Back to Journals » OncoTargets and Therapy » Volume 14
Case Report: SMARCB1 (INI-1)-Deficient Carcinoma of the Nasal Cavity with Pure Yolk Sac Tumor Differentiation and Elevated Serum AFP Levels
Authors Li CY, Han Y, Xu K, Wu S, Lin XY , Cao H
Received 18 January 2021
Accepted for publication 11 March 2021
Published 29 March 2021 Volume 2021:14 Pages 2227—2233
DOI https://doi.org/10.2147/OTT.S302613
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Prof. Dr. Nicola Silvestris
Chun-Yu Li, Ya-mei Han, Ke Xu, Shu-yue Wu, Xu-Yong Lin, Hong-yi Cao
Department of Pathology, The First Affiliated Hospital and College of Basic Medical Sciences, China Medical University, Shenyang, 110001, People’s Republic of China
Correspondence: Hong-yi Cao
Department of Pathology, The First Affiliated Hospital and College of Basic Medical Sciences, China Medical University, Shenyang, 110001, People’s Republic of China
Email [email protected]
Abstract: In adults, yolk sac tumors (YSTs) in the nasal cavity and paranasal sinuses are very rare. To date, only six cases have been reported in the English literature. YSTs in adults are often accompanied by cancer, teratocarcinosarcoma, and other malignant components. Here, we have reported a case of nasal tumor in a 55-year-old man with nasal obstruction and epistaxis. Morphologically, the tumor showed histological characteristics of pure YST. Immunohistochemical staining showed diffuse expression of SALL4, CDX2, and GPC-3 accompanied by sporadic expression of alpha-fetoprotein (AFP) and CD117. After 20 and 40 days of operation, the serum AFP level was 220.30 and 43.60 ng/mL (normal, < 7 ng/mL), respectively, which supported the pathological diagnosis of YST. However, we further performed immunohistochemical staining and fluorescence in situ hybridization using an INI-1 probe to detect the status of INI-1 in tumor cells. The results revealed that INI-1 was absent in tumor cells. Hence, we corrected the diagnosis to SMARCB1 (INI-1)-deficient carcinoma of the nasal cavity with YST differentiation. The patient underwent surgery and adjuvant radiotherapy in our hospital without evidence of recurrence or metastasis at the 6-month follow-up. The serum AFP level had also normalized. In conclusion, our case demonstrates that INI-1-deficient carcinoma may exhibit, a pure YST differentiation and immunophenotype, and elevated serum AFP levels. In adults, YST in the nasal cavity may represent INI-1-deficient carcinoma, which may be a potential diagnostic pitfall.
Keywords: yolk sac tumor, SMARCB1 (INI-1)-deficient carcinoma, sinonasal carcinoma, gonadal germ cell tumor, INI-1
Background
SMARCB1 (INI-1)-deficient carcinoma was recently described in a group of sinonasal carcinomas, and it behaves like an aggressive high-grade neoplasm.1,2 Histologically, it shows a broad spectrum of high-grade morphology, including a basaloid pattern with inverted papilloma-like growth, numerous rhabdoid or plasmacytoid cells, oxyphilic squamoid cell population, focal glandular differentiation, rare spindle cell sarcomatoid differentiation, and occasional pagetoid spread.2,3
Yolk sac tumor (YST) is a malignant tumor of germ cell origin. YSTs very rarely occur in extragonadal sites, especially in adults. To our knowledge, only six cases of adult YST in the sinonasal area have been reported in the English literature.4 Unlike YSTs in children with pure YST morphology, YSTs in adults are frequently accompanied by cancer, teratocarcinosarcoma, and other components.4–7
Here, we have reported a case of a neoplasm with a morphology and immunophenotype of pure YST. However, SMARCB1 (INI-1) immunohistochemical staining and fluorescence in situ hybridization (FISH) revealed SMARCB1 (INI-1)-deficient carcinoma. This indicates that YSTs in adults may be at least partially derived from somatic epithelial neoplasms. This finding expands the spectrum of SMARCB1 (INI-1)-deficient carcinoma and provides new evidence for differential diagnosis.
Case Presentation
A 55-year-old man presented to our hospital with persistent right nasal obstruction and recent aggravation accompanied by right nasal hemorrhage. Magnetic resonance imaging (MRI) of the paranasal sinuses showed long T1 and long T2 soft tissue signal shadows in the right nasal cavity, which were significantly and nonuniformly enhanced on contrast-enhanced imaging. The lesions invaded the sphenoid sinus and the right ethmoid sinus upward, invading the posterior nostril, the right orbital lateral wall, and the right maxillary sinus (Figure 1). Laboratory examination findings revealed the following: carcinoembryonic antigen (CEA) level: 1.37 ng/mL (normal, <4 ng/mL), NSE level: 25.18 ng/mL (normal, <16 ng/mL), and CYfra21-1 (CK19 fragment) level: 8.73 ng/mL (normal, <3.3 ng/mL). The patient then underwent lumpectomy in our hospital.
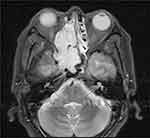 |
Figure 1 MRI presentation of the tumor. MRI revealed a solid mass involved the nasal cavity and sinuses, destroying bone tissue. |
The resected specimens were fixed with 10% neutral-buffered formalin and embedded in paraffin blocks. Tissue blocks were cut into 4-μm slides, deparaffinized in xylene, rehydrated with graded alcohols, and stained with hematoxylin and eosin or immunostained with the following antibodies (ZSGB-BIO, China) using a streptavidin-peroxidase system (KIT-9720, Ultrasensitive TM S-P, MaiXin, China): cytokeratin (CK), SALL4, glypican 3, CDX2, P53, Ki-67, epithelial membrane antigen (EMA), P16, vimentin, CD117, CK20, alfa-fetoprotein (AFP), CEA, CK5/6, glial fibrillary acidic protein, P63, S-100, CK7, smooth muscle actin, OCT3/4, CD10, carbonic anhydrase IX, Pax-8, HepPar-1, and synaptophysin. The chromogen used was a diaminobenzidine tetrahydrochloride substrate (DAB kit, MaiXin, China). Appropriate positive and negative controls were used to exclude false positivity and negativity, respectively.
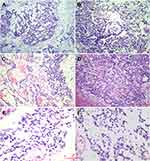
Grossly, the resected tissue was cut into sections measuring approximately 3 × 3 cm. The cut sections were gray-red and grayish-brown in color. Histologically, the cells were predominantly arranged in a microcystic, reticular, or papillary pattern with a myxoid background. The cells were distributed around the blood vessels, forming a Schiller-Duval (S-D) body. In addition, eosinophilic basement membrane materials or numerous round hyaline globules were frequently seen inside and outside the microcysts. The lining cells demonstrated cuboidal or polygonal shapes, a lightly stained cytoplasm, and mild-to-moderate atypia (Figure 2).
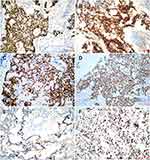
Immunohistochemically, the tumor cells showed diffuse expression of CK, GPC-3, CDX-2, and SALL4 and focal or partial expression of AFP, CEA, P16, EMA, vimentin, CK20, and CD117. The expression of other antibodies was uniformly negative. The Ki-67 proliferative index was approximately 60% (Figure 3).
According to the morphologic pattern and immunohistochemical staining, our preliminary pathological diagnosis was a malignant tumor, consistent with YST. Therefore, we performed further examinations to exclude metastasis and evaluate serum AFP levels. After 20 and 40 days of operation, the serum AFP level was 220.30 and 43.60 ng/mL (normal, <7 ng/mL), respectively. The patient denied having any other disease.
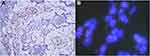
Additional immunohistochemistry revealed absence of INI-1 expression (Figure 4A). FISH using a SMACRB1 missing probe (LBP, China) showed that 1R2G accounted for 3% and 2G accounted for 75% of all cells, indicating the deficiency of SMACRB1 (Figure 4B).
Finally, we revised the diagnosis to SMARCB1 (INI-1)-deficient carcinoma with pure YST differentiation.
The patient is currently receiving adjuvant radiotherapy at our hospital. There was evidence of recurrence or metastasis at the 6-month follow-up. The serum AFP level also normalized.
Discussion
As the core subunit of the chromatin remodeling complex SWI/SNF, SMARCB1 (INI-1) causes abnormal inactivation in various tumors, including rhabdoid tumor, epithelioid sarcoma, renal medullary carcinoma, epithelioid schwannoma, epithelioid malignant schwannoma, myoepithelial carcinoma, and extraosseous myxoid chondrosarcoma.8
INI-1-deficient carcinoma of the nasal and paranasal sinuses is a relatively rare neoplasm that was briefly described in the 2017 World Health Organization classification for head and neck tumors as an undifferentiated carcinoma.9 This neoplasm usually occurs in adults. Its clinical manifestations include nasal congestion, epistaxis, and headache. Imaging findings show a mass invading the nasal cavity or paranasal sinuses. Histologically, the tumor has a relatively broad morphological spectrum, with nests of undifferentiated basaloid cells and fibroblasts in the stroma. They are characterized by scattered rhabdoid cells, but these cells are not always seen in all tumors. Rare patterns, including glandular, papillary, or sarcomatoid patterns, with mucous distribution in the stroma, have been reported.1–3,10,11
Immunohistochemically, the tumor shows consistent CK expression, INI-1 deletion, and varying degrees of CK5, P63, CK7, P16, CD117, and synaptophysin, chromogranin A expression. FISH shows EBV and HPV negativity. It should be noted that FISH results are not completely consistent with the histochemical examination findings. Most cases show homozygous or heterozygous deletions of SMARCB1, but in a few cases, FISH cannot detect the deletions.2
YST is a highly malignant germ cell tumor. To our knowledge, adult YST in the nasal area is very rare, and only six cases have been reported in the English literature. YSTs of the nasal cavity and paranasal sinuses in children are mostly pure YSTs, while those in adults are mostly accompanied by cancer, such as teratocarcinosarcoma or choriocarcinoma,4–7,12,13 suggesting that YSTs of the nasal cavity and paranasal sinuses in adults may originate from somatic cells.
The present case occurred in an adult. Microscopically, the tumor had a characteristic YST-like morphology. The cells were arranged into multiple irregular nests, with microcystic, tubular, or pseudopapillary patterns. Locally, the cells were seen around the blood vessels, forming an S-D corpuscular structure. Diffuse expression of CK, SALL4, CDX2, and GPC-3 was accompanied by sporadic expression of AFP and CD117 on immunohistochemical analysis. Further, the serum AFP level was higher than normal value. These findings supported the pathological diagnosis of YST.
However, we further reviewed the literature and found that Akeesha et al and Michal Zamecnik et al have reported occasional yolk sac-like differentiation (or characteristics) in INI-1-deficient nasal and paranasal carcinomas, also expressing markers such as SALL4, CDX2, and GPC-3.14,15 It should be noted that these markers are also occasionally expressed in non-yolk sac-like regions. Therefore, to further confirm the diagnosis, we performed immunohistochemical analysis and found that the expression of INI-1 was missing in tumor cells; FISH also revealed the same result. Therefore, we diagnosed the patient with INI-1-deficient carcinoma with pure YST differentiation. In addition, we found that the patient had an abnormal serum AFP level. This finding has not been reported in the literature previously.
Further, we hypothesized that exogonadal YSTs require attention for possible somatic origin with yolk sac differentiation. However, further confirmation is needed regarding whether YSTs in the nasal cavity of adults are INI-1-deficient carcinomas showing YST differentiation. Among the six cases of YST of the nasal cavity and paranasal sinuses in adults, four cases were associated with transitional cell carcinoma or poorly differentiated carcinoma, one case was associated with teratocarcinosarcoma, and one case was associated with choriocarcinoma.4 Yasuo et al reported a case of rhabdoid tumor with yolk sac-like morphology and expression of SALL4 and glypican 3, indicating that other tumors with SMARCB1 deletion could also show yolk sac-like morphology or differentiation.16 Deficiency of SMARCA4, another important member of the SWI/SNF complex, has recently been reported in teratocarcinosarcoma.17 Therefore, we believe that teratoma carcinosarcoma with yolk sac differentiation may also be the result of the absence of SMARCA4. Thus, SWI/SNF complex member-deficient tumors may be characterized by YST differentiation.
In general, the differential diagnosis of INI-1-deficient carcinoma of the nasal cavity and paranasal sinuses mainly includes poorly differentiated, undifferentiated, and myoepithelial carcinoma. Since this case presented a cystic structure, we suggest that HPV-related multiphenotype carcinoma, salivary gland carcinoma, embryonal carcinoma, and other tumors with cystic or adenoid arrangement should be added for differential diagnosis. As listed in Table 1, the prognosis of adult nasal YSTs varied, may also depend on the accompanying components.4-7,12,13,18 The reported treatment included surgery, radiotherapy or chemotherapy. SMARCB1-deficient sinonasal carcinomas in general are thought to be aggressive neoplasms. Recommended treatment methods are surgery with additional radiotherapy or chemotherapy.2 It is not clear the difference in prognosis and clinical significance between SMARCB1-deficient sinonasal carcinoma with YST differentiation and non-deficient YST. And we should gather more information to test it.
 |
Table 1 Reported Cases of Adult Nasal YST or Tumors with YST Morphology |
Conclusion
In summary, we encountered an interesting case with diagnostic pitfalls. With the help of INI-1 immunohistochemistry and FISH, the actually diagnosis in this case of adult nasal YST was uncovered. This case highlights that carcinomas lacking INI-1 expression and those with abnormal serum AFP levels may exhibit YST differentiation. Further studies are needed to confirm whether YSTs in the nasal cavity of adults represent SWI/SNF complex-deficient tumors with YST differentiation.
Data Sharing Statement
Not applicable.
Ethical Approval and Informed Consent
Ethical approval is not required for this Case report. Written informed consent was obtained from the patient.
Consent for Publication
Written informed consent was obtained from the patient for publication of this paper and any accompanying images.
Acknowledgments
We sincerely thank Guangzhou LBP Medicine Science and Technology Co., Ltd. for generously providing us technical assistance on SMARCB1 FISH detection.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
The present study was supported by the Liaoning Provincial Natural Science Foundation (grant no. 20180530040) and Xinjiang Autonomous Region Natural Science Foundation (grant no. 2020D01A123).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bishop JA, Antonescu CR, Westra WH. SMARCB1 (INI-1)-deficient carcinomas of the sinonasal tract. Am J Surg Pathol. 2014;38(9):1282–1289. doi:10.1097/PAS.0000000000000285
2. Agaimy A, Hartmann A, Antonescu CR, et al. SMARCB1 (INI-1)-deficient sinonasal carcinoma: a series of 39 cases expanding the morphologic and clinicopathologic spectrum of a recently described entity. Am J Surg Pathol. 2017;41(4):458–471. doi:10.1097/PAS.0000000000000797
3. Tang SJ, Zhai CW, Yuan CC, Zhang JH, Wang SY. SMARCB1 (INI1)-deficient sinonasal carcinoma: a clinicopathological analysis of six cases. Zhonghua Bing Li Xue Za Zhi. 2020;49(1):47–51. doi:10.3760/cma.j.issn.0529-5807.2020.01.009
4. Chuang HC, Kang CJ, Lee LY. Sinonasal pure yolk sac tumor: a case report and literature review. Fetal Pediatr Pathol. 2014;33(3):127–134. doi:10.3109/15513815.2013.839013
5. Filho BC, McHugh JB, Carrau RL, Kassam AB. Yolk sac tumor in the nasal cavity. Am J Otolaryngol. 2008;29(4):250–254. doi:10.1016/j.amjoto.2007.09.001
6. Mei X, Xia Y, Sasano H, Gao H. Sinonasal yolk sac (Endodermal sinus) tumor in an adult female–a case report and review of the literature. APMIS. 2015;123(9):810–814. doi:10.1111/apm.12409
7. Mishra A, El-Naggar AK, DeMonte F, Hanna EY. Endodermal sinus tumor of the paranasal sinuses. Head Neck. 2008;30(4):539–543. doi:10.1002/hed.20711
8. Shaverdashvili K, Azimi‐Nekoo E, Cohen P, et al. INI‐1 (SMARCB1)–deficient undifferentiated sinonasal carcinoma: novel paradigm of molecular testing in the diagnosis and management of sinonasal malignancies. The Oncologist. 2020;25(9):738–744. doi:10.1634/theoncologist.2019-0830
9. Thompson LDR, Franchi A. New tumor entities in the 4th edition of the World Health Organization classification of head and neck tumors: nasal cavity, paranasal sinuses and skull base. Virchows Arch. 2018;472(3):315–330. doi:10.1007/s00428-017-2116-0
10. Gomez-Acevedo H, Patterson JD, Sardar S, et al. SMARC-B1 deficient sinonasal carcinoma metastasis to the brain with next generation sequencing data: a case report of perineural invasion progressing to leptomeningeal invasion. BMC Cancer. 2019;19(1):827. doi:10.1186/s12885-019-6043-0
11. Huang SC, Zhang L, Sung YS, et al. Secondary EWSR1 gene abnormalities in SMARCB1-deficient tumors with 22q11-12 regional deletions: potential pitfalls in interpreting EWSR1 FISH results. Genes Chromosomes Cancer. 2016;55(10):767–776. doi:10.1002/gcc.22376
12. Thomas J, Adegboyega P, Iloabachie K, Mooring JW, Lian T. Sinonasal teratocarcinosarcoma with yolk sac elements: a neoplasm of somatic or germ cell origin? Ann Diagn Pathol. 2011;15(2):135–139. doi:10.1016/j.anndiagpath.2010.01.004
13. Arumugam D, Thandavarayan P, Chidambaram L, Boj S, Marudasalam S. Primary nasopharngeal yolk sac tumor: a case report. J Clin Diagn Res. 2016;10(5):ED06–7. doi:10.7860/JCDR/2016/17620.7760
14. Shah AA, Jain D, Ababneh E, et al. SMARCB1 (INI-1)-deficient adenocarcinoma of the sinonasal tract: a potentially under-recognized form of sinonasal adenocarcinoma with occasional yolk sac tumor-like features. Head Neck Pathol. 2020;14(2):465–472. doi:10.1007/s12105-019-01065-7
15. Zamecnik M, Rychnovsky J, Syrovatka J. Sinonasal SMARCB1 (INI1) deficient carcinoma with yolk sac tumor differentiation: report of a case and comparison with INI1 expression in gonadal germ cell tumors. Int J Surg Pathol. 2018;26(3):245–249. doi:10.1177/1066896917741549
16. Kubota Y, Arakawa Y, Sekiguchi M, et al. A case of malignant rhabdoid tumor mimicking yolk sac tumor. Pediatr Blood Cancer. 2019;66(8):e27784. doi:10.1002/pbc.27784
17. Rooper LM, Gagan J, Brosens LAA, et al. Recurrent loss of SMARCA4 in sinonasal teratocarcinosarcoma. Am J Surg Pathol. 2020;44(10):1331–1339. doi:10.1097/PAS.0000000000001508
18. Manivel C, Wick MR, Dehner LP. Transitional (cylindric) cell carcinoma with endodermal sinus tumor-like features of the nasopharynx and paranasal sinuses. Clinicopathologic and immunohistochemical study of two cases. Arch Pathol Lab Med. 1986;110(3):198–202.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.