Back to Journals » OncoTargets and Therapy » Volume 12
Camellia nitidissima Chi Extract Potentiates the Sensitivity of Gastric Cancer Cells to Paclitaxel via the Induction of Autophagy and Apoptosis
Authors He X, Li H, Zhan M, Li H, Jia A , Lin S, Sun L, Du H , Yuan S, Li Y
Received 22 June 2019
Accepted for publication 9 November 2019
Published 11 December 2019 Volume 2019:12 Pages 10811—10825
DOI https://doi.org/10.2147/OTT.S220453
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr XuYu Yang
Xu He,1,* Hang Li,2,* Meixiao Zhan,1,* Hongyang Li,3 Aiqun Jia,4 Sensen Lin,2 Li Sun,2 Hongzhi Du,5 Shengtao Yuan,2 Yong Li1
1Zhuhai Interventional Medical Center, Zhuhai Precision Medical Center, Zhuhai People’s Hospital, Zhuhai Hospital Affiliated with Jinan University, Zhuhai 519000, People’s Republic of China; 2Jiangsu Key Laboratory of Drug Screening and Jiangsu Center for Pharmacodynamics Research and Evaluation, China Pharmaceutical University, Nanjing 210009, People’s Republic of China; 3Institute of Dermatology, Chinese Academy of Medical Sciences, Peking Union Medical College, Nanjing 210042, People’s Republic of China; 4School of Environmental and Biological Engineering, Nanjing University of Science and Technology, Nanjing 210094, People’s Republic of China; 5School of Pharmacy, Hubei University of Chinese Medicine, Wuhan 430065, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Hongzhi Du; Yong Li Email [email protected]; [email protected]
Background: Camellia nitidissima Chi (CNC) has been applied as a traditional folk medicine for the effective treatment of various diseases. However, there is little research regarding the mechanism of CNC on pharmaceutical function including anticancer effect.
Materials and methods: JHC-4 is a n-butanol extract from CNC. The anti-proliferation effect was evaluated by MTT assays. Monodansylcadaverine (MDC) staining, Western blotting and autophagy inhibitors (CQ and BafA1) were applied to determine whether JHC-4 induced autophagy. The synergistic anticancer effect was evaluated by MTT assays, flow cytometry, Western blotting and autophagy inhibitors. Western blotting was used to explore the influence of PI3K/Akt/mTOR signaling pathway induced by drug treatment.
Results: JHC-4 caused significant growth inhibition and induced autophagy in human gastric cancer cells. Moreover, JHC-4 as an autophagy agonist synergistically potentiated the sensitivity of gastric cancer cells to paclitaxel. Meanwhile, JHC-4 could significantly enhance the growth inhibition effect of paclitaxel by the induction of autophagy and apoptosis. Finally, we demonstrated that the PI3K/Akt/mTOR signaling pathway was involved in the synergistic anti-proliferation effect of JHC-4 and paclitaxel.
Conclusion: All these data indicated that JHC-4 was a novel autophagy inducer when combination with paclitaxel for gastric cancer, which provided the scientific evidence for the use of this Chinese traditional medicine against cancer.
Keywords: Camellia nitidissima Chi, paclitaxel, autophagy, apoptosis, PI3K/Akt/mTOR
Introduction
Camellia nitidissima Chi (CNC) is a well-known economical camellia with golden-yellow flowers and called as the “pandas in plant kingdom”. In China, CNC might be first recorded in Ben Cao Gang Mu and also be recorded as medicinal plant in Guangxi Ethnomedicines Compendium. Importantly, CNC has been applied as a traditional folk medicine for the effective treatment of various diseases, such as hypertension, hyperlipidemia, infection, and cancer.1 However, most previous researches focused on the phytology, plant genetics and breeding of CNC.2 Only our published reports3–8 and several other studies2,9 revealed that CNC contains a variety of physiologically active ingredients, such as tea polysaccharides, polyphenols, flavonoids, and tea saponins. Our group has been engaged in the research of Camellia nitidissima Chi for over 6 years. As our previous studies reported, chemical constituents from CNC could inhibit the formation of AGEs (Advanced Glycation Endproducts)7,8 and reduce pyocyanin production.6 Meanwhile, we preliminarily reported that CNC inhibited the proliferation of EGFR (Epidermal Growth Factor Receptor)-mutant lung cancer cells.3 Importantly, our previous data showed that n-butanol extract (JHC-4) from CNC significantly inhibited AOM/DSS (Azoxymethane/Dextran sulfate sodium) induced colorectal cancer, relieved the colonic pathology of inflammation and ameliorated the serum biochemistry, and noticeably reversed the disturbed metabolic profiling toward the normal state.4 Therefore, we want to evaluate the anti-proliferation effect of the JHC-4 and explore the mechanism.
The accumulation of cytoplasmic vacuoles coincidently was observed after treatment, we thus suspected that autophagy might be the mechanism as abundant previously reported.10,11 Autophagy is a lysosomal-dependent degradation pathway characterized by cytoplasmic vacuolization to maintain cell metabolism. Undoubtedly, it also plays a key role in cancer cell proliferation, chemosensitivity, immunoregulation, migration and so on. Consecutively, Cancer Cell, Nature Reviews Clinical Oncology, Autophagy, and other abundant journals have reported that diet control, gene interference, radiotherapy, immunotherapy and chemotherapy can induce cancer cell autophagy to exert the anti-cancer effect.12 Obviously, induction of autophagy has been a novel strategy for cancer treatment. In recent years, plenty small molecule compounds were discovered and will be potential anticancer drugs.13,14 Similarly, certain natural products such as vitamin D (VD), resveratrol, matrine, etc., also have been confirmed as autophagy inducer in cancer cells.15,16 Therefore, the International Journal of Molecular Sciences published a review:17 'Natural Compounds from Herbs that can Potentially Execute as Autophagy Inducers for Cancer Therapy', which indicates that the induction of autophagy may be the potential mechanism behind Chinese herbal medicine as cancer treatments.
In clinical practice, Chinese herbal medicine plays a vital role in cancer treatment, especially in enhancing efficacy and reducing toxicity when combined with chemical drugs.18,19 But most of the mechanism of the increasing sensitivity is still confused. Interestingly, previous studies have reported that autophagy could potentiate sensitivity to chemoradiotherapy.20,21 In fact, CNC has been applied as a traditional folk medicine for cancer treatment. Whether CNC potentiate the sensitivity of chemical drugs is still unconfirmed by scientific research. In addition, the PI3K/Akt/mTOR pathway is the most classical pathway involved in autophagy, apoptosis, and sensitivity of chemoradiotherapy.22 And the PI3K/Akt/mTOR has been frequently reported as a significant pathway involved in the anticancer effect of Chinese herbal medicine.23 Therefore, we plan to evaluate the anti-proliferation effect of CNC, screen the combination strategies and explore the mechanism.
In this study, we firstly evaluated the anti-cancer effects of JHC-4 on gastric cancer cells. The results suggested that JHC-4 could significantly induce autophagy and inhibit the proliferation. In further experiments, we found that JHC-4 could synergistically potentiate the sensitivity of gastric cancer cells to paclitaxel by the induction of apoptosis. Moreover, it is confirmed that the combination of JHC-4 and paclitaxel-induced apoptosis through autophagy. Finally, we found that the drug combination exhibited synergistic effects might be via the PI3K/Akt/mTOR pathway. In brief, this research provides a scientific basis for the anti-cancer effect of CNC which may be a potential therapeutic strategy for the treatment of gastric cancer in the future.
Materials and Methods
Reagents
The n-butanol extract of CNC (JHC-4) was prepared as previously reported.6,7 The leaves were simmered in hot filtered water for 3 hrs. The clear solution was collected and evaporated using a rotary evaporator at 50°C to a volume of a half. The concentrated solution was extracted with water-saturated n-butanol, which was concentrated to dryness in a vacuum and further lyophilized to obtain a yellow-brown powder of C. nitidissima Chi extract (JHC-4).
Paclitaxel (TAX) was obtained from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). Chloroquine (CQ) and Bafilomycin Al (BafA1) were obtained from Sigma (Saint Louis, MO, USA). MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide, a tetrazole) was obtained from Nanjing Sunshine Biotechnology Ltd (Nanjing, China). Annexin V-PI Detection Kit was purchased from BD Biosciences (San Jose, CA, USA). Monodansylcadaverine (MDC) was purchased from Keygen Company (Nanjing, China). Antibodies against LC3, Atg-7, Atg-3, PARP, cleaved PARP, caspase 9, cleaved caspase 9, PI3K, p-PI3K, Akt, p-Akt, mTOR, p-mTOR and β-actin were obtained from Cell Signaling Technology (Beverly, MA, USA).
Cell Culture
The human cancer cell lines BGC-823 and SGC-7901 were obtained from the BeNa Culture Collection. Furthermore, all cancer cells were authenticated by short tandem repeat (STR profiles are attached in supplemental materials) and tested for mycoplasma contamination. The human cell culture had gained the ethical approval in Zhuhai Hospital Affiliated with Jinan University. Both cell lines were cultured in RPMI-1640 medium (Gibco, Grand Island, NY, USA) with 10% fetal bovine serum (FBS, HyClone, Logan, UT, USA), 100 U/mL penicillin, and 100 U/mL streptomycin (Gibco, Grand Island, NY, USA). The cells were grown at 37°C in a humidified incubator (BB15, Thermo Fisher Scientific Inc., Langenselbold, Germany) with 5% CO2.
MTT Assay
Cell viability was determined by MTT assay as reported.3 Cells were plated in 96-well plates at a density of 2000 cells per well. After 24 hrs incubation, the cells were treated with different concentrations of JHC-4 for 24, 48, or 72 hrs. Subsequently, 20 μL of MTT (0.5 mg/mL) was added into each well for another 4 h. The blue MTT formazan precipitate was then dissolved in 150 μL of DMSO, and the absorbance was measured at 570 nm by a Microplate reader (Tecan, Durham, NC, USA). Cell viability was shown as a percentage of control viability.
Western Blotting
After the cells were treated with JHC-4 or (and) TAX for the indicated time, cell lysates were prepared by RIPA buffer supplemented with complete protease and phosphatase inhibitor mixture (Roche Diagnostics, 4906845001). The concentrations of the protein sample were detected by Bio-Rad Bradford protein assay. Forty microgram of proteins were used to separate in SDS-PAGE system, and the proteins inside the gel were then transferred to Polyvinylidene fluoride (PVDF) membranes. After blocking with 5% non-fat milk in Tris-buffered saline containing 0.1% Tween 20 (TBST), the membranes were incubated with primary antibodies (diluted in 5% BSA in TBST) overnight at 4°C. Following incubation of secondary antibody for 1 hr at room temperature, the immune complexes were visualized by chemiluminescence reagents (Millipore) and then exposed by an image analyzer (Gel 2000, Bio-Rad Laboratories, Inc., Richmond, CA, USA).
Apoptosis Analysis
Apoptosis was determined by flow cytometry.3 BGC-823 cells were treated with JHC-4 or/and TAX in medium for 48 hrs. Cells were then harvested and washed with PBS twice. Subsequently, cells were double-stained with 5 μL of FITC-conjugated Annexin V and 5 μL of propidium iodide (PI) for 15 mins and detected by BD FACS Calibur flow cytometry (BD Biosciences). The data were analyzed by Cellquest software.
MDC Staining
Monodansylcadaverine (MDC) was purchased from Keygen Company (Nanjing, China), it can be used to detect the formation of macro-phage bubble autophagic vacuoles and acidic organelles.24 Gastric cancer cells were treated with JHC-4 for 48 hrs, incubated with MDC (1 mM) for 30 mins. Then, the samples were analyzed by the laser scanning confocal microscope (excitation 380–420 nm, emission 450 nm).
Statistical Analysis
The results were expressed as mean±S.D. from three independent experiments. The data were analyzed by Graph Pad Prism 6.0 and statistical analysis was performed by Student’s t-test (two-tailed). The criterion for statistical significance was taken as P<0.05.
The combination efficacy was expressed by Q value, which was modified from the Burgi formula, and calculated according to the following formula:25
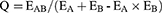
Among them, EAB was the effect when the two drugs (eg. CNC and TAX in this study) are combined, and EA and EB were the effects when Drug A (eg. CNC) and Drug B (eg. TAX) treated alone. When Q ≥ 1.15, it means that the two drugs exert a synergistic effect; when 0.85 <Q <1.15, it means that the two drugs exert an additive effect; when Q ≤ 0.85, it means that the two drugs play an antagonistic role when combined.
Results
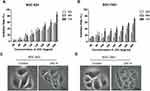
JHC-4 Inhibited the Proliferation of Human Gastric Cancer BGC-823 and SGC-7901 Cells
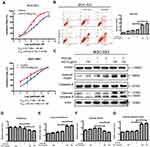
To evaluate the anti-cancer effect on different human gastric cancer cells, we evaluated the anti-cancer effect of JHC-4 on the proliferation of human gastric cancer BGC-823 and SGC-7901 cells by MTT assay. Both cells were treated with indicated concentrations of JHC-4 for 24, 48, and 72 hrs, respectively. As shown in Figure 1A and B, JHC-4 inhibited proliferation inhibition. After 72 hrs, the IC50 value of BGC-823 and SGC-7901 cells were 147.62±16.40 μg/mL and 157.68±18.16 μg/mL, respectively. Interestingly, we found that JHC-4 at 100 μg/mL for 48 hrs could significantly induce the accumulation of cytoplasmic vacuoles (Figure 1C and D), which were often observed in cells undergoing autophagy as abundant previous reports.10,11 These data indicated that JHC-4 exhibited cytotoxic effect on gastric cancer cells and possibly induced autophagy.
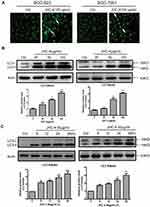
JHC-4 Upregulated the Protein Level of Autophagic Markers in Human Gastric Cancer Cells
As reported,24 MDC staining is a classical method to detect autophagy. To confirm that JHC-4 induced autophagy in gastric cancer cells, the gastric cancer cells were incubated with MDC (1 mM) after JHC-4 treated. Then, the staining results indicated that the acidic vacuoles were enhanced after JHC-4 treatment (Figure 2A), suggesting that JHC-4 induced autophagy possibly. During autophagy, the microtubule-associated protein 1 light chain 3 (LC3) is the mammalian homologue of yeast ATG8. Then, LC3 is converted to lipidated LC3 II and is associated with the autophagic membrane. The accumulation of LC3 II is generally regarded as the marker for autophagy. Therefore, the expression of LC3 is now widely applied to monitor autophagy. To explore the effect of JHC-4 on autophagy, we detected the expression of LC3-II affected by JHC-4. The results showed that the protein levels of LC3-II were induced in a dose-dependent manner in BGC-823 and SGC-7901 cells after JHC-4 treatment (Figure 2B), and the elevation of LC3-II could be detected as early as 12 h after JHC-4 treatment (Figure 2C). These data suggested that autophagy might be involved in the proliferation inhibition induced by JHC-4 treatment.
JHC-4 Induced Autophagic Flux in Human Gastric Cancer Cells
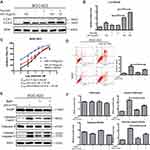
To distinguish the upregulation of LC3-II was due to the accumulation of autophagosomes or the block of the fusion between autophagosomes and lysosomes.26 We pretreated gastric cancer cells with autophagy inhibitors (CQ and BafA1) to evaluate the function of autophagy. As shown in Figure 3A–D, after pretreatment with CQ (A-B) or BafA1 (C-D) in gastric cancer cells for 2 h, JHC-4 (50μg/mL) further increased the protein levels of LC3-II, compared with CQ or BafA1 treated alone. The results were consistent in BGC-823 and SGC-7901 cells. Obviously, these data clearly showed that JHC-4 induced the degradation of the autophagic contents and triggered the autophagic flux in human gastric cancer cells.
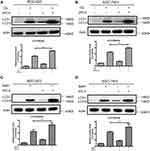
JHC-4 Induced the Formation of Autophagosomes
Morphologically, autophagy is characterized by the formation of double-membrane vesicles called autophagosomes, which are essential for the isolation and degradation of cytoplasmic components.27 To confirm whether the autophagy induced by JHC-4 is caused by the accumulation of autophagosomes, we explored biomarkers of autophagosomes such as the ATG family, according to widely recognized evidences.28 Therefore, the essential proteins involved in the generation of autophagosomes were detected by Western blotting assays. As shown in Figure 4A and B, consistently, JHC-4 treatment dramatically increased the protein levels of Atg-7 and Atg-3 in a time-dependent manner. At the same time, the up-regulation of these proteins was observed as early as 12 hrs after 50 μg/mL JHC-4 treatment (Figure 4C and D). In addition, the experiments on SGC-7901 cells also showed consistent results. Consequently, these findings suggested that JHC-4 induced autophagy via the formation of autophagosomes.
JHC-4 Potentiated the Sensitivity of Gastric Cancer Cells to Paclitaxel via Induction of Apoptosis
In clinical practice, most medicines are not used alone but are combined with other drugs. Especially, Chinese herbal medicine is often applied to enhance the pharmaceutical effect, reduce the toxicity and promote the life quality, when combined with other chemotherapeutic drugs.29 To discover the chemotherapeutic drugs that could exhibit synergistic effects with JHC-4, common chemotherapy drugs were used to screen the combination effects with JHC-4 in gastric cancer cells. The screening results of JHC-4 combined with six common drugs (shown in Tables 1–6) suggested that paclitaxel (TAX) exhibited a significant synergistic effect, navelbine (NVB) showed a slight synergistic effect, while cisplatin (DDP), topotecan (TPT), adriamycin (ADR) and 5-fluorouracil (5-FU) had only additive effect.
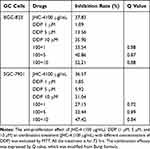 |
Table 1 Q-Value Assess the Combined Effect of JHC-4 and DDP on GC Cells |
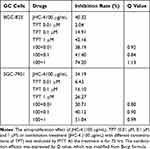 |
Table 2 Q-Value Assess the Combined Effect of JHC-4 and TPT on GC Cells |
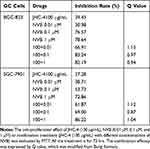 |
Table 3 Q-Value Assess the Combined Effect of JHC-4 and NVB on GC Cells |
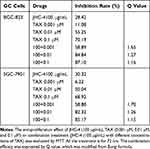 |
Table 4 Q-Value Assess the Combined Effect of JHC-4 and TAX on GC Cells |
 |
Table 5 Q-Value Assess the Combined Effect of JHC-4 and ADR on GC Cells |
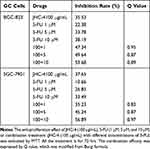 |
Table 6 Q-Value Assess the Combined Effect of JHC-4 and 5-FU on GC Cells |
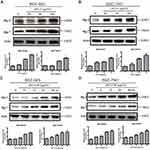
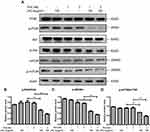
Therefore, further experiments were applied to evaluate the anti-proliferation effect of the combination of JHC-4 with TAX. As is shown in Figure 5A, after drug combination with JHC-4, the IC50 value of TAX was decreased approximately 10 times suggesting that JHC-4 could significantly enhance the anticancer effect of TAX. Usually, apoptosis is the key anticancer effect after treatment. To determine whether apoptosis was induced by the combination treatment, we performed flow cytometry analysis by Annexin V and PI staining. As shown in Figure 5B, the apoptotic fraction was significantly increased by the treatment of JHC-4 combined with TAX, compared with treatment of JHC-4 or TAX alone. In consideration of PARP and the caspase family as markers of apoptosis, we detected the protein levels of apoptosis-related proteins to further investigate the pro-apoptotic effect of JHC-4 combined with TAX. As shown in Figure 5C–G, the results indicated that the combination could decrease the expression of caspase-9 and increase the levels of cleaved caspase-3 and cleaved PARP compared with JHC-4 or TAX alone. In brief, the above results indicated that JHC-4 significantly increased the drug sensitivity of paclitaxel by the induction of apoptosis.
JHC-4 Combined with Paclitaxel-Induced Apoptosis Through Autophagy in Gastric Cancer Cells
Based on the above data shown, then we investigated the relationship between JHC-4-induced autophagy and JHC-4 combined with paclitaxel-induced apoptosis. Firstly, we examined the autophagy effects on gastric cancer cells after combination treatment. The results (Figure 6A and B) showed that drug combination significantly further increased the protein levels of LC3-II compared with JHC-4 or TAX alone. Obviously, JHC-4 combined with paclitaxel-enhanced autophagy in gastric cancer cells. To investigate the relationship between autophagy and the synergistic effect, gastric cancer cells were pretreated with an autophagy inhibitor (BafA1) for 2 hrs to suppress autolysosome formation. After treatment with JHC-4 and TAX for 48 hrs, the combined efficacy was measured by MTT assay. The results showed that the synergistic effect was reversed after BafA1 pretreatment, which indicated that autophagy was involved in the combined efficacy (Figure 6C). Consistently, the data also showed that the apoptosis induced by the combination was reversed after the addition of BafA1 compared with the no inhibitor group (Figure 6D). Meanwhile, the expression of the apoptotic protein decreased with the addition of the inhibitor (Figure 6E and F). In conclusion, all of these results indicated that the drug combination induced apoptosis was via autophagy.
JHC-4 Combined with TAX Induced Autophagy via PI3K/Akt/mTOR Signaling Pathway
In tumor cells, autophagy is regulated by a variety of signaling pathways, PI3K/Akt/mTOR pathway is the most classic one as previously reported.22 mTOR is a key regulator of autophagy initiation and controls the process of autophagy.30 To determine the effects of combination therapy on the PI3K/Akt/mTOR signaling pathways, we detected the related proteins by Western blots. The results showed that JHC-4 treatment with TAX significantly decreased the protein levels of p-PI3K, p-Akt, and p-mTOR compared with JHC-4 or TAX treatment alone (Figure 7A), which indicated that the PI3K/Akt/mTOR signaling pathway was extremely inhibited after the combination therapy. Taken together, JHC-4 enhanced the sensibility of TAX by induction of autophagy and apoptosis might via PI3K/AKT/mTOR pathway.
Discussion
Recently, an increasing number of studies have demonstrated that autophagy plays an important role in numerous aspects of human health, especially the development of cancer and chemoresistance.31 Many researches have shown that natural products can induce autophagy to exert an anti-cancer effect.17 At present, the anti-cancer effect of CNC was poorly revealed, and whether the effect of CNC on cancer is through autophagy was still unknown. Therefore, we reported that the CNC extract (JHC-4) induced autophagic flux and enhanced the efficacy of paclitaxel by apoptosis in gastric cancer cells. Meanwhile, we discovered that the enhanced apoptosis was induced by autophagy. We further confirmed that the PI3K/Akt/mTOR signaling pathway might be involved in the anti-cancer effects of JHC-4 combined with TAX. Since autophagy is an important target for chemo-sensitivity, our discovery may provide a new strategy for reducing the clinical dosage and overcoming the drug resistance of paclitaxel.
Autophagy is regulated by plentiful proteins and pathways. Light chain-3 (LC3-II) is the most representative one, as previously reported.32 The results (Figure 2) showed that LC3-II expression was upregulated in gastric cancer cells after JHC-4 treatment. But the increased expression of LC3-II does not absolutely represent an increase in autophagy. The enhancement of autophagic function is not only relatives to the increase of autophagosomes but also depends on the speed of its degradation. In further experiment, we analyzed the function of autophagy. Bafilomycin (BafA1) is a vacuolar enzyme (proton pump) inhibitor that blocks the fusion of autophagosomes and lysosomes thereby inhibits autophagosome degradation. Meanwhile, chloroquine (CQ) can also inhibit autophagosome degradation. Because both CQ and BafA1 blocked the process of autophagosome degradation, lysosomes accumulate intracellularly. If the drug-induced autophagy, the expression of LC3-II would be further increased.33 Therefore, after CQ or BafA1 pretreatment, JHC-4 further dramatically enhanced the expression of LC3-II compared with CQ or BafA1 alone (Figure 3). In brief, these data indicated that JHC-4 induced autophagy in gastric cancer cells.
In clinical practice, natural medicines are often applied in combination with classical chemotherapy drugs to exert better therapeutic effects.19 After screening combination treatment of JHC-4 with six common chemotherapy drugs, we found that the combination of JHC-4 with TAX exhibited the most remarkable synergistic effects based on different combination strategies (data are shown in Tables 1–6). Paclitaxel is a common drug for the treatment of gastric cancer. Interestingly, recent studies have shown that paclitaxel also induces autophagy while inducing apoptosis.34 SUN’s studies have reported that paclitaxel-induced autophagy can copromote cell death with apoptosis.35 Therefore, the autophagy, respectively, induced by JHC-4 or TAX resulted in apoptosis and death after combination treatment in gastric cancer cells.
According to the characteristics that the excessive cell autophagy can lead to autophagic cell death, the induction of autophagy promoted cell apoptosis and death. Both autophagy and apoptosis are important regulatory mechanisms of cell activities, involved in many physiological and pathological processes. So far, a number of studies have shown that the induction of autophagy synergistically enhanced the chemotherapeutic effect of many cytotoxic drugs on cancer cells.36,37 It has been reported that the relationship between autophagy and apoptosis can be divided into three types: autophagy and apoptosis do not interfere with each other, autophagy induced apoptosis, autophagy inhibited apoptosis.38 In this study, it was found that JHC-4 significantly increased the cytotoxicity of paclitaxel. JHC-4 combined with paclitaxel dramatically induced upregulated of apoptosis-related proteins and apoptosis (Figure 5). Meanwhile, after pretreatment with autophagy inhibitors, the apoptosis induced by combination treatment was somewhat reversed (Figure 6). Therefore, JHC-4 as potential autophagy agonist showed synergistic effect when combined with TAX on gastric cancer cells.
Autophagy and apoptosis are important cellular processes and are regulated by complex signaling networks of different stimulatory factors. PI3K/Akt, a regulator of rapamycin (mTOR), mediated a variety of physiological functions such as cell proliferation, differentiation, migration, apoptosis, and autophagy.39 Therefore, as a key regulator of autophagy, the PI3K/AKT/mTOR signaling pathway is widely activated in a variety of cells and is an important apoptotic signaling pathway. Hence, we found that combination therapy could inhibit the PI3K/AKT/mTOR pathway activity (Figure 7). In brief, these data indicated that JHC-4 potentiated the sensitivity of gastric cancer cells to paclitaxel by the inhibition of the PI3K/AKT/mTOR pathway.
In summary, the anti-proliferation and synergistic effects of CNC have been fully confirmed by a series of experiments. Our research showed that JHC-4 could synergistically potentiate the sensitivity of paclitaxel by inducing autophagy to trigger apoptosis via PI3K/Akt/mTOR signaling in gastric cancer cells. These data provide scientific evidence for CNC as a Chinese traditional medicine for cancer treatment and indicate that CNC might be a potential therapeutic strategy for cancer in the future.
Acknowledgments
This work was financially supported by the National Key Research and Development Program of China (No. 2017YFA0205200), the TCM research project of Health Commission of Hubei Province (ZY2019Q005), the “Young Crops” project of Hubei University of Chinese Medicine (2019ZZX028), the Natural Science Foundation of Jiangsu Province (BK20181330) and the National Natural Science Foundation of China (No: 81903845, 81773766).
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors declare no conflicts of interest in this work.
References
1. Rahayu RP, Prasetyo RA, Purwanto DA, Kresnoadi U, Iskandar RPD, Rubianto M. The immunomodulatory effect of green tea (Camellia sinensis) leaves extract on immunocompromised Wistar rats infected by Candida albicans. Vet World. 2018;11(6):765–770. doi:10.14202/vetworld.2018.765-770
2. Zhou X, Li J, Zhu Y, et al. De novo assembly of the camellia nitidissima transcriptome reveals key genes of flower pigment biosynthesis. Front Plant Sci. 2017;8:1545. doi:10.3389/fpls.2017.01545
3. Hou X, Du H, Yang R, et al. The antitumor activity screening of chemical constituents from Camellia nitidissima Chi. Int J Mol Med. 2018;41(5):2793–2801. doi:10.3892/ijmm.2018.3502
4. Li MH, Du HZ, Kong GJ, et al. Nuclear magnetic resonance-based metabolomics approach to evaluate the prevention effect of Camellia nitidissima Chi on colitis-associated carcinogenesis. Front Pharmacol. 2017;8:447. doi:10.3389/fphar.2017.00447
5. Yang R, Guan Y, Wang W, Chen H, He Z, Jia AQ. Antioxidant capacity of phenolics in Camellia nitidissima Chi flowers and their identification by HPLC Triple TOF MS/MS. PLoS One. 2018;13(4):e0195508. doi:10.1371/journal.pone.0195508
6. Yang R, Guan Y, Zhou J, et al. Phytochemicals from Camellia nitidissima Chi Flowers reduce the pyocyanin production and motility of pseudomonas aeruginosa PAO1. Front Microbiol. 2017;8:2640. doi:10.3389/fmicb.2017.02640
7. Wang W, Liu H, Wang Z, et al. Phytochemicals from Camellia nitidissima Chi inhibited the formation of advanced glycation end-products by scavenging methylglyoxal. Food Chem. 2016;205:204–211. doi:10.1016/j.foodchem.2016.03.019
8. Yang R, Wang WX, Chen HJ, He ZC, Jia AQ. The inhibition of advanced glycation end-products by five fractions and three main flavonoids from Camellia nitidissima Chi flowers. J Food Drug Anal. 2018;26(1):252–259. doi:10.1016/j.jfda.2017.03.007
9. Lu J, Chen R, Zhang M, da Silva JA, Ma G. Plant regeneration via somatic embryogenesis and shoot organogenesis from immature cotyledons of Camellia nitidissima Chi. J Plant Physiol. 2013;170(13):1202–1211. doi:10.1016/j.jplph.2013.03.019
10. Zientara-Rytter K, Subramani S. AIM/LIR-based fluorescent sensors-new tools to monitor mAtg8 functions. Autophagy. 2018;14(6):1074–1078. doi:10.1080/15548627.2018.1454238
11. Ding GB, Sun J, Wu G, et al. Robust anticancer efficacy of a biologically synthesized tumor acidity-responsive and autophagy-inducing functional Beclin 1. ACS Appl Mater Interfaces. 2018;10(6):5227–5239. doi:10.1021/acsami.7b17454
12. Janku F, McConkey DJ, Hong DS, Kurzrock R. Autophagy as a target for anticancer therapy. Nat Rev Clin Oncol. 2011;8:528. doi:10.1038/nrclinonc.2011.71
13. Zhang E, Wang S, Li LL, et al. Discovery of novel jaspine B analogues as autophagy inducer. Bioorg Med Chem Lett. 2017;28:497–502.
14. Guo Q, Yu C, Zhang C, et al. Highly selective, potent, and oral mTOR inhibitor for treatment of cancer as autophagy inducer. J Med Chem. 2018;61(3):881–904. doi:10.1021/acs.jmedchem.7b01402
15. Ansari SS, Sharma AK, Soni H, et al. Induction of ER and mitochondrial stress by the alkylphosphocholine erufosine in oral squamous cell carcinoma cells. Cell Death Dis. 2018;9(3):296. doi:10.1038/s41419-018-0342-2
16. Hu W, Zhang L, Li MX, et al. Vitamin D3 activates the autolysosomal degradation function against Helicobacter pylori through the PDIA3 receptor in gastric epithelial cells. Autophagy. 2019;15:1–19.
17. Lin SR, Fu YS, Tsai MJ, Cheng H, Weng CF. Natural compounds from herbs that can potentially execute as autophagy inducers for cancer therapy. Int J Mol Sci. 2017;18(7). doi:10.3390/ijms18071412
18. Lu Q, Li CL. Therapeutic efficacy and safety of Kang-ai injection combined with platinum-based doublet chemotherapy in advanced NSCLC: a meta-analysis. Life Sci. 2018;210:9–19. doi:10.1016/j.lfs.2018.08.055
19. Li H, Sun L, Li H, et al. DT-13 synergistically enhanced vinorelbine-mediated mitotic arrest through inhibition of FOXM1-BICD2 axis in non-small-cell lung cancer cells. Cell Death Dis. 2017;8(5):e2810. doi:10.1038/cddis.2017.218
20. Wu J, Hu D, Zhang R. Depletion of Bmi-1 enhances 5-fluorouracil-induced apoptosis and autophagy in hepatocellular carcinoma cells. Oncol Lett. 2012;4(4):723–726. doi:10.3892/ol.2012.805
21. Chiu HW, Yeh YL, Wang YC, et al. Combination of the novel histone deacetylase inhibitor YCW1 and radiation induces autophagic cell death through the downregulation of BNIP3 in triple-negative breast cancer cells in vitro and in an orthotopic mouse model. Mol Cancer. 2016;15(1):46. doi:10.1186/s12943-016-0531-5
22. Wang SSS, Chen YHH, Chen N, et al. Hydrogen sulfide promotes autophagy of hepatocellular carcinoma cells through the PI3K/Akt/mTOR signaling pathway. Cell Death Dis. 2017;8. doi:10.1038/cddis.2017.518
23. Li HY, Sun L, De Carvalho EL, et al. DT-13, a saponin monomer of dwarf lilyturf tuber, induces autophagy and potentiates anti-cancer effect of nutrient deprivation. Eur J Pharmacol. 2016;781:164–172. doi:10.1016/j.ejphar.2016.04.016
24. Zhang J, Li Y, Chen X, et al. Autophagy is involved in anticancer effects of matrine on SGC-7901 human gastric cancer cells. Oncol Rep. 2011;26(1):115–124. doi:10.3892/or.2011.1277
25. Ren Y, Zhou X, Mei M, et al. MicroRNA-21 inhibitor sensitizes human glioblastoma cells U251 (PTEN-mutant) and LN229 (PTEN-wild type) to taxol. BMC Cancer. 2010;10:27. doi:10.1186/1471-2407-10-27
26. Yang YP, Liang ZQ, Gu ZL, Qin ZH. Molecular mechanism and regulation of autophagy. Acta Pharmacol Sin. 2005;26(12):1421–1434. doi:10.1111/j.1745-7254.2005.00235.x
27. Ryabovol VV, Minibayeva FV. Molecular MECHANISMS OF AUTOPHAGY IN PLANTS: role of ATG8 proteins in formation and functioning of autophagosomes. Biochem Biokhimiia. 2016;81(4):348–363. doi:10.1134/S0006297916040052
28. Acharya M, Sokolovska A, Tam JM, et al. ⍺v Integrins combine with LC3 and atg5 to regulate Toll-like receptor signalling in B cells. Nat Commun. 2016;7:10917. doi:10.1038/ncomms10917
29. Ji HF, Li XJ, Zhang HY. Natural products and drug discovery. Can thousands of years of ancient medical knowledge lead us to new and powerful drug combinations in the fight against cancer and dementia? EMBO Rep. 2009;10(3):194–200. doi:10.1038/embor.2009.12
30. Brech A, Ahlquist T, Lothe RA, et al. Autophagy in tumour suppression and promotion. Mol Oncol. 2009;3(4):366–375. doi:10.1016/j.molonc.2009.05.007
31. Chen S, Rehman SK, Zhang W, et al. Autophagy is a therapeutic target in anticancer drug resistance. Biochim Biophys. 2010;1806(2):220–229.
32. Kuma A, Matsui M, Mizushima N. LC3, an autophagosome marker, can be incorporated into protein aggregates independent of autophagy: caution in the interpretation of LC3 localization. Autophagy. 2007;3(4):323–328. doi:10.4161/auto.4012
33. Abdel-Mohsen MA, Abdel Malak CA, Abou Yossef MA, El-Shafey ES. Antitumor activity of Copper (I)-nicotinate complex and autophagy modulation in HCC1806 breast cancer cells. Anticancer Agents Med Chem. 2017;17(11):1526–1536. doi:10.2174/1871520617666170327144122
34. Xi G, Hu X, Wu B, et al. Autophagy inhibition promotes paclitaxel-induced apoptosis in cancer cells. Cancer Lett. 2011;307:141–148. doi:10.1016/j.canlet.2011.03.026
35. Sun J, Jin L, Liu J, et al. Effect of autophagy on paclitaxe-l induced CaSki cell death. Cent South Univ T. 2010;36(6):557–565.
36. Li C, Wang Y, Wang C, Yi X, Li M, He X. Anticancer activities of harmine by inducing a pro-death autophagy and apoptosis in human gastric cancer cells. Phytomedicine. 2017;28:10–18. doi:10.1016/j.phymed.2017.02.008
37. Long Z, Chen B, Li L, et al. The reverse-mode NCX1 activity inhibitor KB-R7943 promotes prostate cancer cell death by activating the JNK pathway and blocking autophagic flux. Oncotarget. 2017;7(27):42059–42070.
38. Qian HR, Shi ZQ, Zhu HP, Gu LH, Wang XF, Yang Y. Interplay between apoptosis and autophagy in colorectal cancer. Oncotarget. 2017;8(37):62759–62768. doi:10.18632/oncotarget.18663
39. Gasparri ML, Bardhi E, Ruscito I, et al. PI3K/AKT/mTOR pathway in ovarian cancer treatment: are we on the right track? Geburtshilfe Frauenheilkd. 2017;77(10):1095–1103. doi:10.1055/s-0043-118907
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.