Back to Journals » Infection and Drug Resistance » Volume 11
Carriage of colistin-resistant, extended-spectrum β-lactamase-producing Escherichia coli harboring the mcr-1 resistance gene after short-term international travel to Vietnam
Authors Nakayama T, Kumeda Y, Kawahara R , Yamaguchi T, Yamamoto Y
Received 3 October 2017
Accepted for publication 23 January 2018
Published 12 March 2018 Volume 2018:11 Pages 391—395
DOI https://doi.org/10.2147/IDR.S153178
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Dr Joachim Wink
Tatsuya Nakayama,1,2 Yuko Kumeda,3 Ryuji Kawahara,4 Takahiro Yamaguchi,4,5 Yoshimasa Yamamoto5
1Center for Global Initiatives, Osaka University, Suita, Osaka, Japan; 2Division of Biomedical Food Research, National Institute of Health Science, Kawasaki, Kanagawa, Japan; 3Center for Microorganism Control, Osaka Prefecture University, Sakai, Osaka, Japan; 4Division of Microbiology, Osaka Institute of Public Health, Osaka, Japan; 5Graduate School of Pharmaceutical Sciences, Osaka University, Suita, Osaka, Japan
Background: Due to increasing colistin usage, the dissemination of the colistin-resistant gene mcr-1 has been increasingly investigated. The aim of this study was to determine whether a traveler on a short-term international trip to a developing country could bring mcr-1 back to their home country.
Materials and methods: Thirty-four travel events from Japan to Vietnam encompassing 19 travelers were assessed. A fecal specimen was collected from each traveler before and after each travel event and was inoculated on CHROMagar containing cefotaxime (CTX). Three to seven colonies exhibiting the characteristics of Escherichia coli were collected. Susceptibility to antibiotics and extended-spectrum β-lactamase (ESBL) production were determined by the disk diffusion method and the double-disk synergy test, respectively. ESBL-encoding genes were genotyped, and phylogenetic groupings were determined by multiplex polymerase chain reaction (PCR). The presence of mcr-1 was also confirmed by PCR and sequencing.
Results: A total of 175 ESBL-producing E. coli isolated before and up to 2 weeks after traveling to Vietnam were analyzed. Genotyping of ESBL-producing isolates showed that blaCTX-M-1/blaTEM (27.7%) and blaCTX-M-9 (45.9%) were the most prevalent genotypes, while the most frequently detected phylogenetic group was D (41.9%) followed by B2 (23.0%). In a significant number of travel events, travelers brought ESBL-producing E. coli back to Japan and three events by three travelers carried mcr-1. ESBL-producing E. coli isolates harboring mcr-1 were identified as those carrying both blaCTX-M-14 or blaCTX-M-55 and mcr-1.
Conclusion: Using Vietnam as an example, we have shown that even a short-term trip to some countries may result in ESBL-producing mcr-1-positive E. coli carriage by international travelers.
Keywords: traveler, ESBL-producing E. coli, mcr-1, Vietnam, Japan
Introduction
With increasing globalization, people frequently travel to other countries. Under such circumstances, travelers can represent potential reservoirs for the dissemination of extended-spectrum β-lactamase (ESBL)-producing Enterobacteriaceae.1
Our previous study revealed that veterinarians in Vietnam prefer to prescribe colistin-based drugs for use on chicken farms.2 Although colistin is effective for treating multidrug-resistant (MDR) bacteria, including carbapenem-resistant bacteria, this has resulted in the widespread dissemination of colistin-resistant bacteria harboring plasmids encoding the colistin-resistant gene, mcr-1.3 Therefore, the prevalence of mcr-1 requires investigation. Additionally, a recent report showed that colistin-resistant Escherichia coli harboring mcr-1 had been isolated from food animals as well as from farmers and urban persons in Vietnam.4,5 Haenni et al6 reported that the ESBL-related gene blaCTX-M is encoded on mobile plasmids with mcr-1. Recently, transmission of colistin-resistant ESBL-producing E. coli by international travelers from China and India to developed countries has been reported.7,8 Accordingly, we investigated whether travelers were bringing ESBL-producing E. coli harboring mcr-1 back to their home countries after short-term stays in developing countries, such as Vietnam.
Materials and methods
A prospective cohort study was approved by the ethics committees of Osaka University (no eki31) and Osaka Prefectural Institute of Public Health (no 1602-02-2). All participants provided written informed consent. For any participant younger than 18 years, the parents provided written informed consent. A total of 34 travel events from Japan to Vietnam made by 19 travelers (age: 12–61 years, males: 11, length of stay in Vietnam: 2–12 days) between June 2015 and August 2016 were investigated.
An ~0.1 g fecal specimen was collected from each traveler within 6 days before and up to 21 days after each travel event, and 10 mL of sterile phosphate-buffered saline was added to each sample and homogenized. A 100 μL serially diluted homogenized solution was inoculated on CHROMagar™ ECC (Chromagar, Paris, France) containing 1 µg/mL of cefotaxime (CTX) and cultured at 37°C for 24 h. Three to seven colonies exhibiting the characteristics of E. coli were collected from each sample plate.
For E. coli identification, biochemical tests were conducted, including assays in lysine indole motility medium (Nissui, Tokyo, Japan), cellobiose lactose indole β-glucuronidase medium (Kyokuto Pharmaceutical Industrial, Tokyo, Japan), triple sugar iron slants (Nissui), and API 20E (bioMérieux, Marcy l’Etoile, France).
Susceptibility to antibiotics (ampicillin, fosfomycin, cefoxitin, CTX, ceftazidime, meropenem, gentamycin, kanamycin, streptomycin, tetracycline, ciprofloxacin, chloramphenicol, nalidixic acid, and sulfamethoxazole–trimethoprim) was determined by disk diffusion assay. ESBL production was determined by the double-disk synergy test using ceftazidime and CTX with and without clavulanic acid. Both antibiotic susceptibility and ESBL production tests were conducted as recommended by the Clinical and Laboratory Standards Institute.9
Bacterial DNA was extracted by boiling the bacterial suspension in tris(hydroxymethyl)aminomethane–ethylenediaminetetraacetic acid buffer. Genotyping of ESBL-encoding genes and phylogenetic groupings were determined by multiplex polymerase chain reaction (PCR).10 The presence of the colistin-resistant gene mcr-1 was also confirmed by PCR.3 For both ESBL-encoding genes and mcr-1, the PCR products were identified by sequencing.3,10 In mcr-1-positive strains, the colistin minimum inhibitory concentration (MIC) was evaluated by E-test® (bioMérieux).
The phylogenetic grouping of identified E. coli isolates was also determined by triplex PCR using a combination of two genes (chuA and yjaA) and the DNA fragment TSPE4.C2, as previously described.11
The clonal relationships of isolated E. coli strains were assessed by pulsed-field gel electrophoresis (PFGE), as previously described.12
The conjugation assay was performed as previously described.13
Statistical analysis was performed using Student’s t-test. The significance level was set at P<0.05.
Results
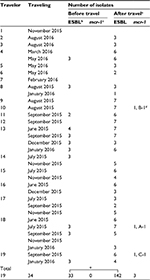
A total of 207 CTX-resistant E. coli strains (43 and 164 before and after travel, respectively) were isolated from the fecal specimens of travelers before and up to 2 weeks after traveling to Vietnam. Among these isolates, 175 strains were identified as ESBL-producing E. coli (Table 1).
A significant number of travel events (88.2%, 30/34) by the 19 travelers brought ESBL-producing E. coli back to Japan, while in 32.3% (11/34) of travel events, it was detected before traveling in seven travelers (Table 1). Among these, 42.9% of ESBL-producing E. coli isolates showed the same PFGE pattern as before traveling.
Genotyping of ESBL-producing isolates showed that blaCTX-M-1 group/blaTEM and blaCTX-M-9 group were the most prevalent genotypes, while blaCTX-M-2 and blaCTX-M-8 groups and blaSHV were not detected in travelers. Phylogenetic analysis of ESBL-producing E. coli isolates was also conducted. The most frequently detected phylogenetic group was D (41.9%, 62/142) followed by B2 (23.0%, 34/142), while B1 (11.5%, 17/142) and A (16.2%, 24/142) were the least common.
ESBL-producing E. coli isolates showed 100% resistance to ampicillin and CTX. In the case of quinolones, ESBL-producing E. coli isolates exhibited a rate of resistance of 76.1% (108/142) to nalidixic acid, and >50% of isolates were resistant to sulfamethoxazole–trimethoprim (66.9%, 95/142) and tetracycline (52.1%, 74/142).
Moreover, three events by three travelers brought back mcr-1. The three travelers with mcr-1, who visited Vietnam on business, did not carry mcr-1 before traveling. ESBL-producing E. coli harboring mcr-1 was identified by the co-occurrence of blaCTX-M and mcr-1. Furthermore, the transmissibility of these mcr-1 genes was confirmed by conjugation assay.
Characteristics of these three ESBL-producing mcr-1- positive E. coli isolates are summarized in Table 2. Traveler A, a 38-year-old man, traveled to Nha Trang by the way of Hanoi for 4 days. One day after returning to Japan, a fecal sample was collected and investigated for the presence of ESBL-producing mcr-1-positive E. coli, and three ESBL-producing isolates with different phylogenetic patterns were identified. These belonged to the blaCTX-M-9 group and the phylogenetic group A, the blaCTX-M-1 group/blaTEM and the phylogenetic group D, and the blaCTX-M-14 genotype and the phylogenetic group B2, the latter of which harbored mcr-1. However, no ESBL-producing E. coli harboring mcr-1 was detected in a sample collected 17 days after returning to Japan. Traveler B traveled to Ho Chi Minh City for 12 days. Three days after returning to Japan, a fecal specimen was collected and four types of ESBL-producing E. coli were identified. These belonged to the blaCTX-M-1 group and the phylogenetic group D, the blaCTX-M-9 group and the phylogenetic group A, the blaCTX-M-1 group/blaTEM and the phylogenetic group D, and the blaCTX-M-55/blaTEM genotype and the phylogenetic group B2, the latter of which harbored mcr-1. Traveler C traveled to Can Tho by the way of Ho Chi Minh City for 5 days. Six days after returning to Japan, a fecal specimen was collected and four types of ESBL-producing E. coli were identified. These belonged to the blaCTX-M-9 group and the phylogenetic group A, the blaCTX-M-1 group and the phylogenetic group D, and the blaCTX-M-14 genotype and the phylogenetic group D, the latter of which harbored mcr-1. In travelers B and C, no ESBL-producing E. coli harboring mcr-1 was detected 20 and 19 days, respectively, after returning to Japan.
Discussion
Widespread dissemination of colistin-resistant MDR bacteria represents a worldwide threat owing to the limited number of effective antibiotics for treatment. In particular, the emergence of the mobile colistin-resistant gene mcr-1 suggests the possibility of the rapid dissemination of MDR bacteria with colistin resistance in a community because of the potential for horizontal gene transfer. In these circumstances, international travelers could act as carriers, transferring mcr-1-positive MDR bacteria to their home countries. Even if only a limited number of travelers carry mcr-1-positive bacteria, the possibility of genetic transfer and expansion of mcr-1 across borders seems to be likely. In this regard, there have been several reports of healthy travelers that carried ESBL-producing E. coli back to their home countries.1,14 However, it is not clear whether a traveler on a short trip can carry mcr-1 back to his/her home country.
In our previous reports, a high prevalence of human ESBL carriage (51%–71%) was demonstrated among Southeast Asian countries, such as Vietnam, Thailand, and Laos.11 In contrast, the dissemination of ESBL-producing E. coli is limited in Japan, where there is a low prevalence of human ESBL carriers.15 To date, only a limited number of bacterial isolates have been reported to harbor mcr-1 in E. coli isolates from healthy animals, but in sick swine, there were high mcr-1- positive bacteria detected in Japan.16,17
It has been reported that in Hanoi, Vietnam, 37.5% of food animals are contaminated with E. coli harboring mcr-1.4 In this study, we revealed that 15.8% of the travelers assessed became the carriers of ESBL-producing E. coli harboring mcr-1 after a short stay in Vietnam. Even though the details of these carriage mechanisms are not clear, our results indicate that travelers may come to resemble Vietnamese residents, a percentage of whom are carriers of mcr-1-positive bacteria (unpublished data), during their stay in Vietnam. The three travelers who became mcr-1 carriers ate breakfast in four-star hotels every morning, and they frequented middle-class restaurants for lunch and dinner. They experienced upper-class conditions during their stays in Vietnam and after returning to Japan. Their genotyping results were similar to those of healthy Vietnamese residents in terms of blaCTX-M genotypes, such as blaCTX-M-1 and blaCTX-M-9 groups.10 Conversely, an abundance of phylogenetic group D was observed in the isolates. However, the reason for this difference from healthy Vietnamese residents10 was not clear. It may be due to differences in intestinal flora between Japanese travelers and Vietnamese residents.
More than 40% of isolates from each traveler before and after travel showed the same PFGE patterns, suggesting low genetic diversity within each traveler. Further study regarding the clonal relationships with residents may provide valuable information on the carriage of MDR bacteria by travelers.
Since the intestinal flora in travelers can be changed by diet and environment factors during travel, it may be responsible for the carriage of ESBL-producing E. coli with mcr-1 after short-term travel.
Our follow-up study showed that the ESBL-producing E. coli harboring mcr-1 disappeared in the three travelers, who were not treated with any antibiotics, within 3 weeks of returning home. Therefore, the stability of the carriage of mcr-1-positive bacteria may be limited if there is no exposure to antibiotics.
The three ESBL-producing E. coli isolates harboring mcr-1 were further characterized, and the colistin MICs of the isolates were relatively low in comparison with those of isolates from food animals (4–8 µg/mL).4 Since antibiotic MICs can be increased by treatment with a high concentration of another antibiotic, the acquisition of high colistin MICs may be possible.
Conclusion
The findings in this study suggest that even a short-term international trip to a developing country may result in ESBL-producing mcr-1-positive E. coli being carried back to the travelers’ home countries. Although the dissemination of E. coli harboring mcr-1 is limited in developing countries, the number of international travelers continues to increase, thereby increasing the risk of spreading E. coli harboring mcr-1 into developed countries.
Acknowledgments
This work was supported by the Japan Agency for Medical Research and Development (AMED)/Japan International Cooperation Agency (JICA) as a part of the Science and Technology Research Partnership for Sustainable Development (SATREPS), and MEXT/JSPS KAKENHI Grant 17H01687.
Disclosure
The authors report no conflicts of interest in this work.
References
Zhong LL, Stoesser N, Doi Y, et al. Carriage of β-lactamase-producing Enterobacteriaceae by Chinese travelers. Lancet Infect Dis. 2017;17(2):138–139. | ||
Nakayama T, Jinnai M, Kawahara R, et al. Frequent use of colistin-based drug treatment to eliminate extended-spectrum beta-lactamase-producing Escherichia coli in backyard chicken farms in Thai Binh Province, Vietnam. Trop Anim Health Prod. 2017;49(1):31–37. | ||
Liu YY, Wang Y, Walsh TR, et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis. 2015;16(2):161–168. | ||
Malhotra-Kumar S, Xavier BB, Das AJ, et al. Colistin-resistant Escherichia coli harboring mcr-1 isolated from food animals in Hanoi, Vietnam. Lancet Infect Dis. 2016;16(3):286–287. | ||
Trung NV, Matamoros S, Carrique-Mas JJ, et al. Zoonotic transmission of mcr-1 colistin resistance gene from small-scale poultry farms, Vietnam. Emerg Infect Dis. 2017;23(3):529–532. | ||
Haenni M, Poirel L, Kieffer N, et al. Co-occurrence of extended spectrum beta-lactamase and mcr-1 encoding genes on plasmids. Lancet Infect Dis. 2016;16(3):281–282. | ||
Payne M, Croxen MA, Lee TD, et al. mcr-1-positive colistin-resistant Escherichia coli in traveler returning to Canada from China. Emerg Infect Dis. 2016;22(9):1673–1675. | ||
Bernasconi OJ, Kuenzli E, Pires J, et al. Travelers can import colistin-resistant enterobacteriaceae, including those possessing the plasmid-mediated mcr-1 gene. Antimicrob Agents Chemother. 2016;60(8):5080–5084. | ||
Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing: Twenty-Fourth Information Supplement. CLSI Document M100-S24. Wayne, PA: CLSI; 2014. | ||
Hoang TA, Nguyen TN, Ueda S, et al. Common findings of blaCTX-M-55- encoding 104-139 kbp plasmids harbored by extended-spectrum β-lactamase-producing Escherichia coli in pork meat, wholesale market workers, and patients with urinary tract infection in Vietnam. Curr Microbiol. 2017;74(2):203–211. | ||
Nakayama T, Ueda S, Huong BTM, et al. Wide dissemination of extended-spectrum beta-lactamase-producing Escherichia coli in community residents in the Indochinese peninsula. Infect Drug Resist. 2015;8:1–5. | ||
Hirai I, Fukui N, Taguchi M, et al. Detection of chromosomal blaCTX-M-15 in Escherichia coli O25b-B2-ST131 isolates from the Kinki region of Japan. Int J Antimicrob Agents. 2013;42(6):500–506. | ||
Kawahara R, Seto K, Taguchi M, et al. Characterization of third-generation-cephalosporin-resistant Shiga toxin-producing strains of Esherichia coli O157:H7 in Japan. J Clin Microbiol. 2015;53(9):3035–3038. | ||
Paltansing S, Vlot JA, Kraakman ME, et al. Extended-spectrum beta-lactamase-producing Enterobacteriaceae among travelers from the Netherlands. Emerg Infect Dis. 2013;19(8):1206–1213. | ||
Luvsansharav UO, Hirai I, Niki M, et al. Prevalence of fecal carriage of extended-spectrum β-lactamase-producing Enterobacteriaceae among healthy adult people in Japan. J Infect Chemother. 2011;17(5):722–725. | ||
Kawanishi M, Abo H, Ozawa M, et al. Prevalence of colistin resistance gene mcr-1 and absence of mcr-2 in Escherichia coli isolated from healthy food-producing animals in Japan. Antimicrob Agents Chemother. 2017;61(1):e2057–e2016. | ||
Kusumoto M, Ogura Y, Gotoh Y, et al. Colistin-resistant mcr-1-positive pathogenic Escherichia coli in swine, Japan, 2007–2014. Emerg Infect Dis. 2016;22(7):1315–1317. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.