Back to Journals » Clinical Ophthalmology » Volume 15
Characteristics and Outcomes of a Series of COVID-Associated Mucormycosis Patients in Two Different Settings in Egypt Through the Third Pandemic Wave
Authors Fouad YA , Bakre HM, Nassar MA , Gad MOA, Shaat AAK
Received 18 October 2021
Accepted for publication 14 December 2021
Published 22 December 2021 Volume 2021:15 Pages 4795—4800
DOI https://doi.org/10.2147/OPTH.S344937
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Scott Fraser
Yousef A Fouad,1,2 Hatem M Bakre,3 Mahmoud A Nassar,4 Mohamed Omar A Gad,5 Ashraf Abdelsalam Kandeel Shaat1,2
1Department of Ophthalmology, Ain Shams University Hospitals, Cairo, Egypt; 2Oculoplastic Service, Al Mashreq Eye Center, Cairo, Egypt; 3Faculty of Medicine, Aswan University, Aswan, Egypt; 4Faculty of Medicine, Assuit University, Assuit, Egypt; 5ENT Department, Assuit University Hospitals, Assuit, Egypt
Correspondence: Yousef A Fouad
Ophthalmology Department, Ain Shams University, Ramses Street, Abbassiya, Cairo, 11517, Egypt
Tel +201063781237
Email [email protected]
Purpose: To report on the underlying risk factors, clinical characteristics, presentation, and survival of coronavirus disease 2019 (COVID-19) associated mucormycosis (CAM) cases referred for ophthalmological consultation in two different Egyptian settings during the third viral pandemic wave (April to August of 2021).
Methods: A multicentric, retrospective analysis involving one public and two private hospitals in metropolitan Cairo, and two university hospitals in upper Egypt (Aswan and Assuit). Cases that fulfilled the diagnostic criteria of mucormycosis, with a prior or concurrent COVID-19 infection, were included.
Results: Of the 26 patients included in the final analysis, 15 (57.7%) were from Cairo and 11 (42.3%) were from upper Egypt. Twenty-five patients (96.2%) had poorly controlled diabetes mellitus and 20 (76.9%) had received corticosteroid treatment for COVID-19, of which 30.8% had mild or moderate disease. The median duration from COVID-19 till the onset of CAM was 20.5 days. No visual function could be detected in the affected eye of 16 patients (61.6%) on presentation. The mortality rate was 46.2% despite surgical debridement in half of the cases that died.
Conclusion: CAM continues to be a concrete threat throughout Egypt, especially in patients with poorly controlled diabetes mellitus and those who inadvertently receive steroid therapy. It remains to be seen if increased vaccination coverage and early detection due to increased awareness would result in declining rates and adverse sequelae of CAM. A national registry would allow for better monitoring of the disease trends.
Keywords: coronavirus disease 2019, COVID-19, mucormycosis, COVID-associated mucormycosis, invasive fungal sinusitis
Introduction
The coronavirus disease 2019 (COVID-19) pandemic has brought about a multitude of complications that occur during the acute or post-acute stages of the viral infection, some of which are yet to be fully understood.1,2 Reported ocular complications of the viral infection have varied from common but simple ones as ocular surface inflammation,3 to less common but serious ones as retinal vascular occlusion,4 optic neuritis, and ocular cranial nerve palsies.5 Adverse ocular sequelae of the pandemic have also been linked to treatment protocols,6 positioning during intensive care unit stay,7 lockdown restrictions,8 and immunization.9
Mucormycosis is a life-threatening, opportunistic, angioinvasive fungal infection that occurs in immunocompromised individuals, especially those with uncontrolled diabetes mellitus (DM).10 The disease often starts in the paranasal sinuses with spread to the surrounding tissues, including the orbit.11 A resurgence of the fungal infection has been reported during the pandemic, either coexisting with or following recovery from the viral infection, justifying a separate entity: COVID-associated mucormycosis (CAM).12 The exact link between the viral and fungal infection remains unclear, but the most widely spread hypothesis entails the inadvertent use of immunosuppressive agents, particularly corticosteroids, to treat the viral infection in an already immune-compromised individual, particularly those with poorly controlled DM.13 Another possible hypothesis is that the combination of biochemical alterations caused by the viral infection, including raised ferritin levels, acidosis, and acute cortisol stress response with elevated serum glucose levels, create the perfect environment for the growth and propagation of the fungal species.14,15 Although most of CAM cases have been reported from India,16,17 other countries have reported spikes in the fungal infection rates during the viral pandemic, including Egypt,18 Chile,19 and Iran.20
In this report, we describe a series of CAM cases presenting to different settings within different geographical locations across Egypt during the third wave of the viral pandemic.
Methods
Study Setting
This multicentric, retrospective, clinical-record analysis included patients diagnosed with CAM that were referred for ophthalmological consultation at 5 different centers: 3 in Metropolitan Cairo (1 public and 2 private hospitals) that mainly receive patients from an urban setting, and 2 university hospitals in upper Egypt (Aswan and Assuit University hospitals) that receive patients from urban, semirural, and rural settings. The analyzed period spanned the duration of the third pandemic wave, in the four-month interval between April 2021 and August 2021. The period coincided with the world-wide spread of the delta variant of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which processed a high transmission rate.21,22
Ethical Approval
The study adhered to the tenets of the Declaration of Helsinki and ethical approval from Aswan faculty of medicine ethical committee (ASWU/552/7/21) was obtained prior to study initiation. Data deidentification was ensured prior to retrospective analysis and, consequently, informed consent was waived.
Study Population and Data Collection
Patients were included if they had 1) prior proof of COVID-19 infection, and 2) a confirmed mucormycosis diagnosis. Validation of prior COVID-19 infection in the patients’ records was by evidence of a positive reverse transcription polymerase chain reaction (RT-PCR) test for SARS-CoV-2. The patients’ clinical, laboratory, imaging, and surgical records were also reviewed for a confirmed diagnosis of CAM, which was based on the global guideline for the diagnosis and management of mucormycosis published by the European Confederation of Medical Mycology in cooperation with the Mycoses Study Group Education and Research Consortium.23 Patients were excluded if there was no prior documentation of COVID-19 infection or if the diagnosis of the fungal infection was only suspected.
Record analysis included demographic variables (age and sex), medical comorbidities, diabetic status and control (glycated hemoglobin levels, HbA1c), COVID-19 severity (based on the world health organization’s clinical progression scale24), treatment details and setting, and temporal relation between the viral and fungal infection. Collected CAM data included presentation, laterality, corrected distance visual acuity (CDVA) on presentation, medical and surgical treatment details, and final clinical outcome.
Data Analysis
Data were recorded and coded in a spreadsheet. Descriptive statistics were conducted using the statistical package for social sciences (SPSS) version 25. For normally distributed data, central tendency and dispersion were measured and expressed in terms of mean ± standard deviation (SD), while for non-normally distributed data, they were expressed as median (interquartile range, IQR).
Results
Sample Description
The final analysis included 26 patients with CAM, of which 15 (57.7%) were from metropolitan Cairo, and 11 (42.3%) were from upper Egypt (Aswan and Assuit). The demographic characteristics of the patients are depicted in Table 1. Although the median age of the whole series was 62.5 years, the ages of the cases widely ranged from 21 to 77 years across the whole analyzed group, with a slight male predominance (1.17:1).
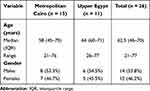 |
Table 1 Demographic Data of the Analyzed CAM Cases Grouped by Setting |
Twenty-five of the 26 patients (96.2%) had poorly controlled DM; records of HbA1c level were available in 14 of them (56%) and it had a median (IQR) value of 9.3% (8–10.7%). Seven (26.9%) of the cases had hypertension, 3 (11.5%) had chronic kidney disease, 2 (7.7%) had ischemic heart disease, 2 (7.7%) had bronchial asthma, and 1 (3.8%) had decompensated liver disease.
COVID-19 Variables
Table 2 demonstrates the analysis of COVID-19-related variables in the studied series. Fourteen of the 15 cases (93.3%) that developed CAM in metropolitan Cairo had moderate or severe COVID-19, and consequently, a total of 13 patients (86.7%) from that group were treated in a hospital setting. Conversely, 5 of the 11 cases (45.5%) that developed CAM in upper Egypt had mild disease and most cases (10 out of 11, 90.9%) were treated in a home setting. Most of the patients (20, 76.9%) received corticosteroid treatment for COVID-19, of which 4 (20%) received oral corticosteroids, and 16 (80%) received intravenous corticosteroids, while 5 patients (19.2%) received tocilizumab. Since the Egyptian protocol for management of COVID-19 recommends limiting corticosteroid therapy to severe cases, 8 cases (30.8%) were considered to have received inadvertent corticosteroid therapy.
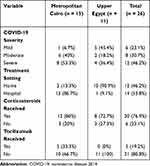 |
Table 2 Distribution of COVID-19-Related Variables Across the Studied Series (n = 26), Grouped by Setting |
CAM Presentation and Outcomes
The details of mucormycosis presentation and management are summarized in Table 3. Fifteen patients (57.7%) developed mucormycosis in association with the active viral infection, while 11 (42.3%) presented following recovery and negative SARS-CoV-2 RT-PCR. The median duration from the onset of COVID-19 symptoms till the onset of CAM symptoms was 20.5 days. On presentation, the right eye was involved in 15 patients (57.7%), the left eye was involved in 10 patients (38.5%), and bilateral eye involvement was seen in 1 patient (3.8%). The most common initial symptom was facial swelling (13 cases, 50%). On ophthalmological evaluation, no visual function could be detected in the affected eye of 16 (61.6%) of the referred cases.
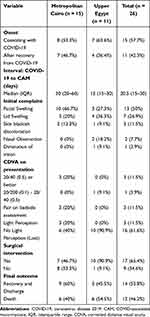 |
Table 3 CAM Presentation, Management, and Outcomes, Grouped by Setting |
All patients received intravenous antifungal treatment (Amphotericin B). Seventeen patients (65.4%) were deemed fit for general anesthesia and underwent surgical sinus debridement that was associated with orbital exenteration in 5 of them (29.4%). The overall survival rate was 53.8% (n = 14), of which half of them (50%) had underwent surgical debridement.
Discussion
CAM continues to pose a threat for immunocompromised individuals during the latest viral pandemic waves. We report on the experience of two settings from Egypt, separated geographically and differing in the background of the presenting patients, during the third pandemic wave in the country that spanned the Summer of 2021. The results of our analysis show that, with minor exceptions, the clinical characteristics and outcomes of CAM across both patient groups were similar to each other and to globally reported ones.
Most of the CAM cases globally reported so far are from the Indian population.16 The burden of mucormycosis in the Indian population has, however, been high prior to the beginning of the COVID-19 pandemic.25 The country has further experienced a surge in COVID-19 cases during the second wave which probably relates to the prevalent strain variants.26 The largest Indian nation-wide survey of CAM to date is the Collaborative OPAI-IJO study on mucormycosis in COVID-19 (COSMIC),27 which analyzed the data of 2826 patients presenting from the start of the pandemic (January 2020) till May 2021. DM was present in 78% of the patients and 87% had received corticosteroids for COVID-19, suggesting that both factors are the most important predisposing ones for CAM. This is in line with the findings of our analysis in which 96.2% of the patients had DM and 76.9% had prior history of corticosteroid treatment. It is worth mentioning that our current understanding of the link between COVID-19 and mucormycosis is incomplete, based on observational analyses and unvalidated by basic experimentation. A link between both infections might be through the biochemical alterations caused by the viral infection, with DM and corticosteroid therapy merely contributing to the such alterations.14,15 The overall mortality in the COSMIC report was 14%,27 which is markedly lower than that in our patient group (46.2%). This could probably be attributed to the delay in presentation which is reflected by the median onset of CAM following COVID-19 symptoms (13 days in the COSMIC report versus 20.5 days in our group); in fact, patients with advanced stages of the disease in the COSMIC analysis had a mortality rate of 39%.27
Reports of CAM outside the Indian population are scarce,25 most of which are case reports.28 In Iran, two cross-sectional studies from Tehran20 and west Iran29 have reported on 15 and 12 CAM cases, of which 86% and 87.3% had DM, with a mortality rate of 47% and 66.6%, respectively. A series of 10 cases with CAM were also reported from Pakistan,30 of which 70% were diabetic and 80% had received corticosteroids. Sixteen cases were also reported from Chile,19 15 of which had received corticosteroids, but only 4 (25%) were diabetic and none were immunocompromised, suggesting different predisposing factors within different settings.
In Egypt, a report of 12 cases with rhino-orbital-cerebral mucormycosis presenting to a university hospital in Cairo during the first viral pandemic wave included 6 patients with prior or concurrent COVID-19.18 Ashour et al31 also retrospectively analyzed the imaging spectrum of 8 cases with invasive rhino-orbital fungal disease during the first and second pandemic waves in Cairo, Egypt, of which 6 were diagnosed as mucormycosis. El-Kholy et al32 conducted a prospective longitudinal study on invasive fungal sinusitis that spanned the duration of the second Egyptian pandemic wave at a university hospital in another governorate, Mansoura, and detected 28 cases with CAM. Alfishawy et al33 reported on 21 patients with CAM from 11 different hospitals in metropolitan Cairo during the third pandemic wave, of which 19 (90.5%) had DM and all had received corticosteroid treatment.
The previous numbers lead to the conclusion that Egypt comes second to India in the number of published reports of CAM cases. The high prevalence of DM in the country, which ranges in recent cluster analyses in different regions from 16.7%34 to 20.9%,35 could explain the burden of CAM.33 Other factors noticed in our sample that can contribute to the disease load include the delay in presentation (median 20.5 days), inadvertent use of corticosteroids in mild-moderate cases (30.8% of the patients), and self-medication for the viral infection in homes instead of seeking hospital care for moderate-severe cases, especially in Upper Egypt (6.7% in metropolitan Cairo, and 45.4% in Upper Egypt).
Limitations to our study include the relatively small sample size which does not allow for meaningful regression analysis of survival-related factors, in addition to the retrospective analysis of the data. As for any emerging infectious diseases, the establishment of an online national case registry for CAM would certainly be of benefit to monitor the trends of the disease.36 With increasing vaccination coverage worldwide, and with increased awareness among the public and practitioners about CAM,37 it remains to be seen whether CAM incidence and mortality rates would change over the coming months.
Conclusions
CAM continues to be a valid infectious threat in Egypt with the country’s high prevalence of DM and with inadvertent steroid therapy. Although this is the first multicentric study on CAM in Egypt that include patients from different settings, a national online registry is needed to monitor the disease trends during the upcoming pandemic waves. Further studies are needed to clarify the geographic predilection of CAM, demonstrate whether the relation between the viral and fungal infection is causal or an association, and analyze survival-related factors in CAM cases.
Abbreviations
COVID-19, coronavirus disease 2019; CAM, COVID-associated mucormycosis; COSMIC, collaborative OPAI-IJO study on mucormycosis in COVID-19; DM, diabetes mellitus; IQR, interquartile range; RT-PCR, reverse transcription polymerase chain reaction; SARS-CoV-2, Severe Acute Respiratory Syndrome Coronavirus 2; SD, standard deviation.
Funding
There is no funding to report.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Drake TM, Riad AM, Fairfield CJ, et al. Characterisation of in-hospital complications associated with COVID-19 using the ISARIC WHO Clinical Characterisation Protocol UK: a prospective, multicentre cohort study. Lancet. 2021;398(10296):223–237. doi:10.1016/S0140-6736(21)00799-6
2. Nalbandian A, Sehgal K, Gupta A, et al. Post-acute COVID-19 syndrome. Nat Med. 2021;27(4):601–615.
3. Aggarwal K, Agarwal A, Jaiswal N, et al. Ocular surface manifestations of coronavirus disease 2019 (COVID-19): a systematic review and meta-analysis. PLoS One. 2020;15(11):e0241661. doi:10.1371/journal.pone.0241661
4. Sheth JU, Narayanan R, Goyal J, Goyal V. Retinal vein occlusion in COVID-19: a novel entity. Indian J Ophthalmol. 2020;68(10):2291. doi:10.4103/ijo.IJO_2380_20
5. Tisdale AK, Chwalisz B. Neuro-ophthalmic manifestations of coronavirus disease 19. Curr Opin Ophthalmol. 2020;31(6):489–494. doi:10.1097/ICU.0000000000000707
6. Bertoli F, Veritti D, Danese C, et al. Ocular findings in COVID-19 patients: a review of direct manifestations and indirect effects on the eye. J Ophthalmol. 2020;2020:1–9. doi:10.1155/2020/4827304
7. Sanghi P, Malik M, Hossain IT, Manzouri B. Ocular complications in the prone position in the critical care setting: the COVID-19 pandemic. J Intensive Care Med. 2020;36(3):361–372. doi:10.1177/0885066620959031
8. Alqudah AA, Al Dwairi RA, Alqudah NM, Abumurad SK. COVID-19 lockdown and eye injury: a case series from Jordan. Int Med Case Rep J. 2020;13:493–501. doi:10.2147/IMCRJ.S274284
9. Pichi F, Aljneibi S, Neri P, Hay S, Dackiw C, Ghazi NG. Association of ocular adverse events with inactivated COVID-19 vaccination in patients in Abu Dhabi. JAMA Ophthalmol. 2021;139(10):1131–1135. doi:10.1001/jamaophthalmol.2021.3477
10. Skiada A, Pavleas I, Drogari-Apiranthitou M. Epidemiology and diagnosis of mucormycosis: an update. J Fungi. 2020;6(4):265. doi:10.3390/jof6040265
11. Werthman-Ehrenreich A. Mucormycosis with orbital compartment syndrome in a patient with COVID-19. Am J Emerg Med. 2021;42:
12. Garg D, Muthu V, Sehgal IS, et al. Coronavirus disease (Covid-19) associated mucormycosis (CAM): case report and systematic review of literature. Mycopathologia. 2021;186(2):289–298. doi:10.1007/s11046-021-00528-2
13. Narayanan S, Chua JV, Baddley JW. COVID-19 associated mucormycosis (CAM): risk factors and mechanisms of disease. Clin Infect Dis. 2021. doi:10.1093/cid/ciab726
14. Pandiar D, Kumar NS, Anand R, Kamboj M, Narwal A, Shameena PM. Does COVID 19 generate a milieu for propagation of mucormycosis? Med Hypotheses. 2021;152:110613. doi:10.1016/j.mehy.2021.110613
15. Pasternak M, Olszanecki R. Mucormycosis in head and neck area - the emerging health problem in COVID-19 pandemic. The perspective of a dental practitioner. Folia Med Cracov. 2021;61(2):117–127. doi:10.24425/fmc.2021.137228
16. Singh AK, Singh R, Joshi SR, Misra A. Mucormycosis in COVID-19: a systematic review of cases reported worldwide and in India. Diabetes Metab Syndr Clin Res Rev. 2021;15(4):102146. doi:10.1016/j.dsx.2021.05.019
17. Selarka L, Sharma S, Saini D, et al. Mucormycosis and COVID‐19: an epidemic within a pandemic in India. Mycoses. 2021;64(10):1253–1260. doi:10.1111/myc.13353
18. Fouad YA, Abdelaziz TT, Askoura A, et al. Spike in rhino-orbital-cerebral mucormycosis cases presenting to a tertiary care center during the COVID-19 pandemic. Front Med. 2021;8:716. doi:10.3389/fmed.2021.645270
19. Rabagliati R, Rodríguez N, Núñez C, Huete A, Bravo S, Garcia P. COVID-19-associated mold infection in critically ill patients, Chile. Emerg Infect Dis. 2021;27(5):1454–1456. doi:10.3201/eid2705.204412
20. Pakdel F, Ahmadikia K, Salehi M, et al. Mucormycosis in patients with COVID‐19: a cross‐sectional descriptive multicentre study from Iran. Mycoses. 2021;64(10):1238–1252. doi:10.1111/myc.13334
21. Kupferschmidt K, Wadman M. Delta variant triggers new phase in the pandemic. Science. 2021;372(6549):1375. doi:10.1126/science.372.6549.1375
22. Del Rio C, Malani PN, Omer SB. Confronting the delta variant of SARS-CoV-2, Summer 2021. JAMA. 2021;326(11):1001. doi:10.1001/jama.2021.14811
23. Cornely OA, Alastruey-Izquierdo A, Arenz D, et al. Global guideline for the diagnosis and management of mucormycosis: an initiative of the European Confederation of Medical Mycology in cooperation with the Mycoses Study Group Education and Research Consortium. Lancet Infect Dis. 2019;19(12):e405–e421. doi:10.1016/S1473-3099(19)30312-3
24. Marshall JC, Murthy S, Diaz J, et al. A minimal common outcome measure set for COVID-19 clinical research. Lancet Infect Dis. 2020;20(8):e192–e197. doi:10.1016/S1473-3099(20)30483-7
25. Muthu V, Rudramurthy SM, Chakrabarti A, Agarwal R. Epidemiology and pathophysiology of COVID-19-associated mucormycosis: India versus the rest of the world. Mycopathologia. 2021;186(6):739–754. doi:10.1007/s11046-021-00584-8
26. Thiagarajan K. Why is India having a covid-19 surge? BMJ. 2021;373:n1124. doi:10.1136/bmj.n1124
27. Sen M, Honavar SG, Bansal R, et al. Epidemiology, clinical profile, management, and outcome of COVID-19-associated rhino-orbital-cerebral mucormycosis in 2826 patients in India – collaborative OPAI-IJO study on mucormycosis in COVID-19 (COSMIC), report 1. Indian J Ophthalmol. 2021;69(7):1670–1692.
28. Hussain S, Baxi H, Riad A, et al. COVID-19-associated mucormycosis (CAM): an updated evidence mapping. Int J Environ Res Public Health. 2021;18(19):10340. doi:10.3390/ijerph181910340
29. Avatef Fazeli M, Rezaei L, Javadirad E, et al. Increased incidence of rhino-orbital mucormycosis in an educational therapeutic hospital during the COVID-19 pandemic in western Iran: an observational study. Mycoses. 2021;64(11):1366–1377. doi:10.1111/myc.13351
30. Nasir N, Farooqi J, Mahmood SF, Jabeen K. COVID-19 associated mucormycosis: a life-threatening complication in patients admitted with severe to critical COVID-19 from Pakistan. Clin Microbiol Infect. 2021;27(11):1704–1707. doi:10.1016/j.cmi.2021.07.038
31. Ashour MM, Abdelaziz TT, Ashour DM, Askoura A, Saleh MI, Mahmoud MS. Imaging spectrum of acute invasive fungal rhino-orbital-cerebral sinusitis in COVID-19 patients: a case series and a review of literature. J Neuroradiol. 2021;48(5):319–324. doi:10.1016/j.neurad.2021.05.007
32. El-Kholy NA, El-Fattah AMA, Khafagy YW. Invasive fungal Sinusitis in post COVID-19 patients: a new clinical entity. Laryngoscope. 2021;131(12):2652–2658. doi:10.1002/lary.29632
33. Alfishawy M, Elbendary A, Younes A, et al. Diabetes mellitus and Coronavirus disease (Covid-19) associated mucormycosis (CAM): a wake-up call from Egypt. Diabetes Metab Syndr. 2021;15(5):102195. doi:10.1016/j.dsx.2021.102195
34. Assaad Khalil SH, Megallaa MH, Rohoma KH, et al. Prevalence of type 2 diabetes mellitus in a sample of the adult population of Alexandria, Egypt. Diabetes Res Clin Pract. 2018;144:63–73. doi:10.1016/j.diabres.2018.07.025
35. AlSawahli H, Mpyet CD, Ezzelarab G, et al. Population-based cross-sectional prevalence survey of diabetes and diabetic retinopathy in Sohag—Egypt, 2019. BMJ Open. 2021;11(6):e047757. doi:10.1136/bmjopen-2020-047757
36. Arora S, Hemmige VS, Mandke C, et al. Online Registry of COVID-19–associated mucormycosis cases, India, 2021. Emerg Infect Dis. 2021;27(11):2963–2965. doi:10.3201/eid2711.211322
37. Farias LABG, Damasceno LS, Bandeira SP, Barreto FKDA, Leitão TDMJS, Cavalcanti LPDG. COVID-19 associated mucormycosis (CAM): should Brazil be on alert? Rev Soc Bras Med Trop. 2021;54:e04102021. doi:10.1590/0037-8682-0410-2021
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
