorbidity index were not different between the groups. Patients with ACO tended to use more respiratory medications, especially inhaled corticosteroids (ICS) compared to patients with COPD alone (P < 0.001).
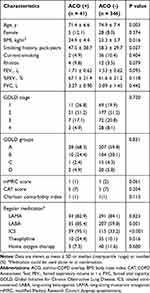 |
Table 1 Characteristics of the Patients with and without ACO |
Inflammatory biomarkers including FENO, eosinophil count, total IgE level, and presence of allergen-specific IgE were significantly higher in patients with ACO than in those with COPD alone (Table 2).
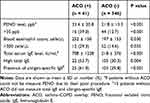 |
Table 2 Inflammatory Biomarkers in Patients with and without ACO |
Clinical Outcomes of the Patients with ACO
Among 387 patients, 385 (99.5%) completed the 1-year follow-up. Exacerbations were observed in 22.5% (9/40) and 11.6% (40/345) of the ACO and non-ACO group during the study period, respectively (P=0.087). The frequency of exacerbations was not different between the ACO and non-ACO groups (0.27 ± 0.55 vs 0.14 ± 0.43 events per person, P=0.078). Exacerbations that required hospital admissions were observed in 12.5% (5/40) and 8.1% (28/345) of the ACO and non-ACO groups, respectively (P=0.522). The frequency of exacerbations that required hospital admissions were not different between the ACO and non-ACO groups (0.15 ± 0.42 vs 0.095 ± 0.34 events per person, respectively; P=0.353).
Among 387 patients, 359 (92.8%) completed the 3-year follow-up. Exacerbations were observed in 28.9% (11/38) and 28.0% (90/321) of the ACO and non-ACO group, respectively, during the study period (P=0.941, Table 3). The annual rate of exacerbations was not different between the ACO and non-ACO groups (0.20 ± 0.37 vs 0.14 ± 0.31 events per person per year, P=0.338). Exacerbations that required hospital admissions were observed in 18.4% (7/38) and 20.2% (65/321) of the ACO and non-ACO groups, respectively (P=0.958, Table 3). The annual rate of exacerbations that required hospital admissions were not different between the ACO and non-ACO groups (0.12± 0.31 vs 0.10 ± 0.26 events per person per year, respectively; P=0.732). Patients with ACO had an odds ratio of 1.05 (95% confidence interval [CI] 0.50–2.20) for the risk of all exacerbations and 0.89 (95% CI 0.38–2.11) for risk of hospital admission relative to COPD alone. Patients with ACO had an odds ratio of 1.05 (95% confidence interval [CI] 0.45–2.20) for the risk of all exacerbations and 0.89 (95% CI 0.38–2.11) for risk of hospital admission relative to COPD alone. Patients with ACO had an odds ratio of 1.27 (95% confidence interval [CI] 0.59–2.77) for the risk of all exacerbations and 1.29 (95% CI 0.52–3.23) for risk of hospital admission relative to COPD alone even adjusted age, smoking history, and BMI.
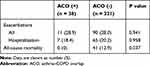 |
Table 3 Outcomes in Patients with and without ACO |
All-cause mortality was 0% (0/38) in ACO group and 12.9% (41/319) in non-ACO during the 3-year study period (Table 3). Mortality was significantly higher in non-ACO group compared with ACO group (P=0.037). The estimated survival rates at 3 years were significant worse in COPD alone compared with ACO (hazard ratio, 0.874; 95% CI, 0.832–0.906).
Discussion
In this study, we have shown the prevalence, clinical characteristics and outcomes of ACO in community-dwelling Japanese patients with COPD.
The results of our study show that approximately 10% of community-dwelling patients with COPD fulfilled the ACO phenotype. In epidemiological studies, reported prevalence rate for ACO have ranged between 9% and 55%, with variation by sex and age, which reflects the different criteria that have been used by different investigators for diagnosing asthma and COPD.1 Our findings are consistent with a previous report by Inoue and colleagues that the prevalence of ACO was 9.2% in outpatient Japanese patients with COPD,20 and are similar to reports from Europe7,21 and the US.8
The results of our study demonstrated that patients with ACO tended to be younger, had higher BMI, which is consistent with the previous systematic review.5 Recent studies described that FENO levels are increased in a subset of population with COPD,22,23 whereas blood and sputum eosinophil counts are increased in COPD patients with asthmatic symptoms.24 We have previously demonstrated that the biomarkers related with asthma including FENO, eosinophil count, and IgE could provide additional diagnostic information on ACO.11 The findings of the present study are concurrent with the results from these studies.
To date, ACO has been considered to be associated with poorer clinical features and outcomes compared to those with asthma or COPD alone.5–10 However, the findings in our study did not indicate that the patients with ACO displayed worse lung function nor increased dyspnea and symptoms than the COPD patients without ACO. Furthermore, ACO was not associated with more frequent exacerbations nor higher mortality than those with COPD alone. These results suggest that asthma overlap may not be associated with poorer clinical features nor outcomes in patients with COPD receiving appropriate treatment. Recent studies reported that exacerbation rates12,20,21 and mortality21,25,26 were not different between COPD patients with or without ACO. Our findings are consistent with those of recent studies. Previous studies have attempted to identify the ACO phenotype by using different criteria, which could explain the discrepancies of outcomes among previous studies.27 In addition, the inappropriate treatment, such as a lack of consideration to ICS,28,29 could result in the disagreement of outcomes in ACO among the studies. The presence of asthma-like features such as bronchodilator reversibility, high blood eosinophils and atopy have been reported as better clinical outcomes in patients with COPD.30 High FENO level31 and blood eosinophilia32,33 have been identified as surrogate markers in response to steroid treatments in patients with COPD, suggesting that inhaled corticosteroid treatment may be beneficial in COPD patients with eosinophilic inflammation. The findings of these study support the view that ACO may not be associated with poor outcomes if receiving appropriate treatment.
The primary strengths of our study are its prospective observational design and inclusion of community-dwelling patients with COPD, reflecting the real-world COPD population in Japan.
A major limitation in this study is that the results were not confirmed by an external validation cohort. The results of our study cannot be generalized or applied directly to a different setting, such as an asthma cohort. The number of patients between ACO and COPD only was obviously different in this study. This might lead to be a potential bias of comparison between the groups. In addition, we could not estimate the effect of smoking on mortality in ACO because of no mortality events in patients with ACO. The patients with ACO had less smoking history compared with those with COPD alone in this study. Smoking cessation is an essential treatment of COPD to improve pulmonary function and symptoms.34–36 Impact of smoking history may be affected on the mortality in the study participants.
Conclusion
We have shown the prevalence and clinical characteristics of ACO in a cohort of Japanese patients with COPD. The results of this study provide evidence that the presence of asthma overlap may not be associated with poor clinical features nor outcomes in patients with COPD receiving appropriate treatment. The results of our study encourage better recognition of ACO and may guide appropriate treatment in patients with ACO.
Data Sharing Statement
Data for the individual study participants are not available because of the privacy policy of ICON, which is the local registry of patients with COPD enrolled from Ishinomaki and surrounding cities in Japan.
Acknowledgments
We would like to thank Fumi Chiba, Natsumi Kagabu, Kazue Morozumi, Keiko Miyamoto (Outpatient Clinic of Japanese Red Cross Ishinomaki Hospital, Ishinomaki, Japan) for their help with data collection and management. We are also grateful to the healthcare professionals affiliated with ICON for their kind help and cooperation.
Disclosure
Dr Seiichi Kobayashi reports personal fees from Boehringer Ingelheim, Bristol-Myers Squibb, TEIJIN PHARMA LIMITED., and Kyowa Kirin Co., Ltd., grants from GlaxoSmithKline, and grants and personal fees from AstraZeneca and Novartis, outside the submitted work. The authors report no other potential conflicts of interest in this work.
References
1. Global Initiative for Asthma. Global strategy for asthma management and prevention; 2020. Available from: http://www.ginasthma.org/.
2. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management and prevention of COPD; 2020. Available from: http://www.goldcopd.org/.
3. Gibson PG, Simpson JS. The overlap syndrome of asthma and COPD: what are its features and how important is it? Thorax. 2009;64(68):728–735. doi:10.1136/thx.2008.108027
4. Zeki AA, Schivo M, Chan A, et al. The asthma-COPD overlap syndrome: a common clinical problem in the elderly. J Allergy. 2011;2011:861–926. doi:10.1155/2011/861926
5. Alshabanat A, Zafari Z, Albanyan O, Dairi M, FitzGerald JM, Short P. Asthma and COPD overlap syndrome (ACOS): a systematic review and meta analysis. PLoS One. 2015;10(9):e0136065. doi:10.1371/journal.pone.0136065
6. Kauppi P, Kupiainen H, Lindqvist A, et al. Overlap syndrome of asthma and COPD predicts low quality of life. J Asthma. 2011;48(3):279–285. doi:10.3109/02770903.2011.555576
7. Miravitlles M, Soriano JB, Ancochea J, et al. Characterisation of the overlap COPD-asthma phenotype. Focus on physical activity and health status. Respir Med. 2013;107(7):1053–1060. doi:10.1016/j.rmed.2013.03.007
8. Hardin M, Silverman EK, Barr RG, et al. The clinical features of the overlap between COPD and asthma. Respir Res. 2011;12(1):127. doi:10.1186/1465-9921-12-127
9. Menezes AM, Montes de Oca M, Pérez-Padilla R, et al. Increased risk of exacerbation and hospitalization in subjects with an overlap phenotype: COPD-asthma. Chest. 2014;145(2):297–304. doi:10.1378/chest.13-0622
10. Kendzerska T, Sadatsafavi M, Aaron SD, et al. Concurrent physician-diagnosed asthma and chronic obstructive pulmonary disease: a population study of prevalence, incidence and mortality. PLoS One. 2017;12(3):e0173830.
11. Kobayashi S, Hanagama M, Yamanda S, et al. Inflammatory biomarkers in asthma-COPD overlap syndrome. Int J Chron Obstruct Pulmon Dis. 2016;11:2117–2123. doi:10.2147/COPD.S113647
12. Kobayashi S, Hanagama M, Ishida M, et al. Clinical characteristics and outcomes in Japanese patients with COPD according to the 2017 GOLD classification: the Ishinomaki COPD network registry. Int J Chron Obstruct Pulmon Dis. 2018;13:3947–3955. doi:10.2147/COPD.S182905
13. The Japanese Respiratory Society. Guidelines for the Diagnosis and Treatment of COPD (Chronic Obstructive Pulmonary Disease).
14. Jones PW, Harding G, Berry P, et al. Development and first validation of the COPD assessment test. Eur Respir J. 2009;34(3):648–654. doi:10.1183/09031936.00102509
15. Tsuda T, Suematsu R, Kamohara K, et al. Development of the Japanese version of the COPD assessment test. Respir Investig. 2012;50(2):34–39. doi:10.1016/j.resinv.2012.05.003
16. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383. doi:10.1016/0021-9681(87)90171-8
17. Miller MR, Hankinson J, Brusasco V, et al. Standardisation of spirometry. Eur Respir J. 2005;26(2):319–338. doi:10.1183/09031936.05.00034805
18. Dweik RA, Boggs PB, Erzurum SC, et al. An official ATS clinical practice guideline: interpretation of exhaled nitric oxide levels (FENO) for clinical applications. Am J Respir Crit Care Med. 2011;184(5):602–615. doi:10.1164/rccm.9120-11ST
19. Kanda Y. Investigation of the freely-available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013;48(3):452–458. doi:10.1038/bmt.2012.244
20. Inoue H, Nagase T, Morita S, et al. Prevalence and characteristics of asthma–COPD overlap syndrome identified by a stepwise approach. Int J Chron Obstruct Pulmon Dis. 2017;12:1803–1810. doi:10.2147/COPD.S133859
21. Cosio BG, Soriano JB, López-Campos JL, et al. Defining the asthma-COPD overlap syndrome in a COPD cohort. Chest. 2016;149(1):45–52. doi:10.1378/chest.15-1055
22. Donohue JF, Herje N, Crater G, et al. Characterization of airway inflammation in patients with COPD using fractional exhaled nitric oxide levels: a pilot study. Int J Chron Obstruct Pulmon Dis. 2014;9:745–751. doi:10.2147/COPD.S44552
23. Tamada T, Sugiura H, Takahashi T, et al. Biomarker-based detection of asthma-COPD overlap syndrome in COPD populations. Int J Chron Obstruct Pulmon Dis. 2015;10:2169–2176. doi:10.2147/COPD.S88274
24. Kitaguchi Y, Komatsu Y, Fujimoto K, et al. Sputum eosinophilia can predict responsiveness to inhaled corticosteroid treatment in patients with overlap syndrome of COPD and asthma. Int J Chron Obstruct Pulmon Dis. 2012;7:283–289.
25. Fu JJ, Gibson PG, Simpson JL, et al. Longitudinal changes in clinical outcomes in older patients with asthma, COPD and asthma-COPD overlap syndrome. Respiration. 2014;87(1):63–74. doi:10.1159/000352053
26. Yamauchi Y, Yasunaga H, Matsui H, et al. Comparison of in-hospital mortality in patients with COPD, asthma and asthma-COPD overlap exacerbations. Respirology. 2015;20(6):940–946. doi:10.1111/resp.12556
27. Tho NV, Park HY, Nakano Y. Asthma-COPD overlap syndrome (ACOS): a diagnostic challenge. Respirology. 2016;21(3):410–418. doi:10.1111/resp.12653
28. Gershon AS, Campitelli MA, Croxford R, et al. Combination long-acting β-agonists and inhaled corticosteroids compared with long-acting β-agonists alone in older adults with chronic obstructive pulmonary disease. JAMA. 2014;312(11):1114–1121. doi:10.1001/jama.2014.11432
29. Kendzerska T, Aaron SD, To T, et al. Effectiveness and safety of inhaled corticosteroids in older individuals with chronic obstructive pulmonary disease and/or asthma. A population study. Ann Am Thorac Soc. 2019;16(10):1252–1262. doi:10.1513/AnnalsATS.201902-126OC
30. Suzuki M, Makita H, Konno S, et al. Asthma-like features and clinical course of chronic obstructive pulmonary disease. An analysis from the Hokkaido COPD cohort study. Am J Respir Crit Care Med. 2016;194(11):1358–1365. doi:10.1164/rccm.201602-0353OC
31. Zietkowski Z, Kucharewicz I, Bodzenta-Lukaszyk A. The influence of inhaled corticosteroids on exhaled nitric oxide in stable chronic obstructive pulmonary disease. Respir Med. 2005;99(7):816–824. doi:10.1016/j.rmed.2004.12.008
32. Chanez P, Vignola AM, O’Shaugnessy T, et al. Corticosteroid reversibility in COPD is related to features of asthma. Am J Respir Crit Care Med. 1997;155(5):1529–1534. doi:10.1164/ajrccm.155.5.9154853
33. Pascoe S, Locantore N, Dransfield MT, et al. Blood eosinophil counts, exacerbations, and response to the addition of inhaled fluticasone furoate to vilanterol in patients with chronic obstructive pulmonary disease: a secondary analysis of data from two parallel randomized controlled trials. Lancet Respir Med. 2015;3(6):435–442. doi:10.1016/S2213-2600(15)00106-X
34. Laniado-Laborín R. Smoking and chronic obstructive pulmonary disease (COPD). Parallel epidemics of the 21 century. Int J Environ Res Public Health. 2009;6(1):209–224. doi:10.3390/ijerph6010209
35. Pezzuto A, Carico E. Effectiveness of smoking cessation in smokers with COPD and nocturnal oxygen desaturation: functional analysis. Clin Respir J. 2020;14(1):29–34. doi:10.1111/crj.13096
36. Pezzuto A, D’Ascanio M, Grieco A, Ricci A. Functional benefit of smoking cessation in severe COPD patients undergoing bronchial valve implantation. Eur J Intern Med. 2019;68:55–59. doi:10.1016/j.ejim.2019.07.032
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
