Back to Journals » International Journal of Nanomedicine » Volume 14
CeO2NPs relieve radiofrequency radiation, improve testosterone synthesis, and clock gene expression in Leydig cells by enhancing antioxidation
Authors Qin F, Shen T, Cao H, Qian J, Zou D, Ye M, Pei H
Received 27 February 2019
Accepted for publication 15 May 2019
Published 24 June 2019 Volume 2019:14 Pages 4601—4611
DOI https://doi.org/10.2147/IJN.S206561
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Lei Yang
Fenju Qin,1,2 Tao Shen,1 Honglong Cao,3 Junchao Qian,4 Dan Zou,1 Mingkang Ye,1 Hailong Pei2
1Department of Biotechnology and Bioengineering, Suzhou University of Science and Technology, Suzhou 215009, People’s Republic of China; 2School of Radiation Medicine and Protection, Medical College of Soochow University, Suzhou 215123, People’s Republic of China; 3School of Electronic & Information Engineering, Soochow University, Suzhou 215006, People’s Republic of China; 4Jiangsu Key Laboratory for Environment Functional Materials, Suzhou University of Science and Technology, Suzhou 215009, People’s Republic of China
Introduction: The ratio of Ce3+,/Ce4+, in their structure confers unique functions on cerium oxide nanoparticles (CeO2NPs) containing rare earth elements in scavenging free radicals and protecting against oxidative damage. The potential of CeO2NPs to protect testosterone synthesis in primary mouse Leydig cells during exposure to 1,800 MHz radiofrequency (RF) radiation was examined in vitro.
Methods: Leydig cells were treated with different concentrations of CeO2NPs to identify the optimum concentration for cell proliferation. The cells were pretreated with the optimum dose of CeO2NPs for 24 hrs and then exposed to 1,800 MHz RF at a power density of 200.27 μW/cm2, (specific absorption rate (SAR), 0.116 W/kg) for 1 hr, 2 hrs, or 4 hrs. The medium was used to measure the testosterone concentration. The cells were collected to determine the antioxidant indices (catalase [CAT], malondialdehyde [MDA], and total antioxidant capacity [T-AOC]), and the mRNA expression of the testosterone synthase genes (Star, Cyp11a1, and Hsd-3β) and clock genes (Clock, Bmal1, and Rorα).
Results: Our preliminary result showed that 128 μg/mL CeO2NPs was the optimum dose for cell proliferation. Cells exposed to RF alone showed reduced levels of testosterone, T-AOC, and CAT activities, increased MDA content, and the downregulated genes expression of Star, Cyp11a1, Hsd-3β, Clock, Bmal1, and Rorα. Pretreatment of the cells with 128 μg/mL CeO2NPs for 24 hrs followed by RF exposure significantly increased testosterone synthesis, upregulated the expression of the testosterone synthase and clock genes, and increased the resistance to oxidative damage in Leydig cells compared with those in cells exposed to RF alone.
Conclusion: Exposure to 1,800 MHz RF had adverse effects on testosterone synthesis, antioxidant levels, and clock gene expression in primary Leydig cells. Pretreatment with CeO2NPs prevented the adverse effects on testosterone synthesis induced by RF exposure by regulating their antioxidant capacity and clock gene expression in vitro. Further studies of the mechanism underlying the protective function of CeO2NPs against RF in the male reproductive system are required.
Keywords: CeO2NPs radiofrequency radiation, Leydig cell, testosterone synthesis, clock genes, antioxidant
Introduction
Exposure to radiofrequency (RF) radiation at frequencies of 850–2,100 MHz, which are used for cellular phones, is increasing rapidly in modern life.1 Consequently, public concerns about the effects of RF radiation on health are also increasing rapidly throughout the world.2 Infertility is common in approximately 15% of the couples globally, and this is attributed to environmental factors, including exposure to RF radiation.3–5 In humans, the testis is located in the area of RF exposure because men usually carry their mobile phones in their trouser pockets or waist-bands or keep computers in their laps. Reports have suggested that RF exposure affects testicular development, damages the seminiferous tubules, and reduces the number of Leydig cells in rats.6 Testosterone, the hormone secreted by Leydig cells in testes, plays a pivotal role in sustaining structural and functional integrity of male reproductive organs.7 The effects of RF exposure on testosterone, male sex hormone, in animals were inconsistent: some studies showed decreased levels in rabbits and rats while others reported increased levels in mice and humans.8–11 The observation in our previous studies indicated that RF exposure inhibited the testosterone synthesis and that clock gene Rorα was involved in its regulatory mechanism.12 There were suggestions that free radical-mediated pathway might be involved in the adverse effects of RF exposure on testosterone secretion.13
Cerium oxide nanoparticles (CeO2NPs) are oxides of rare earth elements and attracted much attention in their biological applications due to the switch between Ce3+ and Ce4+ on the surface of CeO2NPs structure.14 The preparation method, particle size, exposure route, and cell type studied have been found to influence the biological effects of CeO2 NPs.15 Particles with smaller size have a larger surface area per mass unit and hence, they were found to be potentially more active as antioxidant and UV shield.16,17 In our previous studies, CeO2NPs was found to improve the antioxidant capacity of tissue in mice.18 There were reports suggesting that some strong antioxidant may act as a protective agent and keep the male reproductive system from adverse effects of the RF exposure through the elimination of the highly reactive free radicals.19,20 Hence, in this study, the antioxidant capacity of CeO2NPs was investigated on testosterone synthesis and adjusting the clock genes expression in primary Leydig cells under RF exposure.
Materials and methods
CeO2NPs
The CeO2NPs were obtained from Sigma-Aldrich (Shanghai, China) (Product Number: 544,841, APS: <25 nm and purity >99% trace metal basis). Their characteristics were detected by scanning electron microscopy (SEM, Quanta FEG 250, Hillsboro, OR, USA), transmission electron microscope (TEM, JEOL 2100, Tokyo, Japan), and X-ray powder diffractometry (XRD8 Advance X-ray diffractometer; Bruker AXS Endeavor, Billerica, USA) and presented in Figure 1A, B and D. The X-ray diffraction of the precipitated material showed cubic crystals. The intense peaks from the XRD test corresponded to the diffraction peak of CeO2. The size distribution of CeO2NPs with a 18–40 nm size range was analyzed fom the transmission electron microscopy images as shown in Figure 1C, and the average size of CeO2NP was 27.62 ± 3.01 nm.
Primary Leydig cells separation and identification
The experiments were approved (number, 201701A323) by IACUC (Institutional Animal Care and Use Committee) of Soochow University, China. All procedure for the animal experiment was conducted according to the guidelines of IACUC of Soochow University. Primary Leydig cells used in the experiments were isolated from the testis of 9-week-old C57 male mice who exhibited circadian rhythmicity by 0.05% Collagenase I. Approximately 5.0×106 cells were mixed with 3 mL complete medium consisting of 1:1 DMEM/F-12 (Invitrogen, Thermo Fisher, Shanghai, China), 2.5% fetal bovine serum (FBS) and 5% horse serum (Gibco, Grand Island, NY, USA) and cultured in an incubator maintaining 37°C, 5% CO2 atmosphere, and 95% air. The cells were cultured in vitro for 24 hrs at which time they were processed for the cell purity identification using HSD-3β-specific histochemical staining method.21 Those with dark blue particles were identified as viable in Leydig cells.
Experimental design
Firstly, cell proliferation assay was used to evaluate the most suitable addition dose of CeO2NPs according to cell proliferation rate. Secondly, cells which were pretreated with/without CeO2NPs (the optimal addition level obtained from the first step) for 24 hrs were then exposed to 1,800 MHz RF for 1 hr, 2 hrs, or 4 hrs. Immediately after RF exposure, the culture medium was collected to measure testosterone concentration. The cells were kept to determine the antioxidant levels (MDA, CAT, and T-AOC) and to extract the total RNA for real-time PCR to examine mRNA expression of testosterone synthesis and clock genes. The samples were investigated from these groups of Leydig cells as Table 1.
 | Table 1 Experimental design groups |
Cell proliferation assay
Approximately 5.0×106 Leydig cells were seeded in a separate sterile 35-mm petri dish in quadruplicate, and semi-confluent cells were starved in DMEM/F12 medium (without serum for 16 hrs) to synchronize at G0/G1 phase. Then, the cells were cultured in complete medium with 2.5% FBS and 5% horse serum for 8 hrs and then distributed into separate 96-well plates, 10 μL CeO2NPs solution diluted by dispersant (PBS with 0.5% DMSO) was added to the medium of cells by 10 addition gradients as following: 0 (Blank), 0 (Solvent), 4, 8, 16, 32, 64, 128, 256, 512, and 1,024 μg/mL in quadruplicate. MTT assay [3-(4, 5- dimethyl-thiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT)-based colorimetric method] was performed to find the optimal concentration of CeO2NPs for the proliferation of Leydig cells. Twenty microliters MTT (Sigma-Aldrich, China) solution (5 mg/mL in PBS) was added, and the cells were further incubated for 4 hrs at 37°C. Then, 150 μL DMSO was added to dissolve of formazan crystals. Finally, the absorbance in each well was measured at 490 nm using a microplate reader (Beckman, San Jose, CA, USA). Cell proliferation rate was used as the evaluation index of the most suitable addition dose of CeO2NPs: Cell proliferation rate (%) = (OD value in the experimental group/OD value in the solvent group) ×100.
RF exposure system and dosimetry
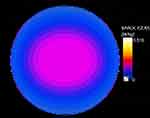
In this experiment, the RF exposure system consists of RF generator (E4438C ESG; Agilent Technologies Inc., Palo Alto, CA, USA) and narrow band amplifier (SN1012; HD Communications Corp, New York, NY, USA). The sensors and fans of the exposure system were connected to a PC that monitors the power densities (200.27 μW/cm2) during RF exposure and maintain a constant temperature and environment (37°C, 5% CO2). The RF exposure system was set to produce an electromotive force similar to that emitted by the global system for mobile communications mobile phones with a frequency of 1,800 MHz. As shown in Table 1, the cells were pretreated with/without CeO2NPs for 24 hrs, and then exposed to 1,800 MHz RF radiation at 200.27 μW/cm2 power densities for 1 hr, 2 hrs, or 4 hrs, respectively. The special absorption rate (SAR) was evaluated and computed using the 3D-FDTD full-wave electromagnetic simulation software, Sim4life 4.0 (ZMT Zurich MedTech, Zürich, Switzerland), and the specific absorption rate distribution inside the culture medium is shown in Figure 2. As shown in Figure 2, the maximum SAR was 0.519 W/kg, and the mass-average SAR was 0.116 W/kg simulated in Sim4life 4.0.
Testosterone assay
Cell culture medium was collected after RF exposure for 1, 2, and 4 hrs. The medium was centrifuged at 3,000 rpm for 10 mins, and the supernatants were kept to evaluate for testosterone concentration using the Beckman DTX-880 Multimode Plate Reader (Beckman Coulter, San Jose, CA, USA). The testosterone levels were obtained using enzyme-immunoassay ELISA kit (Elabscience, Wuhan, China), and the absorbance was read at 450 nm.22 All samples were read in duplicate, and the coefficient of variation was less than 12% within and between measurements. Duplicate samples were used for each assay and repeated three times.
Antioxidant levels
Cells were collected after RF exposure for 1, 2, or 4 hrs with trypsinization, washed and centrifuged at 1,000× g at 4°C for 20 mins. Then, the cells were sonicated to disrupt the cell wall, centrifuged at 8,000× g for 10 mins at 4°C, the supernatant was kept on ice to determine the CAT activities, MDA content, and T-AOC levels using kits according to manufacturers’ instructions (Suzhou Comin, Suzhou, China), respectively.
Real-time PCR
The Leydig cells were collected after RF exposure for 1, 2, or 4 hrs for real-time PCR. Total RNA was isolated using the Mini Kit (Qiagen, Duesseldorf, Germany) and, the quality and quantity were determined using BioPhotometer (Analytik Jena AG, Jena, Germany). Quantitative real-time PCR was conducted on the QuantStudio™ 7 Flex Real-Time PCR system (Life Technologies, Carlsbad, CA, USA) using 2.5X SYBR Green Abstart One Step RT-PCR Mix (Sangon Biotech, Shanghai, China) according to the manufacturer’s instructions. Primers were designed and synthesized by Invitrogen (Life Technologies, Shanghai, China) and the forward and reverse sequences are presented in Table 2. All values were normalized to a mice housekeeping gene β-actin. The fold change in mRNA was calculated by the ΔΔCt method (fold =2ΔΔCt). All samples were tested in duplicates.
 | Table 2 Primer sequence for real-time PCR |
Statistical analysis
All data were presented as mean ± SD (standard deviation). Statistical analyses were performed by one-way analysis of variance between the Blank/Solvent/CeO2NPs/RF/CeO2NPs+ RF groups with SPSS 22 (SPSS Inc, Chicago, IL, USA). When appropriate, LSD test was carried out. A statistical P-value of <0.05 between two groups was indicative of significant difference.
Results
Identification of primary Leydig cell
Figure 3A and B shows the Leydig cells stained with HSD-3β-specific dye after culturing in vitro for 24 hrs. The cytoplasm of most cells was blue-black with dark blue particles, and more than 95% of the cells are stained. Thus, the purity of primary Leydig cells was above 95%.
 | Figure 3 Stain of HSD-3β on primary leydig cells (A, 20×; B, 40×). Abbreviation: HSD-3β, 3β-Hydroxysteroid dehydrogenase. |
Proliferation of Leydig cells affected by different does CeO2NPs
The proliferation rate of Leydig cells which were treated with different addition doses CeO2NPs [0 (Blank), 0 (Solvent), 4, 8, 16, 32, 64, 128, 256, 512, and 1,024 μg/mL] is shown in Figure 4. In terms of the cell proliferation rate, there is no statistical difference between Blank and Solvent groups. The proliferation rate was significantly increased to 127.72%, 181.22%, and 148.53% in cells treated with 64, 128, and 256 μg/mL CeO2NPs, respectively, compared with the solvent (0 μg/mL) group (100%). There was no statistical difference in other CeO2NPs-treated groups. Therefore, the addition concentration of 128 μg/mL CeO2NPs was considered optimum for the proliferation of Leydig cells and also as the protective dose for RF exposure in the next experiments.
Testosterone
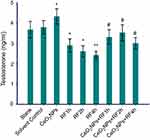
Testosterone synthesis reflects the function of primary Leydig cells, which is easily affected by environmental factors. In this study, for testosterone concentrations in cells medium, solvent control group is no statistical difference from the blank group. Compared with the solvent control group, CeO2NPs (128 μg/mL) addition increased the testosterone concentrations, while RF exposure for 1, 2, and 4 hrs significantly diminished testosterone levels. Cells pretreated with 128 μg/mL CeO2NPs for 24 hrs and then exposed to RF for 1, 2, and 4 hrs, showed increased testosterone concentration compared to that of RF exposure alone groups, respectively (Figure 5). The results showed 128 μg/mL CeO2NPs pretreatment could protect RF exposure-induced toxicity on testosterone synthesis.
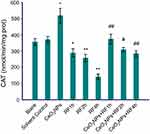
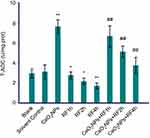
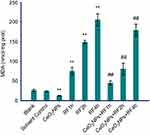
Antioxidant levels (CAT, MDA, and T-AOC)
The data in Figures 6–8 show the results of CAT activities, MDA content, and T-AOC levels. For three antioxidant parameters, there is no difference between the blank group and the solvent control group. There were significant differences between solvent control and CeO2 NP/RF-exposed cells. Cells treated with 128 µg/mL CeO2NPs showed increased the CAT activities and T-AOC levels, reduced MDA content in primary Leydig cells, while RF exposure induced the opposite change on antioxidant levels. Compared to cells exposed to RF for 1, 2, and 4 hrs groups, CeO2NPs + RF each group reduced the MDA content, increased CAT activities and T-AOC levels. The changes in CAT activities, MDA content, and T-AOC levels observed after RF exposure 1, 2, and 4 hrs were reversed by pretreatment with 128 µg/mL CeO2 NPs for 24 hrs.
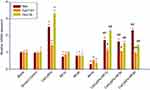
Testosterone synthase genes expression
The data in Figure 9 show the results of the real-time PCR test for Star, Cyp11a1, and Hsd-3β. From the results, there is no difference between the blank group and the solvent control group in the three gene expression. Compared to the solvent control group, exposure of the cells to 128 µg/mL CeO2 NPs quite significantly increased the mRNA expression of testosterone synthesis genes Star, Cyp11a1, and 3β-Hsd in primary Leydig cells, while RF exposure for 2or 4 hrs induced the distinct downregulation of the three testosterone synthesis genes expression. CeO2NPs + RF groups at all RF exposure times distinctly enhanced the genes expression of testosterone synthesis Star, Cyp11a1, and Hsd-3β in primary Leydig cells compared to RF exposure alone for 1, 2, and 4 hrs, respectively. The downregulation effects of RF exposure effect on the mRNA expression of testosterone synthesis genes, Star, Cyp11a1, and Hsd-3β could be effectively reversed by 128 µg/mL CeO2NPs pretreatment for 24 hrs.
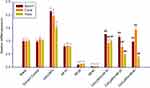
Clock genes expression
The results of real-time PCR for clock genes Baml1, Clock, and Rorα mRNA expression are shown in Figure 10. As reported above indicators, no difference appeared between the blank group and solvent control group in the expression of clock genes. As compared to the solvent control group, Rorα, Clock, and Baml1 mRNA expression remarkably increased in CeO2NPs treatment group. There are significant decreases on the mRNA expressions of Baml1, Clock, and Rorα in the RF exposure 2or 4 hrs group compared to the solvent control cells, and RF exposure 1 hr also reduced the gene expressions of two clock genes Clock and Rorα except for Baml1 expression. As for the genes expression of Rorα, Clock, and Baml1, CeO2NPs + RF each group is higher than that of RF exposure alone for 1, 2, and 4 hrs, respectively. The downregulation toxicity of RF exposure on the mRNA expression of clock genes Rorα, Clock, and Baml1 could be effectively reversed by 128 µg/mL CeO2NPs pretreatment 24 hrs.
Discussion
There were several reports suggesting RF exposure effects on various cellular systems, and highly reactive free radicals were implicated in adverse biological effects (oxidative damage) induced by RF.23–26 The aims of this study were to examine whether 1,800 MHz RF radiation induces oxidative stress and promote lipid peroxidation and also to investigate the role of CeO2NPs, which have antioxidant properties, against possible testosterone synthesis toxicity in primary Leydig cells induced by 1,800 MHz RF radiation in vitro.
Leydig cells are located in interstitial of seminiferous tubules in testis and play an important role in the paracrine regulation of spermatogenesis and, are responsible for the synthesis of testosterone.27 Testosterone has significant effects during the growth and development of the male reproductive organs.28 Long-term exposure to environmental toxins was suggested to be responsible for the decline of testosterone that further induced the defective sperm.29 It is generally considered that Leydig cells are sensitive to the environmental stress, such as heat, radiation, and exposure to heavy metals.30,31 There were reports indicating RF radiation from cell phone, and other electronic equipment may have a negative influence on Leydig cells.25,32 In our previous studies, we found 1,800 MHz RF exposure reduced the level of testosterone synthesis in mice and rats.12,33 The results in the present study showed that 1,800 MHz RF exposure for 1, 2, and 4 hrs negatively affected testosterone synthesis in primary Leydig cells.
In order to investigate the potential mechanisms of RF exposure effect on primary Leydig cells, the expression of genes involved in testosterone synthesis (Star, Cyp11a1, Hsd-3β) was examined, and all were found to be altered. Steroidogenic acute regulatory protein (StAR) is able to mediate the production of steroid hormones which is initiated by translocation of cholesterol through the mitochondrial membrane from the outer to the inner.34 Cholesterol was metabolized into pregnenolone by cytochrome P450 Family 11 subfamily A member 1 (CYP11A1), and the hormonally and developmentally regulated expression of CYP11A1 is mainly driven by a variety of trans-acting factors.35 Due to the close correlation with HSD-3β, pregnenolone transfers from the mitochondria into the smooth endoplasmic reticulum.36 In this study, the mRNA expression levels of Star, Cyp11a1, and Hsd-3β in Leydig cells exposed to RF radiation for 2and 4 hrs decreased significantly, which could be responsible for the inhibition of testosterone synthesis.
In our previous study, we found that RF exposure inhibited testosterone synthesis through CaMKI/RORα signaling pathway. RORα (the retinoid-related receptor alpha) is a critical member of the core clock gene machinery, its ROR response elements (ROREs) have been identified in several clock genes including Bmal1 and Clock, indicating that ROREs are essential for rhythmic transcriptional regulation of Bmal1 and Clock.37 BMAL1 as an essential component of the circadian pacemaker in mammals can regulate the rate-limiting step of steroidogenesis by enhancing the transcription of Star gene.38 Biological clock genes also regulate the expression of cytochrome P450 (CYP) gene family members in which Cyp11family including CYP11A1.39 The gene expression of Hsd-3β is also under transcriptional control of the circadian clock.40 Thus, testosterone synthesis can be changed by clock genes expression. In this present study, the clock genes Rorα, Clock, and Baml1 as well as the three steroidogenic genes, viz., Star, Cyp11a1, Hsd-3β, were downregulated in mouse primary Leydig cells exposed to 1,800 MHz RF radiation for 1, 2, and 4 hrs.
Damage in biological systems induced RF emitted from cellular mobile phones may be due to the accumulation of free radicals which enhance oxidative stress, and change the antioxidant defense systems of tissues.41 Under oxidative stress, circadian rhythmicity is usually affected and the expressions of the key circadian gene are directly inhibited.42 In the present study, 1,800 MHz RF exposure negatively affected testosterone synthesis and clock genes expression, decreased the CAT and T-AOC levels, increased the MDA content, changed the antioxidant capacity of primary Leydig cells, and induced the oxidative stress. Thus, the results demonstrated that the inhibition on testosterone synthesis induced by RF exposure may be due to oxidative stress and downregulation of clock genes.
CeO2NPs have antioxidant properties due to their variable particle sizes, crystal structures, and surface chemistries, and have also been demonstrated that are able to quench ROS produced and protect cells against oxidative damage due to its free radical-scavenging properties.43,44 In the present study, application alone of CeO2 NPs increased the CAT activities and T-AOC levels, decreased the MDA content, which agreed with the literature that reported CeO2NPs reduced the oxidative stress.45 Meanwhile, these nanoparticles further relieved the oxidative stress induced by RF, accompanied by the upregulation of clock genes Rorα, Clock, and Baml1 in this present study. These results were consistent with the report which indicated circadian clock is involved in regulating the response of a cell to oxidative stress.46
Previous studies reported that CeO2NPs materials have positive effects on reproductive cells in sheep and are capable of promoting reproductive performance in rabbits.47,48 It is also reported citrate-stabilized CeO2NPs help to accelerate the proliferation of primary mouse embryonic fibroblasts in vitro.49 In the present study, CeO2NPs treatment alone promoted the proliferation of mouse primary Leydig cells, enhanced the testosterone synthesis and clock genes expression, and increased the antioxidant capacity. Further, we found that pretreatment of the cells with CeO2NPs for 24 hrs reduced the oxidative damage and the clock genes downregulation caused by RF exposure and thus, improved the testosterone synthetase genes expression and testosterone secretion.
As there are always two sides to everything, CeO2NPs also showed some toxicities.50,51 According to our previous studies in animals, it must be noted that doses play a crucial role in the benefits or toxicity of CeO2NPs on the reproductive system.18,52 In the present study, we set ten dose gradients to screen the optimum addition dosage of CeO2NPs by proliferation rate assay of Leydig cells. The results found that the cell proliferation rate was increased most significantly at the dose 128 μg/mL but then gradually decreased above the 128 μg/mL. It suggested there is an optimum addition for the proliferation of mouse primary Leydig cells, and CeO2NPs above the optimum addition level may induce a declining cell proliferation rate, even inhibit the cells proliferation as the dose constantly increases.
Conclusion
In summary, our study provided some evidence that 1,800 MHz RF exposure may first induce the oxidative damage in primary Leydig cells and then, weaken the expression of clock genes Clock, Bmal1, and Rorα which in turn, downregulates its target genes involved in testosterone synthesis, including Star, Cyp11a1, and Hsd-3β, resulted in reduced testosterone production. Preexposure of the cells to CeO2NPs also reduced RF radiation effects, improves testosterone synthesis and clock genes expression in Leydig cells via enhancing antioxidant levels.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Aydogan F, Unlu I, Aydin E, et al. The effect of 2100 MHz radiofrequency radiation of a 3G mobile phone on the parotid gland of rats. Am J Otolaryngol. 2015;36(1):39–46. doi:10.1016/j.amjoto.2014.10.001
2. Hardell L, Carlberg M, Hedendahl LK. Radiofrequency radiation from nearby base stations gives high levels in an apartment in Stockholm, Sweden: a case report. Oncol Lett. 2018;15(5):7871–7883.
3. Poongothai J, Gopenath TS, Manonayaki S. Genetics of human male infertility. Singapore Med J. 2009;50(4):336–347.
4. Bendayan M, Alter L, Swierkowski-Blanchard N, et al. Environment and lifestyle: impacts on male fertility? Gynecol Obstet Fertil Senol. 2018;46(1):47–56.
5. Mortazavi S, Parsanezhad ME, Kazempour M, Davari M, Ghahramani P, Kazempour M. Male reproductive health under threat: short term exposure to radiofrequency radiations emitted by common mobile jammers. J Hum Reprod Sci. 2013;6(2):124–128. doi:10.4103/0974-1208.117178
6. Tenorio BM, Jimenez GC, Morais RN, Torres SM, Albuquerque Nogueira R, Silva Junior VA. Testicular development evaluation in rats exposed to 60 Hz and 1 mT electromagnetic field. J Appl Toxicol. 2011;31:223–230. doi:10.1002/jat.v31.3
7. Teerds KJ, Huhtaniemi IT. Morphological and functional maturation of Leydig cells: from rodent models to primates. Hum Reprod Update. 2015;21:310–328. doi:10.1093/humupd/dmv008
8. Sarookhani MR, Rezaei MA, Safari A, Zaroushani V, Ziaeiha M. The influence of 950 MHz magnetic field (mobile phone radiation) on sex organ and adrenal functions of male rabbits. Afr J Biochem. 2011;5(2):65–68.
9. Sehitoglu I, Tumkaya L, Kalkan Y, et al. Biochemical and histopathological effects on the rat testis after exposure to electromagnetic field during fetal period. Arch Esp Urol. 2015;68(6):562–568.
10. Forgács Z, Somosy Z, Kubinyi G, et al. Effect of whole-body 1800MHz GSM-like microwave exposure on testicular steroidogenesis and histology in mice. Reprod Toxicol. 2006;22(1):111–117. doi:10.1016/j.reprotox.2006.04.011
11. Gutschi T, Mohamad Al-Ali B, Shamloul R, Pummer K, Trummer H. Impact of cell phone use on men’s semen parameters. Andrologia. 2011;43(5):312–316. doi:10.1111/and.2011.43.issue-5
12. Qin F, Cao H, Yuan H, et al. 1800 MHz radiofrequency fields inhibits testosterone production via CaMKI/RORα pathway. Reprod Toxicol. 2018;81:229–236. doi:10.1016/j.reprotox.2018.08.014
13. Shahin S, Mishra V, Singh SP, Chaturvedi CM. 2.45-GHz microwave irradiation adversely affects reproductive function in male mouse, Mus musculus by inducing oxidative and nitrosative stress. Free Radic Res. 2014;48(5):511–525. doi:10.3109/10715762.2014.960411
14. Nelson BC, Johnson ME, Walker ML, Riley KR, Sims CM. Antioxidant cerium oxide nanoparticles in biology and medicine. Antioxidants (Basel). 2016;5(2):15. doi:10.3390/antiox5020015
15. Gagnon J, Fromm KM. Toxicity and protective effects of cerium oxide nanoparticles (Nanoceria) depending on their preparation method, particle size, cell type, and exposure route. Eur J Inorg Chem. 2015;2015(27):4510–4517. doi:10.1002/ejic.201500643
16. Caputo F, De Nicola M, Sienkiewicz A, et al. Cerium oxide nanoparticles, combining antioxidant and UV shielding properties, prevent UV-induced cell damage and mutagenesis. Nanoscale. 2015;7(38):15643–15656. doi:10.1039/C5NR03767K
17. Szuromi P. Dispersing small, bimetallic nanoparticles. Science. 2017;358(6369):1397–1399.
18. Zou D, Chu X, Lu W, et al. Effects of cerium oxide nanoparticles on cognitive function in 48 h sleep deprived male mice. J Hyg Res. 2018;47(1):113–118. Chinese
19. Avci B, Akar A, Bilgici B, Tunçel ÖK. Oxidative stress induced by 1.8 GHz radio frequency electromagnetic radiation and effects of garlic extract in rats. Int J Radiat Biol. 2012;88(11):799–805. doi:10.3109/09553002.2012.711504
20. Oksay T, Naziroğlu M, Doğan S, Güzel A, Gümral N, Koşar PA. Protective effects of melatonin against oxidative injury in rat testis induced by wireless (2.45 GHz) devices. Andrologia. 2014;46(1):65–72. doi:10.1111/and.12080
21. Abarikwu SO, Farombi EO, Kashyap M, Pant AB. Atrazine induces transcriptional change in steroidogenesis marker genes in primary cultures of rat Leydig cells. Toxicol In Vitro. 2011;25:1588–1595. doi:10.1016/j.tiv.2011.06.002
22. Yu Y, Han Y, Niu R, et al. Ameliorative effect of VE, IGF-I, and hCG on the fluoride-induced testosterone release suppression in mice Leydig cells. Biol Trace Elem Res. 2018;181(1):95–103. doi:10.1007/s12011-017-1057-4
23. Yakymenko I, Tsybulin O, Sidorik E, Henshel D, Kyrylenko O, Kyrylenko S. Oxidative mechanisms of biological activity of low-intensity radiofrequency radiation. J Bioelectricity. 2016;35(2):17.
24. Lai H, Singh NP. Melatonin and a spin-trap compound block radiofrequency electromagnetic radiation-induced DNA strand breaks in rat brain cells. Bioelectromagnetics. 2015;18(6):446–454. doi:10.1002/(SICI)1521-186X(1997)18:6<446::AID-BEM7>3.0.CO;2-2
25. Lin YY, Wu T, Liu JY, et al. 1950MHz Radio frequency electromagnetic radiation inhibits testosterone secretion of mouse Leydig cells. Int J Environ Res Public Health. 2017;15(1):17. doi:10.3390/ijerph15010017
26. Zuo WQ, Hu YJ, Yang Y, et al. Sensitivity of spiral ganglion neurons to damage caused by mobile phone electromagnetic radiation will increase in lipopolysaccharide induced inflammation in vitro model. J Neuroinflammation. 2015;12:105. doi:10.1186/s12974-015-0300-1
27. Zirkin BR, Papadopoulos V. Leydig cells: formation, function and regulation. Biol Reprod. 2018;99(1):101–111. doi:10.1093/biolre/ioy059
28. Pomara C, Barone R, Marino Gammazza A, et al. Effects of nandrolone stimulation on testosterone biosynthesis in Leydig cells. J Cell Physiol. 2016;231(6):1385–1391. doi:10.1002/jcp.25173
29. Dasdag S, Tas M, Akdag MZ, Yegin K. Effect of long-term exposure of 2.4 GHz radiofrequency radiation emitted from Wi-Fi equipment on testes functions. Electromagn Biol Med. 2015;34(1):37–42. doi:10.3109/15368378.2013.869752
30. Wang Y, Chen F, Ye L, Zirkin B, Chen H. Steroidogenesis in Leydig cells: effects of aging and environmental factors. Reproduction. 2017;154(4):R111–122. doi:10.1530/REP-17-0064
31. Shetty G, Zhou W, Weng CC, Shao SH, Meistrich ML. Leydig cells contribute to the inhibition of spermatogonial differentiation after irradiation of the rat. Andrology. 2016;4(3):412–424. doi:10.1111/andr.12110
32. Saygin M, Caliskan S, Karahan N, Koyu A, Gumral N, Uguz A. Testicular apoptosis and histopathological changes induced by a 2.45 GHz electromagnetic field. Toxicol Ind Health. 2011;27(5):455–463. doi:10.1177/0748233710389851
33. Qin F, Zhang J, Cao H, et al. Effects of 1800-MHz Radiofrequency fields on circadian rhythm of plasma melatonin and testosterone in male rats. J Toxicol Environ Health. 2012;75(18):1120–1128. doi:10.1080/15287394.2012.699846
34. Rathor PK, Bhat IA, Rather MA, et al. Steroidogenic acute regulatory protein (StAR) gene expression construct: development, nanodelivery and effect on reproduction in air-breathing catfish, clarias batrachus. Int J Biol Macromol. 2017;104(Pt A):1082–1090.
35. Xu Q, Song Y, Chen Y, et al. Molecular cloning and expression patterns of the cholesterol side chain cleavage enzyme (CYP11A1) gene during the reproductive cycle in goose (Anas cygnoides). J Anim Sci Biotechnol. 2015;6:54. doi:10.1186/s40104-015-0053-9
36. Li L, Chen X, Zhu Q, et al. Disrupting androgen production of Leydig cells by resveratrol via direct inhibition of human and rat 3β-hydroxysteroid dehydrogenase. Toxicol Lett. 2014;226(1):14–19. doi:10.1016/j.toxlet.2014.01.022
37. Cook DN, Kang HS, Jetten AM. Retinoic acid-related orphan receptors (RORs): regulatory functions in immunity, development, circadian rhythm, and metabolism. Nucl Receptor Res. 2015;2:101185. doi:10.11131/2015/101185
38. Fahrenkrug J, Georg B, Hannibal J, Jørgensen HL. Altered rhythm of adrenal clock genes, StAR and serum corticosterone in VIP receptor 2-deficient mice. J Mol Neurosci. 2012;48(3):584–596. doi:10.1007/s12031-012-9804-7
39. Johnson BP, Walisser JA, Liu Y, et al. Hepatocyte circadian clock controls acetaminophen bioactivation through NADPH-cytochrome P450 oxidoreductase. Proc Natl Acad Sci USA. 2014;111(52):18757–18762. doi:10.1073/pnas.1421708111
40. Doi M, Takahashi Y, Komatsu R, et al. Salt-sensitive hypertension in circadian clock-deficient Cry-null mice involves dysregulated adrenal Hsd3b6. Nat Med. 2010;16(1):67–74. doi:10.1038/nm1110-1167
41. Ozguner F, Oktem F, Ayata A, Koyu A, Yilmaz HR. A novel antioxidant agent caffeic acid phenethyl ester prevents long-term mobile phone exposure-induced renal impairment in rat. Mol Cell Biochem. 2005;277(1–2):73–80. doi:10.1007/s11010-005-5074-9
42. Yang G, Wright CJ, Hinson MD, et al. Oxidative stress and inflammation modulate Rev-erbalpha signaling in the neonatal lung and affect circadian rhythmicity. Antioxid Redox Signal. 2014;21(1):17–32. doi:10.1089/ars.2013.5539
43. Eriksson P, Tal AA, Skallberg A, et al. Cerium oxide nanoparticles with antioxidant capabilities and gadolinium integration for MRI contrast enhancement. Sci Rep. 2018;8(1):6999. doi:10.1038/s41598-018-25390-z
44. Rubio L, Annangi B, Vila L, Hernández A, Marcos R. Antioxidant and anti-genotoxic properties of cerium oxide nanoparticles in a pulmonary-like cell system. Arch Toxicol. 2015;90(2):269–278. doi:10.1007/s00204-015-1468-y
45. DeCoteau W, Heckman KL, Estevez AY, et al. Cerium oxide nanoparticles with antioxidant properties ameliorate strength and prolong life in mouse model of amyotrophic lateral sclerosis. Nanomedicine. 2016;12(8):2311–2320. doi:10.1016/j.nano.2016.06.009
46. Magnone MC, Langmesser S, Bezdek AC, Tallone T, Rusconi S, Albrecht U. The mammalian circadian clock gene per2 modulates cell death in response to oxidative stress. Front Neurol. 2015;5:289. doi:10.3389/fneur.2014.00289
47. Ariu F, Bogliolo L, Pinna A, et al. Cerium oxide nanoparticles (CeO2NPs) improve the developmental competence of in vitro-matured prepubertal ovine oocytes. Reprod Fertil Dev. 2017;29(5):1046–1056. doi:10.1071/RD15521
48. Akinmuyisitan IW, Gbore FA, Adu OA. Reproductive performance of growing female rabbits (Oryctolagus cuniculus) fed diets supplemented with cerium oxide. Journal of Medical and Bioengineering. 2015;4(3):239–243. doi:10.12720/jomb
49. Popov AL, Popova NR, Selezneva II, Akkizov AY, Ivanov VK. Cerium oxide nanoparticles stimulate proliferation of primary mouse embryonic fibroblasts in vitro. Mater Sci Eng C. 2016;68:406–413. doi:10.1016/j.msec.2016.05.103
50. Mittal S, Pandey AK. Cerium oxide nanoparticles induced toxicity in human lung cells: role of ROS mediated DNA damage and apoptosis. Biomed Res Int. 2014;2014:891934. doi:10.1155/2014/891934
51. Li Y, Li P, Yu H, Bian Y. Recent advances (2010–2015) in studies of cerium oxide nanoparticles’ health effects. Environ Toxicol Pharmacol. 2016;44:25–29. doi:10.1016/j.etap.2016.04.011
52. Qin F, Shen T, Li J, et al. SF-1 mediates reproductive toxicity induced by cerium oxide nanoparticles in male mice. J Nanobiotechnology. 2019;17(1):41. doi:10.1186/s12951-019-0474-2
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.