Back to Journals » International Journal of General Medicine » Volume 15
Characteristics and Follow-Up of Organizing Pneumonia Associated with Haematological Malignancies
Authors Zeng H, Ma Y, He X, Cai S, Chen P , Chen Y , Luo H
Received 2 September 2021
Accepted for publication 23 December 2021
Published 7 January 2022 Volume 2022:15 Pages 301—310
DOI https://doi.org/10.2147/IJGM.S337321
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Huihui Zeng,1– 3 Yiming Ma,1– 3 Xue He,1– 3 Shan Cai,1– 3 Ping Chen,1– 3 Yan Chen,1– 3 Hong Luo1– 3
1Department of Pulmonary and Critical Care Medicine, The Second Xiangya Hospital of Central South University, Changsha, Hunan, People’s Republic of China; 2Research Unit of Respiratory Diseases, Central South University, Changsha, Hunan, People’s Republic of China; 3Hunan Centre for Evidence-Based Medicine Changsha, Hunan, People’s Republic of China
Correspondence: Hong Luo
Department of Pulmonary and Critical Care Medicine, The Second Xiangya Hospital of Central South University, No. 139 Renmin Road, Changsha, Hunan, 410011, People’s Republic of China
Email [email protected]
Background: Organizing pneumonia (OP) is a secondary process in many diseases. Due to its low incidence and indistinct symptoms, there is limited information on OP associated with haematological malignancies. Therefore, the aim of this study was to discuss the characteristics and prognosis of OP associated with haematological malignancies.
Methods: We observed and analysed pathologically confirmed OP cases associated with haematological malignancies in a hospital record database and excluded cases of OP with known causes, including chemotherapy, radiotherapy, targeted therapy, transplantation and infection.
Results: There were five patients with OP underlying only haematological malignancies, including one case each of the following: myelodysplastic syndrome, acute myelogenous leukaemia, multiple myeloma, aplastic anaemia, and T cell lymphoma. Radiological findings did not show a distinct pattern, and two cases mimicked pulmonary aspergillosis with ground-glass opacity (GGO). The diagnosis of OP was confirmed by minimal invasive biopsy. Although all patients developed severe cases, steroids yielded favourable outcomes.
Conclusion: This study demonstrates that haematological malignancies may be a cause of OP and that minimal invasive biopsy may be an effective and safe method to confirm the diagnosis. Although OP associated with haematological malignancies may more frequently develop into severe cases, the OP lesions were steroid-responsive during follow-up.
Keywords: organizing pneumonia, haematological malignancies, characteristics, follow-up
Introduction
Organizing pneumonia (OP) is defined as filling of the distal air spaces with buds of granulation tissue progressing from fibrin exudates to lose collagen.1 This pathological pattern may be encountered in different disease processes, such as infection, infarction, certain treatments (eg, chemotherapy) and immune disorders (eg, connective tissue diseases and graft-versus-host disease).2,3 According to its causes or associated diseases, OP can be categorized into OP with a determined cause, OP with undetermined causes but underlying specific or relevant conditions and cryptogenic OP (COP). Unfortunately, there is a lack of distinct features among these three groups.2 The clinical characteristics of OP are similar to those of respiratory infection (with recurrent or persistent fever, dyspnoea, cough, and weakness as the dominant symptoms) and are difficult to differentiate from other pathological processes. The radiological manifestations are also non-specific, exhibiting patchy consolidation and “ground-glass” opacities. The potentially significant features may be migrating pulmonary infiltrates and less pulmonary fibrosis during follow-up.2
The common pulmonary complications of haematological malignancies are infectious diseases and may be frequently attributed to immune deficiency.4,5 However, non-infectious causes, such as pulmonary oedema, haemorrhage, autoimmune manifestations and pulmonary alveolar proteinosis, could be potential aetiologies for pulmonary infiltrates in patients with haematological malignancies.3–5 In the last decade, OP has been reported in haematological disorder patients.3,6–9 Other related factors, including chemotherapy, stem cell transplantation (SCT) and infection, have been previously described with respect to OP in cases of haematological malignancies.3,7,9 Therefore, there is limited information on OP underlying only haematological malignancies or caused by haematological malignancies. Moreover, due to the lack of specific features, it is difficult to distinguish OP from other pulmonary complications associated with haematological malignancies.
To discuss OP associated with haematological malignancies and exclude other related reasons, we conducted this observational study. Some traditional methods, such as antigen and antibody detection, Gram staining, culture and so on. New and promising NGS was also employed to detect pathogens.
Materials and Methods
Study Design
This observational study was performed at a 3500-bed tertiary teaching hospital, the Second Xiangya Hospital of Central South University, Changsha, in mid-southern China. This study was approved and supervised by the Medical Research Ethics Committee of the Second Xiangya Hospital, Central South University. No interventions were conducted in this study. On the other hand, clear privacy and identity information of patients, such as name, address, telephone number and ID number (all data), were kept confidential and not described in this study. The study was ranked as “not more than the minimum risk” and “the rights and interests of subject will not be invaded” by the Medical Research Ethics Committee of the Second Xiangya Hospital. Therefore, written informed consent was waived. The hospital records’ database was searched for all cases of OP with haematological malignancies from 1 January 2000 to 1 July 2020. The diagnosis of OP was confirmed by two independent pathologists following the American Thoracic Society/European Respiratory Society statement (2013 update).10 Patients were excluded if they had other related conditions, including chemotherapy, radiotherapy, targeted therapy, SCT, bone marrow transplantation (BMT), cord blood transplantation (CBT) and infection.
To exclude infection, sputum, bronchoalveolar lavage fluid (BALF), blood and bone marrow cultures were conducted in these patients, none of which yielded positive results. 1,3-β-
The following information was examined: demographics, underlying cancer diagnoses, physical examination and laboratory findings, pulmonary function before OP diagnosis, findings on chest radiography and computed tomography (CT), steroid treatment, use of concurrent treatments, clinical outcomes and follow-up.
Definitions
The duration of OP diagnosis was defined as the time from symptom onset to the pathological diagnosis of OP. Clinical outcomes during hospitalization were confirmed by two independent pulmonologists, and a favourable clinical response was defined as a complete or partial alleviation of symptoms and signs after 1 week of steroid treatment.
Results
Patients
There were 12 patients with a histopathological diagnosis of OP and concomitant diagnosis of a haematological malignancy in our database from 1 January 2000 to 1 July 2020. Before OP onset, seven patients accepted chemotherapy, including one patient who accepted SCT, one who received targeted therapy (eg, rituximab) and three with ongoing infection. The other five patients had OP associated with haematological malignancies, which were the primary pathological processes without any other related reasons. None of these five patients accepted chemotherapy, targeted therapy or SCT. There were three female patients, including one with myelodysplastic syndrome (MDS), one with acute myelogenous leukaemia (AML, M5), and one with multiple myeloma (MM). One of the two male patients had aplastic anaemia (AA), and the other had T cell lymphoma (TCL). The patients’ ages ranged from 43 to 67 years, and three of them had a smoking history (range from 30 to 80 pack-years).
Clinical Characteristics
The five cases of OP associated with haematological malignancies presented symptoms similar to those of pulmonary infection, such as fever, cough and dyspnoea (Table 1). One patient had a history of haemoptysis, which may have been caused by MDS-induced thrombocytopenia. Two of the patients had a history of productive cough with sputum, but all five patients were observed to have moist inspiratory rales. Consistent with the non-fibrosis on CT scan, none of the patients exhibited Velcro rales.
 |
Table 1 Demographic and Clinical Characteristics of OP Associated with Haematological Malignancies |
All five OP patients were hospitalized for diagnosis, and all were primarily diagnosed with pulmonary infection. Due to unfavourable responses to antibiotics and immune deficiency, pulmonary aspergillosis was the most frequently considered diagnosis before the diagnosis of OP. All patients were found to have anaemia, and four of them (excluding the patient with TCL) showed hypolymphocytosis in routine blood tests. Interestingly, after the use of steroids, all patients showed improvement of anaemia without treatment for haematological malignancies. Before the diagnosis of OP, all patients accepted anti-infection treatments for more than 4 weeks, including piperacillin-tazobactam, cefoperazone-sulbactam, meropenem, and voriconazole. However, none of them received antiviral treatment, indicating that bacterial and fungal infections were the most frequently considered differential diagnoses in OP associated with haematological malignancies. Furthermore, all five OP patients were admitted to the intensive care unit (ICU) for respiratory failure (Table 1).
Radiological Findings
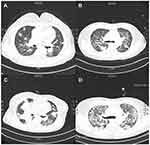
All patients underwent high-resolution computed tomography (HRCT) before OP diagnosis, and one of them even underwent positron emission tomography-computed tomography (PET-CT). As in previous studies,8,9,11 there were patchy consolidative opacities and airspace consolidation along the bronchovascular bundle or in the subpleural area on HRCT (Figure 1). However, some uncommonly described findings on HRCT were observed in these cases. One MDS case and one AML case exhibited multiple nodules along the bronchovascular bundle with ground-glass opacity (GGO). The HRCT scan of the MDS case even showed a halo of GGO, which is a typical early sign of pulmonary fungal infection (Figure 2A). Additionally, the AML patient exhibited interlobular septal thickening and a reticulonodular pattern (Figure 2B). Interestingly, both cases progressed rapidly and developed extensive bilateral opacity on HRCT in less than 2 weeks (Figure 2C and D).
PET-CT was conducted in TCL patients and showed a high standardized uptake value (SUV) max of the lung and pleura (range from 2.5 to 3.5). However, PET-CT showed a higher SUV max of the retroperitoneal and intraperitoneal lymph nodes (>7.5).
Diagnosis
Since OP is a rare disease without distinct symptoms and is defined by pathological pattern, it takes a long time to diagnose this condition.2 The duration from onset to OP diagnosis was extensive, ranging from 1 to 6 months in the 5 OP cases. OP was frequently diagnosed by lung biopsy, including bronchoscopic and CT-guided percutaneous biopsy. All five patients underwent bronchoscopic biopsy, and the results were confirmed by two independent pathologists (Figure 3). Due to the subpleural lesion and limitation of the biopsy sample, one TCL patient failed to obtain a proper diagnosis for the lung lesions even after 2 times of bronchoscopic biopsies. Finally, the OP diagnosis was confirmed via CT-guided percutaneous biopsy.
Cytological detection was conducted in BALF from the five patients, and no malignant cells were found. Four of them had differential cell counts and T cell subsets in BALF, except for the MM case. The ratios of lymphocytes ranged from 15% to 30%, and the ratios of CD4+/CD8+ T cells ranged from 1.5 to 3.0 (Table 1). However, the ratios of neutrophil were under 15% in all 4 cases. In addition, no malignant cells were observed in the alveolar space or wall from the biopsy sample.
Although bronchoscopy revealed purulent sputum in the TCL patient with OP, the pathogen-detection results were negative in sputum and BALF. Bronchoscopy of the other four patients revealed only a few secretions without oedema and congestive mucosa in the airways. To detect possible pathogens, next-generation sequencing (NGS) technology was conducted in blood and BALF from the MDS and AML cases. The sequence analysis did not identify causative pathogens for the two cases.
Before steroid treatment, all AA and MDS patients suffered severe anaemia, and the AML case suffered mild neutropenia. Because all of five patients were severe cases and admitted into ICU, they could not finish the lung function test. All of them lack of the lung function data before OP diagnosis.
Treatment and Outcome
Once the five patients were diagnosed with OP, all were treated with intravenous steroids (methylprednisolone, 40 mg/day) for more than 1 week. Fever and cough were the earliest and most frequently improved symptoms after steroid treatment. The five patients underwent steroid (intravenous and oral) treatment for 3–6 months. During follow-up, we found that the respiratory symptoms and radiological abnormalities were completely reversed after 1–3 months of steroid use (Table 1). One patient died due to AA at the 13th month of follow-up, and the other four patients survived during follow-up. Interestingly, none of the OP case series relapsed.
Due to agranulocytosis, the AML patient was treated with voriconazole and cefoperazone-sulbactam after OP diagnosis. The other four OP patients discontinued antifungal treatment after no evidence of fungal infection was found. Because of mild neutropenia, AA and TCL patients received piperacillin-tazobactam treatment after their OP diagnosis.
After recovery of OP, two of them suffered severe anaemia, one suffered thrombocytopenia, and one was dead. And MDS patient was observed with an improved of anaemia after OP treatment (haemoglobin 57–93 g/L). Only the MDS patients finished the lung function test after OP treatment. The results showed forced expiratory volume in first second/forced vital capacity (FEV1/FVC) 76.0% and FEV1/prediction 90.6% after bronchodilation use. The pulmonary diffusion function of the MDS was valued with diffusion capacity for carbon monoxide of lung single-breath method (DLCO2-sb), and the results of DLCO2 and DLCO2/ VA (alveolar volume) were normal (7.98 and 1.38 mmol/min/kPa/L).
Discussion
Since the aetiology of OP is multifactorial, OP with haematological malignancies could be caused by anti-cancer drugs,12,13 transplantation (SCT, BMT and CBT),11,14 immunodeficiency-induced infection and secondary CTD.2,8,9 The authors summarized the cases of five OP patients with only haematological malignancies after excluding other risk factors, implying that haematological malignancies may be a causative factor for OP.
The patient’s ages in our study ranged from 43 to 68 years, and the average age in OP with haematological malignancies was 52.3 years,8 indicating that younger patients with haematological malignancies may be less likely to develop OP.11 In previous studies, only 55 OP patients were provided rough haematological diagnosis and divided into 3 groups of haematological malignancies (Table 2), 20 cases of lymphoma (36.4%), 22 cases of leukaemia (40%) and 13 cases of MDS (23.6%). Our study demonstrated that AA and MM were two new underlying haematological diseases. Our results also showed an improvement in anaemia following reversal of OP, indicating that OP may aggravate anaemia in haematological malignancies. Unlike previous study of OP with haematological malignancies,9 all patients in our study were admitted to the ICU, and three of them underwent mechanical ventilation. A previous study reported that OP cases with multiple possible causes in other malignancies were also observed with a high ratio of severe cases.8 However, OP with other benign causes was described with a favourable prognosis.2 It is possible that OP associated with haematological malignancies is more likely to develop into a severe case than OP with benign underlying diseases, suggesting that physicians should exercise caution in treating OP patients with haematological malignancies.
 |
Table 2 Summary of Previous Reports on Organizing Pneumonia Associated with Haematological Malignancy |
Previous studies8,9,11 demonstrated that radiological findings were not distinctive, and the authors also found patchy consolidative opacities and airspace consolidation along the bronchovascular bundle or in the subpleural region on HRCT. However, two patients in our case series and 7 OP patients in another study15 were observed to exhibit multiple nodules along the bronchovascular bundle with GGO on HRCT, which were described as highly indicative of invasive pulmonary aspergillosis (IPA). As the disease progressed, the two patients in our study developed extensive consolidation in 1 week. The above findings confirmed that the radiological findings of OP associated with haematological malignancies lacked a unique pattern and could mimic some other diseases, such as IPA. Therefore, physicians cannot exclude OP only by radiological findings in haematological malignancy patients, especially in patients with a poor response to anti-infection treatment. Additionally, there is limited information on PET-CT findings in OP. The SUVmax of the OP lesion in TCL patients was increased but was still lower than that of the malignant lesion, indicating that pulmonary lesions may be different from lymphadenopathy. To discuss whether OP and malignant or metastatic lesions could be distinguished through PET-CT, there is a need of further study with larger sample size.
This study revealed that OP patients with haematological malignancies had similar symptoms to those with pulmonary infection (Table 2), with a long duration from the onset to the diagnosis of OP. Due to the lack of distinctive symptoms and examinations, it is difficult to distinguish OP from infection. Therefore, bronchoscopic, CT-guided percutaneous and surgical lung biopsies were employed in the diagnosis of OP with haematological malignancies (Table 2). At the beginning of the twenty-first century, surgical lung biopsy was more frequently used15–17 (Table 2). However, in the last ten years, bronchoscopic and CT-guided percutaneous biopsies have been more frequently used.12,18,19 Haematological malignancies may lead to contraindications for surgery, such as severe thrombocytopenia and anaemia. The five cases of OP associated with haematological malignancies were confirmed through bronchoscopic and CT-guided biopsy, and all of these patients tolerated the minimally invasive biopsy well. This case series revealed that bronchoscopic and CT-guided percutaneous biopsies were safe and effective in the diagnosis of OP with haematological malignancies. Because of the limited sample size and undiagnosed pathological pattern, the OP patient with underlying TCL in our study underwent bronchoscopic biopsy 2 times, and the diagnosis was confirmed through CT-guided percutaneous biopsy. It seems that CT-guided percutaneous biopsy may be more effective in subpleural lesions than bronchoscopic biopsy.
As described in a previous study,2 steroid treatment led to a favourable outcome in this case series. To discuss steroid treatment in OP associated with haematological malignancies, the authors searched for OP and haematological malignancies in PubMed (Table 2). Most of the identified studies used steroids to treat OP and demonstrated a favourable response. However, other studies found that OP with haematological malignancies could be steroid resistant and improved with SCT18 or without treatment.7 The follow-up demonstrated unfavourable clinical outcomes associated with non-steroidal treatment,7 indicating that observation without treatment may not be suitable for OP with haematological malignancies. Furthermore, our study excluded other possible causative factors, such as infection, could progress after steroid treatment. For the above reasons, the authors suggest that steroids may be the first-line drug treatment for OP associated with haematological malignancies. Due to the limitation of sample size, a prospective study with larger sample size might provide stronger evidence.
Conclusion
Collectively, our findings provide support that haematological malignancies may be a causative factor for OP and that biopsy may be an effective and safe method to confirm the diagnosis. Although OP associated with haematological malignancies may more frequently develop into severe cases, OP lesions seem to be steroid responsive with favourable outcomes during follow-up.
Data Sharing Statement
The datasets analysed during the current study are not publicly available to protect patient privacy but are available from the corresponding author on reasonable request.
Ethics Approval and Consent to Participate
The study was approved and supervised by the Medical Research Ethics Committee of the Second Xiangya Hospital, Central South University. Informed consent was waived. This research was performed in accordance with the Declaration of Helsinki.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This study was supported by National Key Clinical Specialist Construction Programs of China; the National Natural Science Foundation of China (81370143, 81170036, 81400032); Beijing Bethuen Charitable Foundation (BJ-RW2020011J) and the Natural Science Foundation of Hunan Province (09JJ3036, 2019JJ50877).
Disclosure
The authors declare that they have no conflicts of interest.
References
1. Enright H, Jacob HS, Vercellotti G, et al. Paraneoplastic autoimmune phenomena in patients with myelodysplastic syndromes: response to immunosuppressive therapy. Br J Haematol. 1995;91(2):403–408. doi:10.1111/j.1365-2141.1995.tb05310.x
2. Cordier J-F. Organising pneumonia. Thorax. 2000;55(4):318–328. doi:10.1136/thorax.55.4.318
3. Lamour C, Bergeron A. Non-infectious pulmonary complications of myelodysplastic syndromes and chronic myeloproliferative disorders. Rev Mal Respir. 2011;28(6):e18–27. doi:10.1016/j.rmr.2009.04.001
4. Hildebrand FL, Rosenow EC, Habermann TM, et al. Pulmonary complications of leukemia. Chest. 1990;98(5):1233–1239. doi:10.1378/chest.98.5.1233
5. Pennington JE, Feldman NT. Pulmonary infiltrates and fever in patients with hematologic malignancy: assessment of transbronchial biopsy. Am J Med. 1977;62(4):581–587. doi:10.1016/0002-9343(77)90421-1
6. Inoue D, Marumo S, Ishii H, et al. Secondary pulmonary alveolar proteinosis during corticosteroid therapy for organising pneumonia associated with myelodysplastic syndrome. BMJ Case Rep. 2019;12(9). doi:10.1136/bcr-2019-231055
7. Kobara Y, Amemiya Y, Mieno T, et al. Clinical study of three myelodysplastic syndrome (MDS) patients with BOOP-like pulmonary disease. Nihon Kokyuki Gakkai Zasshi. 2000;38(8):605–609.
8. Mokhtari M, Bach PB, Tietjen PA, et al. Bronchiolitis obliterans organizing pneumonia in cancer: a case series. Respir Med. 2002;96(4):280–286. doi:10.1053/rmed.2001.1269
9. Daniels CE, Myers JL, Utz JP, et al. Organizing pneumonia in patients with hematologic malignancies: a steroid-responsive lesion. Respir Med. 2007;101(1):162–168. doi:10.1016/j.rmed.2006.03.035
10. Travis WD, Costabel U, Hansell DM, et al. An official American Thoracic Society/European Respiratory Society statement: update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. Am J Respir Crit Care Med. 2013;188(6):733–748. doi:10.1164/rccm.201308-1483ST
11. Tanaka N, Kunihiro Y, Yujiri T, et al. High-resolution computed tomography of chest complications in patients treated with hematopoietic stem cell transplantation. Jpn J Radiol. 2011;29(4):229–235. doi:10.1007/s11604-010-0544-8
12. Mankikian J, Lioger B, Diot E, et al. Pulmonary toxicity associated with the use of lenalidomide: case report of late-onset acute respiratory distress syndrome and literature review. Heart Lung. 2014;43(2):120–123. doi:10.1016/j.hrtlng.2013.11.007
13. Alnimer Y, Salah S, Abuqayas B, et al. Azacitidine-induced cryptogenic organizing pneumonia: a case report and review of the literature. J Med Case Rep. 2016;10(1):15. doi:10.1186/s13256-016-0803-0
14. Uhlving HH, Andersen CB, Christensen IJ, et al. Biopsy-verified bronchiolitis obliterans and other noninfectious lung pathologies after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2015;21(3):531–538. doi:10.1016/j.bbmt.2014.12.004
15. Kim K, Lee MH, Kim J, et al. Importance of open lung biopsy in the diagnosis of invasive pulmonary aspergillosis in patients with hematologic malignancies. Am J Hematol. 2002;71(2):75–79. doi:10.1002/ajh.10168
16. White DA, Wong PW, Downey R. The utility of open lung biopsy in patients with hematologic malignancies. Am J Respir Crit Care Med. 2000;161(3):723–729. doi:10.1164/ajrccm.161.3.9904016
17. Dunn JC, West KW, Rescorla FJ, et al. The utility of lung biopsy in recipients of stem cell transplantation. J Pediatr Surg. 2001;36(8):1302–1303. doi:10.1053/jpsu.2001.25799
18. Asano T, Fujii N, Niiya D, et al. Complete resolution of steroid-resistant organizing pneumonia associated with myelodysplastic syndrome following allogeneic hematopoietic cell transplantation. SpringerPlus. 2014;3(1):1–5. doi:10.1186/2193-1801-3-3
19. Vieira AL, Vale A, Melo N, et al. Organizing pneumonia revisited: insights and uncertainties from a series of 67 patients. Sarcoidosis Vasc Diffuse Lung Dis. 2018;35(2):129–138. doi:10.36141/svdld.v35i2.6860
20. Dai M-S, Lee S-C, Ho C-L, et al. Impact of open lung biopsy for undiagnosed pulmonary infiltrates in patients with hematological malignancies. Am J Hematol. 2001;68(2):87–90. doi:10.1002/ajh.1158
21. Wohlrab JL, Anderson ED, Read CA. A patient with myelodyplastic syndrome, pulmonary nodules, and worsening infiltrates. Chest. 2001;120(3):1014–1017. doi:10.1378/chest.120.3.1014
22. Karamlou K, Gorn AH. Refractory sweet syndrome with autoimmune organizing pneumonia treated with monoclonal antibodies to tumor necrosis factor. J Clin Rheumatol. 2004;10(6):331–335. doi:10.1097/01.rhu.0000147053.60795.46
23. Garg R, Soud Y, Lal R, et al. Myelodysplastic syndrome manifesting as sweet’s syndrome and bronchiolitis obliterative organizing pneumonia. Am J Med. 2006;119(11):e5–7. doi:10.1016/j.amjmed.2006.03.032
24. Tomonari A, Tsukada N, Takahashi S, et al. Early-onset pulmonary complication showing organizing pneumonia pattern following cord blood transplantation in adults. Int J Hematol. 2007;85(4):364–366. doi:10.1532/IJH97.07016
25. Kamiya H, Ishikawa R, Moriya A, et al. Disseminated cryptococcosis complicated with bilateral pleural effusion and ascites during corticosteroid therapy for organizing pneumonia with myelodysplastic syndrome. Intern Med. 2008;47(22):1981–1986. doi:10.2169/internalmedicine.47.0898
26. Tzelepis E, Kampolis CF, Vlachadami I, et al. Cryptogenic organizing pneumonia in Sweet’s syndrome: case report and review of the literature. Clin Respir J. 2016;10(2):250–254. doi:10.1111/crj.12206
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.