Back to Journals » Clinical, Cosmetic and Investigational Dentistry » Volume 15
CBCT Analysis of Edentulous Mandibular Symphysis in Iraqi Patients for Treatment with Implant-Supported Overdentures. Cross-Sectional Single-Center Study
Authors Hassan NA, Al-Jaboori ASK , Al-Radha ASD , Ali MQ, Albayati RM
Received 14 March 2023
Accepted for publication 29 April 2023
Published 3 May 2023 Volume 2023:15 Pages 79—87
DOI https://doi.org/10.2147/CCIDE.S410620
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Christopher E. Okunseri
Nuhad A Hassan,1 Aseel S Khazaal Al-Jaboori,2 Afya Sahib Diab Al-Radha,3 Maisaa Q Ali,4 Raya M Albayati4
1Department of Oral Medicine, College of Dentistry, Mustansiriyah University, Baghdad, Iraq; 2Department of Prosthetic Dentistry, College of Dentistry, Mustansiriyah University, Baghdad, Iraq; 3Oral Surgery and Periodontology Department, College of Dentistry, Mustansiriyah University, Baghdad, Iraq; 4Department of Dental Radiology, Al-Falah Center, Ministry of Health, Baghdad, Iraq
Correspondence: Afya Sahib Diab Al-Radha, Oral Surgery and Periodontology Department; College of Dentistry, Al- Mustansiriyah University, Central Baghdad Post Office, Post Box (P.o.) 55418, Baghdad, Iraq, Tel +964(0)7816883387, Fax +964(0)0115372237, Email [email protected]; [email protected]
Background: Overdentures supported by dental implants are a useful treatment strategy for patients with edentulous mandibles. The aim of this study was to evaluate certain characteristics of the mandibular symphyseal edentulous cases for dental implant treatment using CBCT; and if it is associated with gender differences.
Methods: Ninety patients (45 females and 45 males) were included in this investigation. A section along the midline of each CBCT image was chosen for the edentulous symphyseal area. Symphysis height, width, and cortical thickness was measured for each patient. Bone density were calculated at four points A two-sample Student’s t-test and Pearson correlation were used for statistical analysis.
Results: Males had a considerably greater symphysis height (26.66± 6.21 mm) than females (24.07± 5.00 mm) (p = 0.02). Similar results were found for bone width; males had greater means than females. In case of cortical bone thickness; males had thicker bone cortex in the buccal region than females (p = 0.01). While the difference in bone density between genders was small. However, a positive relationship was recorded between symphysis height and width.
Conclusion: Height and width of the mandibular symphysis were influenced by gender in edentulous patients, with men showing higher measurements. The reduction of symphyseal height decreased concurrently with the width after teeth were lost, while bone density was maintained in both genders with no difference between them. The lingual cortical bone is significantly thicker than the buccal cortex at the lower part of the symphysis.
Keywords: mandibular symphysis, CBCT, dental implant, cortical thickness
Introduction
Complete dentures were the standard of care for edentulous patients for more than a century. They allowed patients to speak, eat, and interact with the rest of society without interrupting their normal lifestyle. However, many people struggle to use complete dentures due to denture discomfort and mobility, particularly in the case of mandibular dentures. Therefore, during the past decades, clinical investigations have been conducted to determine the advantages of using dental implants to support lower dentures.1,2 These investigations showed that titanium dental implants have a very good survival rate with few clinical impediments, particularly in the anterior mandible, and that implant placement greatly slows down the rate of bone loss in the edentulous jaws.
In the York Consensus Statement of 2009, it was decided that the restoration of the edentulous mandible with an overdenture supported by two inter-foraminal implants should be offered to patients as a first option of treatment other than conventional dentures.3 The prosthesis that is seated on implants offers many benefits, including improved balance, enhanced functional efficiency, safer chewing, reduced ridge resorption, better aesthetics, the elimination of prosthetic flaws, and most importantly, the removal of the anxiety associated with speech or mastication detachment.4 The symphysis of the mandible undergoes numerous modifications after tooth extraction, especially in terms of volume and shape. As about up to 50% of the initial volume may be lost within the first six months after tooth extractions.5,6 The patient appearance affected by the buccal mandibular cortex; the bone cortex is more susceptible to resorption and bone remodeling after any tooth extraction, particularly on the vestibular side of the jaw.7 Therefore, the analysis of alveolar bone is crucial when deciding on implant location, dimensions, and implantation angle.8,9
Implant surface topography and surface energy has been documented in the literature to enhance cell proliferation and accelerate osseointegration process.10,11 Adding to that, the features of the bone and the surgical technique have a considerable impact on the success of dental implants.9 Even though the mandibular symphysis is thought to be a fairly predictable and safe place for dental surgery, the number of surgical mistakes in this area has increased,1 making it important to measure bone sizes correctly.
Cone-beam CT (CBCT) is a relatively new radiological imaging technique that allows for accurate presentation of the anatomical structures and identification of pathological lesions. CBCT is widely used in dentistry because it has a high resolution and a low radiation risk (compared to medical CT).12
The thickness of the jaw and the position of the anatomical landmarks following bone resorption have been examined in a few Iraqi studies.13–15 Nevertheless, the characteristics of the mandibular symphyseal area in completely edentulous patients have not been extensively examined in the Iraqi population. The goal of this CBCT study was to evaluate certain characteristics of the edentulous mandibular symphyseal area (MS), including bone density, cortical thickness, and bone dimensions. Furthermore, evaluation if the characteristics of the edentulous mandibular symphyseal area had gender differences.
Materials and Methods
Study Design
This was an observational cross-sectional single-center study in which the median symphyseal region dimensions and shape were studied in completely edentulous patients using CBCT. The cortical bone thickness and symphyseal width, height, and density were measured and compared between males and females. The relationship of symphysis height with width and density was evaluated. Classification and distribution of the ridge type between males and females were also estimated based on Cawood and Howell’s categorization.
Data Collection
Data was collected from ninety patients during pre-implantation routine CBCT investigations in an Al-Sadar City Specialized Center for Dentistry from first of February 2021 to thirty of March 2022. The ethics were obtained by “The Scientific Committee of the Oral Medicine Department, College of Dentistry, Mustansiriyah University” (PROTOCOL 14-2-2020). The research was conducted in accordance with the “World Medical Association and the Declaration of Helsinki”. After explaining the purpose and the objectives of the study, “informed consent was obtained from the study participants before enrolling in the study.
Study Size
Using the G*Power 3.1.9.7 program and information from our pilot study, the minimal sample size was calculated to be 42 patients in each group.
Participants
Inclusion criteria: were participants with a completely edentulous lower arch for more than 3 months with no history of bone disease. Exclusion criteria: were patients with a previous history of trauma, jaw surgery, postoperative complications, skeletal anomalies, or muscle-skeletal disorders were disqualified.
CBCT Setting
The images were taken by “Kodak 9500, Care Stream, France; 10.8 s exposure time; 10 mA; 90 KV; voxel size of 300 µm; FOV 8×10 cm”.
Data Sources (Measurements)
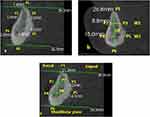
A section along the midline of each CBCT image was chosen for the edentulous symphyseal area. The section was equally distant from each mental foramen. Every patient was given thirteen estimates, which included symphysis height, symphysis width, bone density, and cortex thickness. The mandibular symphyseal cross-section was used to illustrate all measurements (Figure 1).
Cortical Thickness
The cortical thickness was estimated (6 measurements in mm) both buccally and lingually at six reference points: P1: the ridge peak, P2: the upper buccal, P3: the upper lingual, P4: the lower buccal, P5: the lower lingual, P6: the base of the mandible.
Symphysis Height
During the section height measurement, a segment was taken that passed through the symphysis maximum height (HL).
Symphysis Width
Widths of both the upper (W1) and lower (W2) thirds of the mandibular ridge. “Parallel to the mandibular plane” and running through the section, two lines were drawn at the upper-third (W1:P2-P3) and lower-third (W2:P4-P5) of the total height. Sections from the base of the mandible to the alveolar ridge were inclined lingually, except those through the symphysis.
Bone Density
In Hounsfield Units (HU), four measures of bone density (BD) were calculated (P1 and P6 at cortical bone, P7 and P8 at cancellous bone)16 (Figure 1).
Cawood and Howell’s Symphysis Categorization
Classification of mandible ridge type in symphysis was recorded according to gender using the CBCT.
Statistical Analysis
The readings were collected and repeated after one week by one radiologist. Although the two readings were nearly identical, a paired t-test for 10 randomly selected CBCT scans was performed to evaluate the intra-observer reliability and revealed no significant difference. The statistics used the mean of each pair of measurements.
Statistically, SPSS software version 21 was used for data analysis. The homogeneity of variance and normality of the data were evaluated by Levene’s and Shapiro–Wilk tests, respectively. An independent t-test was used to test the difference between genders in the symphysis parameters. Pearson coefficient correlation was used to show the relationship between symphysis height and both width and density. A p-value of ≤ 0.05 was used to define statistical significance.
Results
Ninety patients were enrolled in current study, and the average age of the patients was 53 years, 45 females (mean age: 47±6.80); and 45 males (mean age: 59±8.46).
Males had statistically higher buccal cortical bone thickness than females in P2 and P4 points with p=0.01. as can be seen in Table 1. Symphysis height was significantly higher in males than in females p=0.02. Similar results can be seen for bone width, as the males’ bone width were statistically greater than the females’ bone width in both W1 and W2 points. Regarding the comparison between the cortical thickness for all subjects, a thicker cortex was found at the base of the mandible (P6) than at the crestal point (P1), and the difference was significant (p< 0.001). Furthermore, at the lower part of the symphysis the lingual cortex (P5) was thicker than the buccal cortex (P4) (p= 0.03), as can be seen in Table 2.
 |
Table 1 Comparison of Bone Dimensions and Density of the Mandible Symphysis Between Males (n=45) and Females (n=45) |
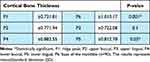 |
Table 2 Comparison of Cortical Thickness for All Subjects (Males and Females) |
A positive correlation was found between the height and width of the symphysis. However, the height was more strongly related to the lower part of the mandible W2 (r = 0.6, P = 0.0001) than to the upper part of W1 (r = 0.3, P = 0.004). Bone density showed no correlation to height except for the lower point of density (P8), which was positively correlated with height (r = 0.3; p = 0.014) Table 3.
 |
Table 3 Correlation Between Bone Height (HL) and Both of Bone Density and Width at the Symphyseal Region (n=90) |
Based on Cawood and Howell’s symphysis categorization, type III was more frequent in both genders followed by class IV; class V and class VI respectively in both genders. However, still the male had a slightly higher percentage at all classes except for class VI. No patients in classes I and II were included in our study cohort based on the inclusion criteria. Cawood and Howell’s classification distribution was not statistically different between males and females (p = 0.25, Fisher’s exact test) (Figure 2, Table 4).
 |
Table 4 The Gender Distribution of Cawood and Howell Classifications of the Symphysis |
 |
Figure 2 According to gender, the distribution of Cawood and Howell classes (45 males and 45 females). Due to the criteria of our study, no patients from classes I or II were allowed to take part. |
Discussion
The present study was conducted to determine the certain characteristics of the edentulous mandibular symphyseal area and if it had gender differences. Males had significantly higher symphysis height and width than females. With a positive relationship between symphysis height and width.
Imaging techniques are routinely used for implant-based treatment choices. They serve as pre-clinical examinations in implantology, providing the data necessary to create the best possible treatment strategy. Kanai et al17 used CBCT and a lateral cephalogram to examine the symphysis thickness of 100 healthy adults aged 15 to 56 years. The lateral cephalogram revealed a larger symphysis thickness at measurement locations than the CBCT, and the differences were statistically significant. They found that CBCT is a better way to measure the shape of MS than a lateral cephalogram.
The present CBCT study recorded a significant gender difference in both height and width of the mandible symphysis, where male patients showed higher values. This outcome is in line with Mense et al,16 who studied 103 CBCTs to compare the heights of males and females in the symphyseal region. They found that the heights were significantly different by gender. This finding is also similar to that of the Abdul-Qadir study, which examined lower alveolar height and symphysis widths using cephalometric radiographs in 60 adults.18 Furthermore, during the study of 125 CBCT images (66 females and 59 males), Uysal et al (2012) discovered that males were larger than females in mandibular symphyseal height and width measurements.19 The outcome was also compatible with the literature, which indicates that the males’ symphyseal measurements were greater as compared with females.20–22
A previous study showed that the males had greater MS height.23 Furthermore, a smaller symphysis height recorded for females in the older age group.24 Moreover, Linjawi et al25 concluded that the symphysis morphology varies by gender and confirmed that the linear dimensions and surface area of the symphysis appeared larger in males. Similar results found in a previous CBCT study,26 males showed significantly higher crestal width than females in the edentulous mandible.
The width of the existing bone is one of the most important clinical factors that affects the survival rate of endosteal implants. A 4 mm dental implant often requires more than 6 mm of bone thickness to ensure an adequate blood supply around the implant.1 Following implant loading, the first crestal bone loss is inversely correlated with the bone’s initial breadth. The least mean value of symphysis width recorded in the current investigation was that of the females (W1: 8.61 mm) which makes the symphysis a suitable site for successful dental implants.
In the current investigation, the buccal cortical bone thickness of the symphysis was significantly higher in the male patients than the females and showed no difference at the ridge crest, base, or lingual side. In a study conducted by Fayed et al27 among young patients (13–27 years old) to compare the buccal and palatal cortical bone thickness of the dentate mandible in females and males using CBCT images, they found no significant difference between the sexes, suggesting that variation in bone thickness that happens at a later stage of life between the sexes is determined by factors such as the bone’s clinical circumstances and remodeling associated with teeth loss. Some authors concluded that the cortical bone thickness varies depending on age.27,28 Furthermore, it has been reported previously that the loss of teeth with aging significantly alters the form of the mandible in both the alveolar and basal regions due to changes in muscle activity and a lack of occlusion.6
When comparing the cortical bone thickness for all subjects in the present work, the lingual cortex of the lower part of the symphysis was thicker than the buccal cortex. Moreover, the base of MS was significantly thicker than the crest. An older report16 found that the lingual and buccal cortical thickness varied significantly, which is in harmony with our results. This suggests that analyzing the symphyseal area before the implant can help determine a good design. Another study was done previously found that the lingual side was thicker than the buccal one; therefore, the level of the mid-root needs more attention during the implantation process.29
This discrepancy can be viewed as one of the reasons for the lingual sides’ slow rate of resorption following tooth extraction. Grunder30 highlighted previously, that for the best implant placement and long-term success, the buccal plate should be at least 2 mm thick. This is because a thick plate will prevent soft and hard tissue recession and reduce future problems with aesthetics and bone loss. If the buccal plate is not thick enough, the surgeon should conduct local bone augmentation.
Al-Ekrish et al31 created a five-class version of an old classification based on CBCT data to measure the cortical/cancellous bone ratio. In this research, CBCT pictures were utilized to quantify bone density (BD). It was between 863.44±231.57 HU and 1269.62 HU in the males and between 865.31 HU and 1263.07±309.44 HU in the females, with no significant differences between the sexes. This BD result was verified by Mense et al16 and Park et al,32 who reported the cortical density range of the mandible in the alveolar bone as being around 700 to 1500 HU. Chae et al33 in 2020, also revealed close values when they examined the BD of mandibular symphysis among young people (10–20 years old) of both gender. This suggests that bone density is preserved with age and after the loss of teeth. Chae et al also indicated that females had a higher mean cortical BD than males, in contrast to the current study. Alnoori et al (2018) in a 3 years retrospective study indicated that the density of bone did not influence the primary and secondary stability of dental implants.34
In the remodeling process of bone, cytokines, and estrogen play a crucial role in regulating osteoclast and osteoblast activity. Thus, females will likely exhibit a higher amount of BD than males since estradiol aids in maintaining higher levels of BD. However, Lerner35 in a previous study, showed that osteoporotic women who received estrogen replacement therapy had denser bones than those who did not.35 In the current work, the females’ mean age was (47±6.80 years), and they may have begun menopause, increasing their exposure to osteoporosis risk factors related to low estrogen levels.
This evaluation also looked at the distribution of “Cawood and Howell ridge classifications” according to gender, and it was not different statistically between men and women. Also, there were no patients in Cawood-Howell classes I and II. Most of the patients were in classes III and IV. In contrast to this study, older research indicated that ridge type differed significantly by gender.16 Nevertheless, it showed the same prevalence of classes. This controversy might be related to the variances in sample size and ethnicity.
The height and width of the symphysis were found to be positively correlated in this study, which is in agreement with Abdul- Qadir’s study18 and in disagreement with Mense et al finding.16 In previous observations, the mandibular height showed the most pronounced atrophy-induced alterations. It lost about 1/3 of its initial bone height at the initial stage of the bone resorption process and about 1/5 of its initial bone mass at next stages. The study also found that the amount of alveolar bone resorption was related to the duration of time following extractions.
The initial clinical conditions and the healing of the sockets have a critical impact on the amount of bone loss in MS. In a systematic review, it was noted that the mean alveolar bone alterations following extraction caused a clinical width loss of 3.87 mm, which was larger than the height loss (1.53 mm).36 Similar outcomes were seen by Tan et al,37 with the horizontal loss being larger than the vertical loss of the bone during the first six months following extraction. Individual symphysis morphology is different due to multiple etiological factors such as facial type, genetics, and ethnicity.21,29 These limitations of this study warrant further research.
Conclusions
In edentulous patients, gender had an impact on the height and width of the mandibular symphysis, with men having higher values; with a positive correlation was found between the height and width of the symphysis. The base of the mandible had a significantly thicker cortical bone than at the crestal. Also, the lingual cortical bone is significantly thicker than the buccal cortex at the lower part of the symphysis.
After teeth extraction, the height and width of the symphyseal decreased in their dimensions, but the bone density was maintained for both men and women with no significant difference between them.
Data Sharing Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Acknowledgment
The Authors would like to thank Mustansiriyah University, Baghdad, Iraq, for its support in the present work.
Disclosure
The authors declared that there are no conflicts of interest in this work.
References
1. Guljé F, Raghoebar GM, Ter Meulen JW, Vissink A, Meijer HJ. Mandibular overdentures supported by 6-mm dental implants: a 1-year prospective cohort study. Clin Implant Dent Relat Res. 2012;14(Suppl 1):59–66. doi:10.1111/j.1708-8208.2011.00358.x
2. Thomason JM, Lund JP, Chehade A, Feine JS. Patient satisfaction with mandibular implant overdentures and conventional dentures 6 months after delivery. Int J Prosthodont. 2003;16(5):467–473.
3. Thomason JM, Feine J, Exley C, et al. Mandibular two implant-supported overdentures as the first choice standard of care for edentulous patients-the York Consensus Statement. Br Dent J. 2009;207(4):185–186. doi:10.1038/sj.bdj.2009.728
4. Imre MM, Marin M, Preoteasa E, Tancu A, Preoteasa C. Two implant overdenture–The first alternative treatment for patients with complete edentulous mandible. J Med Life. 2011;4(2):207.
5. Tan WL, Wong TL, Wong MC, Lang NP. A systematic review of post‐extractional alveolar hard and soft tissue dimensional changes in humans. Clin Oral Implants Res. 2012;23(Suppl 5):1–21. doi:10.1111/j.1600-0501.2011.02375.x
6. Merrot O, Vacher C, Merrot S, Godlewski G, Frigard B, Goudot P. Changes in the edentate mandible in the elderly. Surg Radiol Anat. 2005;27(4):265–270. doi:10.1007/s00276-005-0323-x
7. Rossell J, Puigdollers A, Girabent-Farrés M. A simple method for measuring thickness of gingiva and labial bone of mandibular incisors. Quintessence Int. 2015;46(3):265–271. doi:10.3290/j.qi.a32919
8. de Oliveira RC, Leles CR, Normanha LM, Lindh C, Ribeiro-Rotta RF. Assessments of trabecular bone density at implant sites on CT images. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;105(2):231–238. doi:10.1016/j.tripleo.2007.08.007
9. Rozé J, Babu S, Saffarzadeh A, Gayet-Delacroix M, Hoornaert A, Layrolle P. Correlating implant stability to bone structure. Clin Oral Implants Res. 2009;20(10):1140–1145. doi:10.1111/j.1600-0501.2009.01745.x
10. Ibrahim SW, Rafeeq AK, Ahmedhamdi MS. Histomorphometric assessment of implant coated with mixture of nano-alumina and fluorapatite in rabbits. Saudi Dent J. 2021;33(8):1142–1148. doi:10.1016/j.sdentj.2021.02.005
11. Smeets R, Stadlinger B, Schwarz F, et al. Impact of Dental Implant Surface Modifications on Osseointegration. Biomed Res Int. 2016;2016:6285620. doi:10.1155/2016/6285620
12. Benavides E, Rios HF, Ganz SD, et al. Use of cone beam computed tomography in implant dentistry: the international congress of oral implantologists consensus report. Implant Dent. 2012;21(2):78–86. doi:10.1097/ID.0b013e31824885b5
13. Al-Ghurabi ZH, Mohammed HB, Hassan TAL. Presurgical Assessment of Anatomical Landmark Position Using Two Views of Cone Beam Computed Tomography: a Comparative Radiological Study. J Craniofac Surg. 2022;33(2):165–168. doi:10.1097/SCS.0000000000008182
14. Hassan NA, Al-Jaboori ASK, Al-Radha ASD. Evaluation of Cortical Bone Thickness of Posterior Implant Sites Using CBCT in Iraqi Population. Int J Dent. 2022;2022:5723397. doi:10.1155/2022/5723397
15. Al-Saedi A, Al-Bakhakh B, Al-Taee RG. Using Cone-Beam Computed Tomography to Determine the Prevalence of the Second Mesiobuccal Canal in Maxillary First Molar Teeth in a Sample of an Iraqi Population. Clin Cosmet Investig Dent. 2020;12:505–514. doi:10.2147/CCIDE.S281159
16. Mense C, Saliba-Serre B, Ferrandez AM, Hüe O, Ruquet M, Lalys L. Cone beam computed tomography analysis of the edentulous mandibular symphysis. J Dent Sci. 2021;16(1):115–122. doi:10.1016/j.jds.2020.06.025
17. Kanai M, Yamaguchi T, Maki K. Cone-beam Computed Tomography evaluation of mandibular symphysis. Dental Med Res. 2011;31(1):16–23. doi:10.7881/dentalmedres.31.16
18. Abdul-Qadir MY. Assessment of anterior alveolar bone dimensions in adolescents and adults with class I normal occlusion. Al–Rafidain Dent J. 2014;14(2):161–172. doi:10.33899/rden.2014.160887
19. Uysal T, Yagci A, Ozer T, Veli I, Ozturk A. Mandibular anterior bony support and incisor crowding: is there a relationship? Am J Orthod Dentofacial Orthop. 2012;142(5):645–653. doi:10.1016/j.ajodo.2012.05.017
20. Marghalani HYA, Guan G, Hyun P, Tabbaa S, Linjawi AI, Al-Jewair T. Relationship between mandibular symphysis dimensions and skeletal pattern in adults. Folia Morphol. 2022;81(2):464–471. doi:10.5603/FM.a2021.0045
21. Al-Khateeb SN, Al Maaitah EF, Abu Alhaija ES, Badran SA. Mandibular symphysis morphology and dimensions in different anteroposterior jaw relationships. Angle Orthod. 2014;84(2):304–309. doi:10.2319/030513-185.1
22. Gómez Y, García-Sanz V, Zamora N, et al. Associations between mandibular symphysis form and craniofacial structures. Oral Radiol. 2018;34(2):161–171. doi:10.1007/s11282-017-0292-x
23. Arruda KEM, Valladares Neto J, Almeida GA. Assessment of the mandibular symphysis of Caucasian Brazilian adults with well-balanced faces and normal occlusion: the influence of gender and facial type. Dental Press J Orthod. 2012;17(3):40–50. doi:10.1590/S2176-94512012000300012
24. Ghalib ZA, Agha NF. Evaluation of mandibular symphysis area among Iraqi adolescent. Int J Enhanced Res Sci Tech Eng. 2016;5(10):52–57.
25. Linjawi AI, Afify AR, Baeshen HA, Birkhed D, Zawawi KH. Mandibular symphysis dimensions in different sagittal and vertical skeletal relationships. Saudi J Biol Sci. 2021;28(1):280–285. doi:10.1016/j.sjbs.2020.09.062
26. Koç A, Erdem R. Evaluating posterior edentulous mandible alveolar crest dimensions by cone beam computed tomography. Eastern J Med. 2020;25(3):345–349. doi:10.5505/ejm.2020.24471
27. Fayed MM, Pazera P, Katsaros C. Optimal sites for orthodontic mini-implant placement assessed by cone beam computed tomography. Angle Orthod. 2010;80(5):939–951. doi:10.2319/121009-709.1
28. Swasty D, Lee JS, Huang JC, et al. Anthropometric analysis of the human mandibular cortical bone as assessed by cone-beam computed tomography. J Oral Maxillofac Surg. 2009;67:491–500. doi:10.1016/j.joms.2008.06.089
29. Srebrzyńska-Witek A, Koszowski R, Różyło-Kalinowska I. Relationship between anterior mandibular bone thickness and the angulation of incisors and canines-a CBCT study. Clin Oral Investig. 2018;22(3):1567–1578. doi:10.1007/s00784-017-2255-3
30. Grunder U. Crestal ridge width changes when placing implants at the time of tooth extraction with and without soft tissue augmentation after a healing period of 6 months: report of 24 consecutive cases. Int J Periodontics& Restorative Dent. 2011;31(1):9–17.
31. Al-Ekrish AA, Widmann G, Alfadda SA. Revised, computed tomography-based Lekholm and Zarb jawbone quality classification. Int J Prosthodont. 2018;31(4):342–345. doi:10.11607/ijp.5714
32. Park HS, Lee YJ, Jeong SH, Kwon TG. Density of the alveolar and basal bones of the maxilla and the mandible. Am J Orthod Dentofacial Orthop. 2008;133(1):30–37. doi:10.1016/j.ajodo.2006.01.044
33. Chae JM, Gousman J, Seo JW, Bay RC, Jue SS, Park JH. A retrospective CBCT study of the relationship between mandibular symphysis bone density and mandibular growth direction. J Clin Pediatr Dent. 2020;44(5):356–365. doi:10.17796/1053-4625-44.5.11
34. Alnoori DNM, Al-Adili DSS, Alnuaimi DAS. Evaluation of related factors affecting stability and survival rate of dental implants (meta-analysis of retrospective study). MDJ. 2018;15(1):105–121.
35. Lerner UH. Bone remodeling in post-menopausal osteoporosis. J Dent Res. 2006;85(7):584–595. doi:10.1177/154405910608500703
36. Van der Weijden F, Dell’Acqua F, Slot DE. Alveolar bone dimensional changes of post-extraction sockets in humans: a systematic review. J Clin Periodontol. 2009;36(12):1048–1058. doi:10.1111/j.1600-051X.2009.01482.x
37. Tan WL, Wong TL, Wong MC, Lang NP. A systematic review of post-extractional alveolar hard and soft tissue dimensional changes in humans. Clin Oral Implants Res. 2012;23(Suppl 5):1–21.
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.