Back to Journals » Research Reports in Clinical Cardiology » Volume 13
Clinical and Socio-Demographic Profiles of Patients Seen with Rheumatic Heart Disease in a Cardiac Clinic of a Tertiary Hospital in Ghana
Authors Owusu IK , Acheamfour-Akowuah E, Wiafe YA
Received 19 July 2022
Accepted for publication 30 September 2022
Published 18 October 2022 Volume 2022:13 Pages 85—93
DOI https://doi.org/10.2147/RRCC.S372472
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Kones
Isaac Kofi Owusu,1,2 Emmanuel Acheamfour-Akowuah,1,2 Yaw Amo Wiafe3
1Department of Medicine, School of Medicine and Dentistry, College of Health Sciences, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana; 2Directorate of Medicine, Komfo Anokye Teaching Hospital, Kumasi, Ghana; 3Department of Medical Diagnostics, College of Health Sciences, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana
Correspondence: Isaac Kofi Owusu, Email [email protected]
Background: Rheumatic heart disease (RHD) is mainly among populations in low-to-middle income nations. The burden of this disease on communities and even hospital settings is little understood. This study aimed at analysing the clinical and socio-demographic features of RHD patients in our adult outpatient cardiac department over a 3-year period.
Methods: The study was conducted at the Komfo Anokye Teaching Hospital’s Adult Cardiac Clinic in Kumasi, Ghana. Medical records of patients seen at the adult cardiac clinic between 2016 and 2019 with the diagnosis of rheumatic heart disease were examined. The data were captured using EPI-DATA (version 3.1). Stata 14 was used in analysing the mean (standard deviation) and median of continuous variables (inter-quartile range). The frequency and percentages were used to describe categorical data. The Chi-square test was used to establish the statistical significance of a relationship, with a p-value of less than 0.05.
Results: A total of 140 RHD cases were included in the study. The average age of patients was 39.7 years. Majority of the participants (82.9%) were from rural areas. Palpitation was the commonest symptom (54.29%), followed by chest pains (36.43%), dyspnoea (25.00%), and dizziness (24.29%). Over seventy-seven percent (77.14%) of the patients had moderate-to-severe rheumatic heart disease. The commonest valvular lesion was isolated mitral regurgitation (56.43%), followed by pure aortic regurgitation (30.71%), and isolated tricuspid regurgitation (12.14%).
Conclusion: RHD occurred more commonly in the third decade of life. Rural participants, who are primarily females, were the most affected. Efforts to reduce prevalence rates should be stepped up.
Keywords: rheumatic heart disease, acute rheumatic fever, heart failure, mitral regurgitation, echocardiography
Introduction
Rheumatic heart disease (RHD) has been virtually eradicated in most industrialized, high-income countries, and in many parts of the world where human development indices and healthcare systems have improved.1 However, it is still a prominent cause of cardiovascular morbidity and death in the first fifteen years of life in many low and middle-income countries.1–5 RHD currently affects 40.5 million people in the world, who lose 11.5 million years of life to disability, with RHD-related 305,000 deaths recorded each year, mostly occurring in low-income countries.6–8
Acute rheumatic fever (ARF), a multisystemic inflammatory disease that can cause scarring and long-term damage to the heart valves, usually end up as RHD.6,7 RHD has also been linked to overcrowding, poor living circumstances, poor sanitation, malnutrition, and insufficient access to healthcare, which highlights its high prevalence in sub-Saharan Africa and other low-income countries.9–11 A global RHD registry known as the REMEDY project found that heart failure is the most common complication of RHD. This is followed by pulmonary hypertension, atrial fibrillation, stroke, and infective endocarditis.12
The disease pattern of RHD is expected to vary in different African countries, because of differences in epidemiology, access to diagnostic methods, disparities in rural versus urban population distribution, and variations in public awareness of RHD. However, there is still limited data on clinical presentation and socio-demographic characteristics of patients with RHD in most sub-Saharan African countries. In view of advancing industrialization and the improving income of many low-resource settings, it is important to determine the current clinical presentation and socio-demographic profile of RHD in different settings in order to inform appropriate healthcare policies, innovative techniques, and interventional options for this critical cardiovascular condition.
A recent study of cardiovascular disease patterns encountered in an outpatient cardiac clinic in Ghana suggested that RHD could still be a significant cause of cardiovascular disease in this country.13 Consequently, the goal of this study was to determine the clinical and socio-demographic profile of RHD patients at the Komfo Anokye Teaching Hospital in Kumasi, Ghana.
Methods and Materials
Between 2016 and 2019, we examined the clinical characteristics of patients with RHD who were enrolled in the adult outpatient cardiac clinic of the Komfo Anokye Teaching Hospital, a tertiary care center in Kumasi, Ghana. The clinic has service twice a week and accepts referrals for patients with cardiovascular diseases aged 13 years and above from most of Ghana’s 16 administrative areas, serving a population of about 15 million people. The information was gathered by studying the patient’s medical records. The study gathered information on 140 patients with a definitive diagnosis of RHD, ranging in age from 15 to 65 years old. Patient’s socio-demographics, echocardiography reports, clinical presentation, current diagnoses, and medications were among the information gathered.
Information obtained included patients’ medical histories and physical examinations, 12-lead electrocardiogram findings, and echocardiography findings. At the time of admission to our centre, RHD was diagnosed based on a previous history of acute rheumatic fever with valve disease and/or the World Heart Federation criteria for echocardiographic diagnosis of RHD.
Echocardiography Reports
According to the standard format developed by the American Society of Echocardiography14 and the 2014 AHA/ACC guidelines for the care of patients with valvular heart disease (VHD),15 we retrieved the transthoracic echocardiography data for all research participants. The 2012 World Heart Federation (WHF) criteria for echocardiographic diagnosis of conclusive RHD16 were used to diagnose rheumatic VHD. Based on markers specified in the 2008 ACC/AHA Focused Update Practice Guideline Guidelines for the Management of Patients with VHD,17 the severity of valve disease was classified as mild, moderate, or severe.
Ethical Consideration
The study complied with the Declaration of Helsinki. The Committee on Human Research Publication and Ethics of the School of Medicine and Dentistry, Kwame Nkrumah University of Science and Technology, granted ethical permission (CHRPE/56/15). The study participants gave their written informed consent to the review of their medical records. Parents or legal guardians provided informed consent for patients less than 18 years.
Statistical Design and Analysis
The data were captured using EPI-DATA (version 3.1). After that, the data were cleaned and exported to Stata 14 for analysis. The mean (standard deviation) and median (inter-quartile range) of continuous variables were calculated and displayed in the tables. Frequency and percentages were used to analyze categorical data. The chi-square test was used to obtain the test of significance (p-value). Significant was defined as a p-value of less than 0.05.
Results
Socio-Demographic
This study included a total of 140 RHD patients. The patients’ average age (± standard deviation) was 39.7. (± 9.9 years), and the majority of them were females (n=103; 74.57%) (Table 1). A total of 40 (28.5%) of the patients were aged 15 to 24, Nearly half of the patients (n=69; 49.21%) were under the age of 35. The majority of the patients were Christians (82.14%), with 19 (13.57%) Muslims and 0.2% having no religious affiliation. Those aged at least 55 years did not have any tricuspid valve lesions on their records. Again, RHD appeared to be spared in the 35–44 year old group, with a total of 11 cases (7.8%) (Table 1).
 |
Table 1 Socio-Demographic and Baseline Characteristics of the Patients |
Patients’ occupations ranged from white-collar jobs to farming, artisans, trading, and students, among others. Trading (59%) was the most common occupation among the patients, accounting for 42.14%. Twenty-six (18.57%) of the patients were students. There were 25 patients who were unemployed (17.86%). In terms of marital status, the data revealed that 64 (45.71%) of the patients were married, while 42 (30%) were single (Table 1). Tables 1 and 3 show that the majority of the patients in this study lived in rural areas, with 116 (82.86%) living in rural areas and 24 (17.4%) residing in urban areas.
 |
Table 2 Clinical Characteristics and Valvular Heart Lesions Among Patients |
 |
Table 3 Valvular Lesion Distribution by Gender, Age Groups and Residency Among the Patients |
Table 2 shows the clinical presentation of the patients. The most common presenting symptom was palpitation which accounted for 76 (54.29%) of the patients, followed by chest pains (n= 51; 36.43%), dyspnoea (n=35; 25.00%), dizziness (n=34; 24.29%), and easy fatigability (n=11; 7.89%).
From Table 3 and Figure 1, the commonest valvular disorder seen among the patients was mitral regurgitation (n=79; 56.43%), followed by aortic regurgitation (n=44; 30.71%) and tricuspid regurgitation (n=17; 12.14%). Both mitral regurgitation and aortic regurgitation were found in 1 patient out of a total 140 study population (0.17%). Figures 2 and 3 show an image of a participant with mitral valve calcification due to RHD with evidence of mitral regurgitation. The majority of the patients in this study presented with moderate-to-severe valvular disease (n=108; 77.14%). Female patients had a higher percentage of mitral regurgitation and aortic regurgitation compared to males (79.7% vs.20.3%) and (70.5% vs 29.5%) respectively. Aortic regurgitation and mitral regurgitation were seen more in rural dwellers than patients living in urban communities (81.8% vs.18.2%) and (79.7% vs 20.3%) respectively (Table 3).
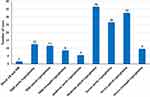 |
Figure 1 Echocardiographic diagnosis of rheumatic heart disease. |
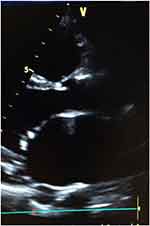 |
Figure 2 Mitral valve leaflets calcifications with sub-valvular thickening and LA volume overload in an RHD patient. |
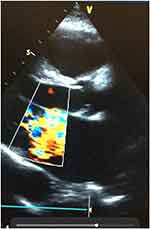 |
Figure 3 Colour Doppler showing mitral regurgitation in the same RHD patient in ventricular systole. |
Discussion
Rheumatic heart disease has mostly disappeared in most industrialized nations, but it remains a significant clinical and public health issue in Ghana and several other developing countries.1,13,18,19 The clinical and socio-demographic profile of patients with RHD attending the adult cardiac clinic of the Komfo Anokye Teaching Hospital in Kumasi, Ghana, is described in this study.
A total of 140 individuals (37 men and 103 women) were investigated retrospectively. Females made up of 73.4% of our RHD patients, with a female to male ratio of 2.8:1. RHD was found to be more prevalent in females than in males in various studies,20 and this was confirmed in this investigation. It is unclear if this phenomenon is due to a genetic predisposition, increased sensitivity, or exposure to group A streptococcus as a result of greater engagement in child rearing, low socio-cultural positions or inequity, and insufficient access to preventive medical treatment for women.18
The average age of the patients in this study was 39.7 ± 9.9 years, with more than half of them falling within the 15–44 year age group. This is in line with the findings from earlier research in sub-Saharan Africa, which found a high prevalence of RHD in young adults.21,22 The majority of the participants in this study were young adults, which supports the theory that rheumatic valvular lesions are more common in young people.23–26 It is worth noting that the disease has the ability to stifle national output because it affects the population’s most active and productive people.
The majority of the patients in this research (79.2%) were Christians. This could be partly due to the fact that the majority of the individuals in the research region are Christians. In terms of occupation, the majority of the study patients (42.1%) were traders, followed by students (18.6%), and 17.9% were jobless. Indeed, the research area is a commercial metropolis with one of Ghana’s largest markets.
The majority of the patients in our study (85.29%) lived in rural areas. As shown in this study, other research work from developing nations have indicated a greater frequency of RHD among rural inhabitants.24,27 Other studies from developing nations have indicated a greater frequency of RHD among rural inhabitants. On the other hand, several investigations have found a higher prevalence of RHD in patients living in metropolitan communities.28 When compared to metropolitan settings, the prevalence of RHD in rural residents may be linked to poor and substandard socioeconomic conditions. Furthermore, due to the larger distance between the nearest healthcare facility and a lack of information about preventive medical measures, rural communities have less access to quality healthcare.
Zhang et al26 and the 75% reported by Ogah et al,27 but significantly higher than the 34.1% recorded by Thakur et al.28 Chest pains were the second most prevalent symptom, affecting 36.4% of the participants in this study. Other investigations by Zhang et al,23 Ogah et al,27 and Thakur et al,28 revealed that 47.7%, 76.4.9%, and 15.9% of patients, respectively, had chest pains. Dyspnoea was found in only 25.0% of patients, compared to 76.4% and 100% in Zhang et al19 and Ogah et al,27 respectively. Other symptoms, such as cough, were observed. Zhang et al19 found much more RHD patients with easy fatigability (89.2%) than the 7.9% seen in this study. The differences in the presenting complaints among patients with RHD seen in various studies may be attributed to differences in sample sizes and the patient populations which were studied.
Mitral regurgitation was the most common valvular lesion among the RHD patients (56.43%), which is consistent with findings from previous studies by Sani et al,18 Okello et al,21 Mirabel et al,29 and Aurakzai et al,30 who all reported mitral regurgitation as the most common valvular lesion in RHD. Other investigations, however, have found that mitral regurgitation with mitral stenosis is the most prevalent valvular presentation in RHD.13,23 Mitral stenosis is the most prevalent valvular lesion in RHD, according to a few studies.31 Surprisingly, there was no evidence of mitral stenosis in this report.
The second most prevalent valvular lesion was aortic regurgitation, which was seen in 30.7% of our RHD patients. Aortic regurgitation prevalence has been shown to range between 3.1% and 47% in other studies.28,32 As indicated in our study, RHD typically affects left-sided valves, with the mitral valve having a stronger affinity and effect. The explanation for the left-sided predominance is unknown, but some studies ascribe it to the left heart’s dominance.33,34
In our study, tricuspid regurgitation was reported in 12.1% of RHD cases, whereas other investigations revealed rates ranging from 5.7% to 48.9%.23,32 Tricuspid regurgitation is more often caused by right ventricular dilatation than by direct rheumatic involvement, and it usually develops after the onset of pulmonary hypertension following left-sided valvular lesions.35
Notably, the majority of the patients in this study (77.14%) had moderate-to-severe valvular disease, which is consistent with earlier study.20 This demonstrates that the majority of the patients had progressive RHD.
This was a hospital-based study, thus it was constrained by the fact that we detailed characteristics of patients we encountered in our healthcare facility, which cannot be adequately represented in the broader population by extrapolation. Despite this flaw, this report includes current, accurate, and dependable data that may be used to characterize the RHD burden in Ghana and other West African sub-region.
In conclusion, this study found that RHD is more common in rural areas, and it affects females more than males; mostly affecting the working-age population (15 to 54 years). It is strongly advised that efficient healthcare systems be built to ensure that patients have access to high-quality care, improved socioeconomic situations, and educational campaigns emphasizing the importance of RHD prevention and early detection. This is especially important in sub-Saharan Africa, where open-heart surgery for patients with severe RHD is not widely available.
Acknowledgment
The authors are grateful to the staff and patients of the cardiac clinic at the Komfo Anokye Teaching Hospital in Kumasi, Ghana, for their assistance. This study would not have been possible without their cooperation.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for profit sectors.
Disclosure
The authors confirm that there are no conflicts of interest in this article’s content.
References
1. Carapetis JR, Steer AC, Mulholland EK, Weber M. The global burden of group A streptococcal diseases. Lancet Infect Dis. 2005;5(11):685–694. doi:10.1016/S1473-3099(05)70267-X
2. Marijon E, Mirabel M, Celermajer DS, Jouven X. Rheumatic heart disease. Lancet. 2012;379:953–964. doi:10.1016/s0140-6736(11)61171-9
3. Eisenberg MJ. Rheumatic heart disease in the developing world: prevalence, prevention, and control. Eur Heart J. 1993;14:122–128. doi:10.1093/eurheartj/14.1.122
4. Haffejee I. Rheumatic fever and rheumatic heart disease: the current status of its immunology, diagnostic criteria, and prophylaxis. QJM Int J Med. 1992;84:641–658. doi:10.1093/oxfordjournals.qjmed.a068703
5. Essop MR, Nkomo VT. Rheumatic and nonrheumatic valvular heart disease. Circulation. 2005;112:3584–3591. doi:10.1161/CIRCULATIONAHA.105.539775
6. World Health Organization. Rheumatic fever and rheumatic heart disease: report of a WHO expert consultation, Geneva, 29 October – 1 November, 2001. World Health Organization; 2004.
7. Myint NPST, Aung NM, Win MS, et al. The clinical characteristics of adults with rheumatic heart disease in Yangon, Myanmar: an observational study. PLoS One. 2018;13:e0192880. doi:10.1371/journal.pone.0192880
8. Watkins DA, Johnson CO, Colquhoun SM, et al. Global, regional, and national burden of rheumatic heart disease, 1990–2015. N Engl J Med. 2017;377:713–722. doi:10.1056/NEJMoa1603693
9. Sharma P, Shakya U, Shrestha KCS. Clinical profile and management in children with rheumatic heart disease in a tertiary cardiac care center of Nepal. Nepal Heart J. 2016;13:33. doi:10.3126/njh.v13i2.15562
10. Jackson SJ, Steer AC, Campbell H. Systematic Review: estimation of global burden of non-suppurative sequelae of upper respiratory tract infection: rheumatic fever and post-streptococcal glomerulonephritis. Trop Med Int Health. 2011;16:2–11. doi:10.1111/j.1365-3156.2010.02670.x
11. Seckeler MD, Hoke TR. The worldwide epidemiology of acute rheumatic fever and rheumatic heart disease. Clin Epidemiol. 2011;3:67–84. doi:10.2147/CLEP.S12977
12. Zühlke L, Engel ME, Karthikeyan G, et al. Characteristics, complications, and gaps in evidence-based interventions in rheumatic heart disease: the Global Rheumatic Heart Disease Registry (the REMEDY study). Eur Heart J. 2015;36:1115–1122a. doi:10.1093/eurheartj/ehu449
13. Owusu IK, Acheamfour-Akowuah E. Pattern of Cardiovascular diseases as seen in an out-patient cardiac clinic in Ghana. World J Cardiovasc Dis. 2018;8:720–726. doi:10.4236/wjcd.2018.81008
14. Gardin JM, Adams DB, Douglas PS, et al. Recommendations for a standardized report for adult transthoracic echocardiography: a report from the American Society of Echocardiography’s nomenclature and standards committee and task force for a standardized echocardiography report. J Am Soc Echocardiogr Off Publ Am Soc Echocardiogr 2002;15:275–290. doi:10.1067/mje.2002.121536.
15. Nishimura RA, Otto CM, Bonow RO, et al. AHA/ACC guideline for the management of patients with valvular heart disease: executive summary: a report of the American College of Cardiology/American Heart Association task force on practice guidelines. Circulation. 2014;2014(129):2440–2492. doi:10.1161/CIR.0000000000000029
16. Reményi B, Wilson N, Steer A, et al. World Heart Federation criteria for echocardiographic diagnosis of rheumatic heart disease--an evidence-based guideline. Nat Rev Cardiol. 2012;9:297–309. doi:10.1038/nrcardio.2012.7
17. Bonow RO, Carabello BA, Chatterjee K, et al. Focused update incorporated into the ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1998 Guidelines for the Management of Patients With Valvular Heart Disease): endorsed by the Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Circulation. 2008;2008(118):e523–661. doi:10.1161/CIRCULATIONAHA.108.190748
18. Sani MU, Karaye KM, Borodo MM. Prevalence and pattern of rheumatic heart disease in the Nigerian Savannah: an echocardiographic study. Cardiovasc J Afr. 2007;18:295–299.
19. Zhang W, Mondo C, Okello E, et al. Presenting features of newly diagnosed rheumatic heart disease patients in Mulago Hospital: a pilot study. Cardiovasc J Afr. 2013;24:28–33. doi:10.5830/CVJA-2012-076
20. Rwebembera J, Manyilirah W, Zhu ZW, et al. Prevalence and characteristics of primary left-sided valve disease in a cohort of 15,000 patients undergoing echocardiography studies in a tertiary hospital in Uganda. BMC Cardiovasc Disord. 2018;18:82. doi:10.1186/s12872-018-0813-5
21. Okello E, Longenecker CT, Beaton A, Kamya MR, Lwabi P. Rheumatic heart disease in Uganda: predictors of morbidity and mortality one year after presentation. BMC Cardiovasc Disord. 2017;17:20. doi:10.1186/s12872-016-0451-8
22. Sliwa K, Carrington M, Mayosi BM, Zigiriadis E, Mvungi R, Stewart S. Incidence and characteristics of newly diagnosed rheumatic heart disease in urban African adults: insights from the heart of Soweto study. Eur Heart J. 2010;31:719–727. doi:10.1093/eurheartj/ehp530
23. Boyarchuk O, Komorovsky R, Kovalchuk T, Olha D. Socio-demographic and medical predictors of rheumatic heart disease in a low-risk population. Pediatr Pol. 2018;93:325–330. doi:10.5114/polp.2018.77998
24. Paar JA, Berrios NM, Rose JD, et al. Prevalence of rheumatic heart disease in children and young adults in Nicaragua. Am J Cardiol. 2010;105:1809–1814. doi:10.1016/j.amjcard.2010.01.364
25. Marijon E, Ou P, Celermajer DS, et al. Prevalence of rheumatic heart disease detected by echocardiographic screening. N Engl J Med. 2007;357:470–476. doi:10.1056/NEJMoa065085
26. Riaz BK, Selim S, MdN K, Chowdhury KN, Chowdhury SH, Rahman M. Risk factors of rheumatic heart disease in Bangladesh: a case-control study. J Health Popul Nutr. 2013;31:70–77.
27. Ogah, O, Adegbite, GD, Udoh, S, Ogbodo, EI . Chronic rheumatic heart disease in Abeokuta, Nigeria: data from the Abeokuta heart disease registry; n.d. Available from: https://www.nigjcardiol.org/article.asp?issn=0189-7969;year=2014;volume=11;issue=2;spage=98;epage=103;aulast=Ogah.
28. Thakur J, Negi P, Ahluwalia S, Vaidya N. Epidemiological survey of rheumatic heart disease among school children in the Shimla Hills of northern India: prevalence and risk factors. J Epidemiol Community Health. 1996;50:62–67. doi:10.1136/jech.50.1.62
29. Mirabel M, Tafflet M, Noël B, et al. Newly diagnosed rheumatic heart disease among indigenous populations in the Pacific. Heart Br Card Soc. 2015;101:1901–1906. doi:10.1136/heartjnl-2015-308237
30. Aurakzai HA, Hameed S, Shahbaz A, et al. Echocardiographic profile of rheumatic heart disease at a tertiary cardiac centre. J Ayub Med Coll Abbottabad. 2009;21:122–126.
31. Faheem M, Hafizullah M, Gul A, Jan HU, Khan MA. Pattern of valvular lesions in rheumatic heart disease. J Postgrad Med Inst. 2007;21:32.
32. Joseph N, Madi D, Kumar GS, Nelliyanil M, Saralaya V, Rai S. Clinical spectrum of rheumatic fever and rheumatic heart disease: a 10 year experience in an urban area of South India. North Am J Med Sci. 2013;5:647–652. doi:10.4103/1947-2714.122307
33. Ravisha MS, Tullu MS, Kamat JR. Rheumatic fever and rheumatic heart disease: clinical profile of 550 cases in India. Arch Med Res. 2003;34:382–387. doi:10.1016/S0188-4409(03)00072-9
34. Jadoon S, Haider N, Hassan M. Retrospective analysis of 54 patients with acute rheumatic fever. Pak Paed J. 2003;27:118–120.
35. WHO Study Group on Rheumatic Fever and Rheumatic Heart Disease (2001): Geneva S, Organization WH. Rheumatic fever and rheumatic heart disease: report of a WHO expert consultation, Geneva, 20 October - 1 November 2001. World Health Organization; 2004.
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
