Back to Journals » International Journal of Nanomedicine » Volume 9 » Issue 1
Bone plate composed of a ternary nano-hydroxyapatite/polyamide 66/glass fiber composite: biomechanical properties and biocompatibility
Authors Qiao B , Li J, Zhu Q, Guo S, Qi X, Li W, Wu J, Liu Y, Jiang D
Received 10 November 2013
Accepted for publication 11 January 2014
Published 17 March 2014 Volume 2014:9(1) Pages 1423—1432
DOI https://doi.org/10.2147/IJN.S57353
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Bo Qiao,1 Jidong Li,2 Qingmao Zhu,1 Shuquan Guo,1 Xiaotong Qi,1 Weichao Li,1 Jun Wu,1 Yang Liu,3 Dianming Jiang1
1Department of Orthopaedics, The First Affiliated Hospital of Chongqing Medical University, Chongqing, 2Research Center for Nano-Biomaterials, Analytical and Testing Center, Sichuan University, Chengdu, 3Department of Orthopaedics, The Second Affiliated Hospital of Chongqing Medical University, Chongqing, People's Republic of China
Abstract: An ideal bone plate for internal fixation of bone fractures should have good biomechanical properties and biocompatibility. In this study, we prepared a new nondegradable bone plate composed of a ternary nano-hydroxyapatite/polyamide 66/glass fiber (n-HA/PA66/GF) composite. A breakage area on the n-HA/PA66/GF plate surface was characterized by scanning electron microscopy. Its mechanical properties were investigated using bone-plate constructs and biocompatibility was evaluated in vitro using bone marrow-derived mesenchymal stem cells. The results confirmed that adhesion between the n-HA/PA66 matrix and the glass fibers was strong, with only a few fibers pulled out at the site of breakage. Fractures fixed by the n-HA/PA66/GF plate showed lower stiffness and had satisfactory strength compared with rigid fixation using a titanium plate. Moreover, the results with regard to mesenchymal stem cell morphology, MTT assay, Alizarin Red S staining, enzyme-linked immunosorbent assay, and reverse transcription polymerase chain reaction for alkaline phosphatase and osteocalcin showed that the n-HA/PA66/GF composite was suitable for attachment and proliferation of mesenchymal stem cells, and did not have a negative influence on matrix mineralization or osteogenic differentiation of mesenchymal stem cells. These observations indicate that the n-HA/PA66/GF plate has good biomechanical properties and biocompatibility, and may be considered a new option for internal fixation in orthopedic surgery.
Keywords: nano-hydroxyapatite, polyamide, glass fiber, biomechanics, biocompatibility
Background
Internal fixation using plates and screws is an effective treatment for long bone fractures. The main materials used in bone plates are metals such as stainless steel, cobalt–chromium alloys, and titanium and its alloys. However, rigid fixation using metal plates with such a high elastic modulus would lead to stress shielding, causing bone atrophy and osteopenia, particularly under the plates, or even refractures.1–3 In addition, metal plates are not bioactive and are not ideal materials for internal fixation because biomaterials should have good biofunctionality and biocompatibility.4,5 For this reason, many types of biomaterials, in particular polymers, have been explored as bone plates, among which biodegradable materials are the latest advance. The rationale for using biodegradable materials for fixing fractures is that their mechanical properties are expected to be similar to those of bone and the degradation processes can meet the requirement of healing of fractures. Poly(lactide), poly(L-lactic acid), and poly (D, L-lactide), along with their fiber-reinforced composites have been studied for this purpose, and biodegradable plates have been used successfully in maxillofacial and malleolar surgery.6–9 However, the rate of degradation for these implants in the body is not the same as the healing period for fractures, which is governed by many factors,7,10,11 and nonspecific foreign body reactions are not uncommon.10,12 Further, the poor mechanical properties of biodegradable plates have been the main reason for their limited application.4 In view of the lack of satisfactory performance using biodegradable bone plates, we attempted to develop a new nondegradable biocomposite bone plate with good bioactivity and appropriate biomechanical strength.
Nano-hydroxyapatite/polyamide 66 (n-HA/PA66) is an osteoconductive material used in bone repair.13,14 Previous studies have suggested that an n-HA/PA66 biocomposite can bond directly with bone, remain permanently in the host tissue without causing a foreign body reaction,14,15 and act as a bioactive nondegradable material meeting the requirements for a bone analog.16,17 However, when we attempted to use n-HA/PA66 alone to prepare a bone plate, its mechanical strength was inadequate. Addition of fibers reinforces the strength of polymer matrix composites, so we constructed an n-HA/PA66/glass fiber (n-HA/PA66/GF) composite with slightly greater strength than cortical bone and a very similar elastic modulus as a potential candidate biomaterial for fixing fractures of long bone. However, adding glass fibers could influence the biocompatibility of n-HA/PA66. Therefore, we investigated the biomechanical properties of the n-HA/PA66/GF plate and the biocompatibility of this composite when in direct contact with mesenchymal stem cells (MSCs) in vitro. We hypothesized that the n-HA/PA66/GF plate could repair fractures of long bone effectively in vitro and have no negative influence on proliferation or differentiation of MSCs.
Materials and methods
Materials
The n-HA/PA66 composite was prepared as described before.18 In brief, HA/PA66 composite grains were obtained and mixed with glass fibers; an extrusion method was then used to prepare the ternary composite. The weight ratio used for hydroxyapatite, the glass fibers, and PA66 was 2:3:5. The processing temperature was 240°C–290°C. Subsequently, the ternary composite plate (10 × 75 × 3 mm, with six holes) was obtained using an injection molding method. The titanium plate (12 × 75 × 3 mm, with six holes) and screw (Φ3.5 mm, 22 mm, or 24 mm length) were purchased from BaiDe Medical Instrument Co, Ltd (Jiangsu, People’s Republic of China).
Given that n-HA/PA66 is known to be biocompatible with MSCs, we performed a 3-(4, 5-dimethylthiazol-2yl)-2, 5-diphenyl-2H-tetrazolium bromide (MTT) assay, an enzyme-linked immunosorbent assay (ELISA), and real-time polymerase chain reaction (RT-PCR) in this study using n-HA/PA66 disks as the control. The n-HA/PA66/GF and n-HA/PA66 composites were cut into 8 × 8 × 1 mm disks for study of their biocompatibility in vitro. The MSCs were obtained from the bone marrow of 4-week-old Sprague Dawley rats and cultured in Dulbecco’s Modified Eagle’s Medium/F12 containing 10% fetal bovine serum (Gibco, Grand Island, NY, USA). MSCs at passage 3–6 were used. The osteogenic differentiation medium consisted of Dulbecco’s Modified Eagle’s Medium/F12 supplemented with 10% fetal bovine serum, and 200 U/mL penicillin/streptomycin, 50 mg/L L-ascorbic acid, 0.01 mol/L β-glycerophosphate, and 10−8 mol/L dexamethasone (Sigma-Aldrich, St Louis, MO, USA).
Characterization of n-HA/PA66/GF plate
Scanning electron microscopy of n-HA/PA66/GF plate fracture surface
The n-HA/PA66/GF plate was fractured by applying a bending load until failure. The surface of the fracture was then coated with gold, and the orientation of the fibers and their adhesion to the n-HA/PA66 matrix was observed by scanning electron microscopy (SEM; JSM-7500F, JEOL Ltd, Tokyo, Japan).
Mechanical properties of n-HA/PA66/GF plate
All the mechanical tests in this study were performed using a mechanical testing device (M-3050; Reger Instrument Co, Ltd, Shenzhen, People’s Republic of China) at room temperature and 70% humidity. The bending yield strength of the n-HA/PA66/GF plate was determined by a three-point bending test. Briefly, the crosshead was located at the third hole of the plate with a preload of 50 N because the plate holes are located where the stress is concentrated and are also the weakest part of a plate. The load was applied at a speed of 5 mm per minute until failure. The span length was 40 mm.
Biomechanical analysis of fixation
In this part of the study, the n-HA/PA66/GF and titanium plates (Figure 1) were used to fix transverse fractures on a fresh canine femur. The canine femur diameters were 12–14 mm. For each group, three specimens were used for the bending and torsion tests with the two ends of the femur embedded in a cylindrical pot with polymethylmethacrylate. Briefly, transverse fractures were made using a thin blade saw. An n-HA/PA66/GF or titanium plate with six holes was then placed at the lateral surface of the femur with a 3 mm gap left between the third and fourth holes. Next, six titanium screws were installed to provide bicortical fixation. All the bone-plate constructs were tested to failure under static loading. For the four-point bending test, the preload was set at 50 N for all constructs. Loading was then applied across the entire length of the plate in the gap-closing direction at a speed of 1 mm per minute until construct failure. The outer and inner supports were separated by 150 mm and 75 mm, respectively (Figure 2A). Failures were defined as breakage of plates/bones or as screws migration. The load (N) and displacement (mm) of the crosshead were recorded until failure. The bending stiffness (N/mm) was defined as the ratio of the linear portion of the load-deformation curve,19 and bending strength was calculated using the failure load and distance between the outer and inner supports.20 For the torsion test, the load was applied at a speed of 0.5 degrees per second until failure of the bone-plate construct (Figure 2B). The load (Nm) and angular deformation (degrees) were recorded until failure. Torsion stiffness (Nm per degree) was calculated from the slope of the load-angular deformation curve.20
  | Figure 1 n-HA/PA66/GF plate (A), titanium plate (B), and titanium screws (C) applied in mechanical tests. |
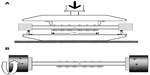  | Figure 2 Schematic drawing of (A) bending and (B) torsion tests using bone-plate constructs. |
In vitro biocompatibility
In this part of the study, MSCs were cocultured with n-HA/PA66/GF disks in osteogenic medium to evaluate the influence of n-HA/PA66/GF on proliferation and osteoblastic differentiation of MSCs. MSCs cultured alone and with n-HA/PA66 disks were used as controls for MTT, ELISA, and RT-PCR.
Culture and seeding of MSCs
MSCs were harvested from the bone marrow of 4-week-old Sprague-Dawley rats. In brief, bone marrow was aspirated from the femur and tibia and subjected to gradient centrifugation (600 g for 20 minutes at 4°C) to obtain mononuclear cells which were cultured in Dulbecco’s Modified Eagle’s Medium/F12 containing 10% fetal bovine serum and 1% penicillin/streptomycin (200 U/mL). An n-HA/PA66/GF or n-HA/PA66 disk was placed in a six-well plate, and the MSCs were then seeded at a density of 1 × 105 in each well. However, for the MTT assay, MSCs were seeded at a density of 5 × 104 in a 24-well plate. The medium was replaced with fresh osteogenic differentiation medium after one day and every 2–3 days thereafter.
SEM
After coculture of the MSCs with n-HA/PA66/GF disks for 3 days, the cell/disk constructs were rinsed with phosphate-buffered saline three times and fixed with 2.5% glutaraldehyde. Adhesion and morphology of the MSCs on the surface of the n-HA/PA66/GF composite was observed by SEM.
MTT assay
An MTT assay was used to determine the viability of MSCs cocultured with the n-HA/PA66/GF composite. After 1, 3, and 7 days of culture, the medium was replaced with serum-free medium and MTT solution (5 mg/mL) was added in each well. After incubation at 37°C for 4 hours, dimethylsulfoxide was added to dissolve the formazan crystals, and optical density was read at a wavelength of 570 nm. Three parallel replicates of each sample were prepared at each time point.
Alizarin Red S staining
Alizarin Red S staining was used to detect matrix mineralization of the MSCs; these were rinsed with phosphate-buffered saline three times and fixed with 4% paraformaldehyde after 21 days of coculture. Next, 1 mL of 1% Alizarin Red S (Sigma-Aldrich) solution was added and incubated for 15 minutes at room temperature. The staining result was observed using a light microscope (BX51; Olympus, Tokyo, Japan).
ELISA of alkaline phosphatase and osteocalcin
Cells from each treatment group were trypsinized and centrifuged on days 3, 7, and 14. After three freeze–thaw cycles, the lysates were centrifuged for 20 minutes at 12,000 g and 4°C. Their protein content was measured using a competitive ELISA kit (BlueGene Biotech Co, Ltd, Shanghai, People’s Republic of China) according to the manufacturer’s directions, in triplicate. Absorbance was read at 450 nm, and the amount of protein was expressed as ng per well.
Gene expression of alkaline phosphatase and osteocalcin
RT-PCR was used to determine mRNA expression in alkaline phosphatase and osteocalcin on days 3, 7, and 14. Total RNA was extracted from the MSCs in the three treatment groups at each time point using Trizol reagent (Takara, Dalian, People’s Republic of China) and reverse-transcribed into complementary DNA (cDNA) using a reverse transcriptase kit (Promega, Madison, WI, USA). RT-PCR analysis was performed with the SYBR Green PCR Master Mix (Toyobo, Tokyo, Japan) on an ABI Prism 7500 sequence detection system (Applied Biosystems Inc, Carlsbad, CA, USA) by monitoring the 18S rRNA gene as a quantitative control. Primer sequences were as follows: rat 18S rRNA (forward, 5′-GAATTCCCAGTAAGTGCGGGTCATA-3′; reverse, 5′-CGAGGGCCTCACTAAACCATC-3′); rat alkaline phosphatase (forward, 5′-CACGGCTGGAGATGGACAAGT-3′; reverse, 5′-GCCTTCACGCCACACAAGTAG-3′); and rat osteocalcin (forward, 5′-CACCACCGTTTAGGGCATGTGT-3′; reverse, 5′-CCGTCCATACTTTCGAGGCAGA-3′). Relative quantification of the target genes was determined using the ΔΔCT method.21
Statistical analysis
Quantitative data are presented as the mean ± standard deviation. A Student’s t-test and one-way analysis of variance was performed to determine statistical significance using Statistical Package for the Social Sciences version 17.0 (SPSS Inc, Chicago, IL, USA). A P-value less than 0.05 was determined to be statistically significant.
Results
SEM observation of fracture surface of n-HA/PA66/GF plate
After bending the n-HA/PA66/GF plate until failure, the fracture surface was observed by SEM. As shown in Figure 3, no obvious n-HA/PA66 matrix cracking on the cross-sectional areas was observed. When the n-HA/PA66/GF plate ruptured, a large number of glass fibers broke, but few fibers were pulled out at the site of the fracture surface. This indicates that adhesion between the n-HA/PA66 matrix and the glass fibers was strong and beneficial with regard to mechanical properties.
  | Figure 3 Scanning electron micrograph of fracture surface on n-HA/PA66/GF plate (×200). |
Mechanical analysis of n-HA/PA66/GF plate
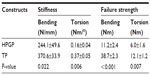
The bending strength of the n-HA/PA66/GF plate was 184.02±5.45 MPa when the load was located at the third hole of a plate. Biomechanical tests of the fixation of the n-HA/PA66/GF plate versus the titanium plate were then performed using transverse fractures in canine femur. In four-point bending and torsion tests, all the n-HA/PA66/GF plate constructs failed by breaking of the plates at the hole sites near the fracture gap (Figure 4A and C), whereas the titanium plate constructs failed because of bone fractures (Figure 4B and D). In the four-point bending test, failure load of n-HA/PA66/GF plate constructs was 596.0±128.4 N compared with 2,065.5±124.3 N for titanium plate constructs, whereas the torsional failure load of the n-HA/PA66/GF plate and titanium plate constructs was 6.0±1.6 Nm and 12.1±1.3 Nm, respectively. The results of both the bending and torsion tests indicate that the stiffness and strength of the n-HA/PA66/GF plate construct was significantly lower than that of the titanium plate constructs (Table 1). The bending and torsion stiffness of the n-HA/PA66/GF plate construct was reduced by 34.1% and 56.8%, respectively, compared with the titanium plate construct. The bending and torsion strength of the n-HA/PA66/GF plate construct was 28.9% and 49.6% that of the titanium plate construct, respectively. The results of the mechanical study show that the fractures fixed by the n-HA/PA66/GF plate had lower stiffness and sufficient strength to decrease the stress shield using rigid fixation.
Attachment and morphology of MSCs on n-HA/PA66/GF disk
The adherence and morphology of the MSCs on the n-HA/PA66/GF disk were observed by SEM. After 3 days of coculture, the MSCs showed a fusiform shape and adhered to the surface of the scaffold by their pseudopodia (Figure 5).
MTT test
Over 7 days of culture, cell numbers increased with culture time and no significant difference was found at any time point between the n-HA/PA66/GF, the n-HA/PA66 composite, and the blank control (Figure 6). The results of the MTT assay showed that the n-HA/PA66/GF biomaterial had no negative effect on proliferation of MSCs.
Alizarin Red S staining
After 21 days of coculture with the n-HA/PA66/GF biomaterial in osteogenic medium, the number of MSCs increased markedly (Figure 7A) and calcium accumulation was visualized by Alizarin Red S staining (Figure 7B). Staining was observed, indicating that most cells formed mineralized calcium deposits.
ELISA assay and RT-PCR for alkaline phosphatase and osteocalcin
To examine the influence of the n-HA/PA66/GF composite on osteoblast differentiation of MSCs, we carried out ELISA and RT-PCR for alkaline phosphatase and osteocalcin, which are indicators of osteogenic differentiation. Alkaline phosphatase and osteocalcin protein content as measured by ELISA is plotted in Figure 8. At 3, 7, and 14 days, the amount of the two proteins increased with culture time when MSCs were cocultured with n-HA/PA66/GF disks; no significant differences were found in comparison with the n-HA/PA66 composite or the blank control. Alkaline phosphatase and osteocalcin gene expression levels were upregulated by 6.1-fold and 12.4-fold on day 14 compared with day 3 in the n-HA/PA66/GF group (Figure 9). Similarly, we did not detect a difference between the three groups in alkaline phosphatase or osteocalcin expression at any time point using RT-PCR. Analyzing the protein and gene expression levels of these osteogenic indicators, we found that n-HA/PA66/GF had no negative influence on osteogenic differentiation of MSCs.
Discussion
Ideally, artificial bone plates should have similar biomechanical properties to bone to guarantee biofunctionality and good biocompatibility in the healing of fractures. Biofunctionality refers to the mechanical properties of biomaterials, whereas biocompatibility represents the compatibility between these materials and the body. Much effort has been made to fabricate bone plates using degradable biomaterials which could replace the metal materials as bone plates. However, degradable plates cannot fix long bones effectively, mainly because of their poor mechanical strength after implantation. Complete degradation and resorption of these composites seemed to proceed quite slowly in vivo.11 Alternative ways of developing fixation devices using bioceramics and biometals have been explored, but their high elastic modulus often results in bone resorption and loosening of implants. Therefore, elimination of stress shielding and enhancement of strength is the primary motivation for development of inorganic/polymer biocomposites. To this end, nondegradable HA/PA66 composites that mimic the structure of natural bone have been studied.
n-HA/PA66 composites have good biocompatibility and desirable mechanical properties, and have been used successfully as scaffolds in orthopedics.13,14,19,22,23 However, n-HA/PA66 composites cannot provide adequate mechanical strength as bone plates for fixation of fractures. In the present study, to enhance the strength of these composites, we added 30 wt% glass fibers to n-HA/PA66 when fabricating the bone plate. The reinforced n-HA/PA66/GF plate has a yield strength of 184 MPa and an elastic modulus of 10–20 GPa,18 and therefore has mechanical properties comparable with those of the human femur.24,25 Improvement in the mechanical properties of the n-HA/PA66 composites was achieved using glass fibers, which are often added to reinforce polymeric matrices. Compared with the titanium plate, the bending and torsion stiffness for fixation of fractures using the n-HA/PA66/GF plate was reduced by 34.1% and 56.8%, respectively. The lower stiffness of fixation provided by the n-HA/PA66/GF plate reduced the stress shield and facilitated interfragmentary movement, allowing the bone to carry more load and stimulating formation of new bone. Hence, secondary healing was more likely to happen when fractures were fixed by an n-HA/PA66/GF plate. In addition, the metal plate would normally be present in the body for at least one year. Even after union of the fracture, stiffness mismatch between the bone and the plate could lead to osteopenia or even refracture of bone.26 Long-term survival of the n-HA/PA66/GF plate in the body would have a less negative influence on bone loss given that the elasticity modulus of the n-HA/PA66/GF composite is much lower than that of metal materials. The bending and torsional strength of fixation using the n-HA/PA66/GF plate was 11.2±2.4 Nm and 6.0±1.6 Nm, respectively, which is 28.9% and 49.6% of the strength of that applying to the titanium plate. To our knowledge, few studies have evaluated the threshold strength of bone-plate constructs when holding fractures because of poor comparability between the different studies with various parameter settings in mechanical tests. However, unlike biodegradable plates, the strength of the nondegradable n-HA/PA66/GF plate can be retained after implantation in the body, indicating that the plates can hold the fractures efficiently until union. Further, the n-HA/PA66/GF bone plate was used instead of the composite without holes in the mechanical tests, and the load was positioned at the screw hole which was the weakest part of the plate, whereas results for the strength of biodegradable composites would normally be based on data from samples without holes. Hence, the n-HA/PA66/GF plate may hold fractures more tightly until fracture union than a biodegradable plate.
In addition to biofunctionality, we evaluated the biocompatibility of n-HA/PA66/GF biomaterials using MSCs. Previous results indicated that the n-HA/PA66/GF biomaterial had no negative effect on adherence or growth of preosteoblastic MC3T3-E1 cells.18 However, MSCs have to be recruited, proliferated, and differentiated into osteogenic cells for bone to regenerate.27 Therefore, MSCs seem to be the best candidate for assessment of the biocompatibility of orthopedic biomaterials. In the present study, the SEM images indicate that the rough surface of the n-HA/PA66/GF composite is suitable for attachment and proliferation of MSCs and that the n-HA/PA66/GF composite had no negative influence on growth of MSCs as assessed by the MTT test. Osteogenic differentiation of MSCs is an important aspect of fracture healing. Expression of alkaline phosphatase reflects early osteoblastic differentiation of MSCs, whereas osteocalcin and matrix mineralization is associated with the final differentiation phase. The results indicate that the n-HA/PA66/GF biomaterial did not affect the expression of osteogenic indicators or matrix mineralization of MSCs. Unlike biodegradable polylactic acids that inhibit proliferation and osteoblastic differentiation of MSCs,28 the nondegradable n-HA/PA66/GF biomaterial neither decreased pH nor released lactic acid with a toxic effect. Therefore, the n-HA/PA66/GF plate can be implanted safely in the body. Given that n-HA/PA66/GF had biomechanical properties similar to that of bone and good biocompatibility, a second surgery to remove the plate after union of fracture can be avoided. However, further study will be needed in vivo to compare the effect of fixation and biocompatibility between the n-HA/PA66/GF plate and the titanium plate, in particular the influence of long-term implantation.
Conclusion
A novel nondegradable n-HA/PA66/GF bone plate was developed in the present study for fixation of long bone fractures. Biomechanical analysis indicate that the n-HA/PA66/GF bone plate has lower stiffness and satisfactory strength compared with the titanium plate. The results of a biocompatibility study in vitro show that the n-HA/PA66/GF biomaterial has no negative influence on proliferation and osteogenic differentiation of bone marrow-derived MSCs. With its good biofunctionality and biocompatibility, the n-HA/PA66/GF bone plate can be expected to be an alternative for fixing fractures in the future.
Acknowledgment
This work was supported by the National Natural Science Foundation of China (51002099) and the Natural Science Foundation Project of CQ CSTC (2009BB5410).
Disclosure
The authors report no conflicts of interest in this work.
References
Uhthoff HK, Finnegan M. The effects of metal plates on post-traumatic remodelling and bone mass. J Bone Joint Surg Br. 1983;65(1):66–71. | |
Uhthoff HK, Boisvert D, Finnegan M. Cortical porosis under plates. Reaction to unloading or to necrosis? J Bone Joint Surg Am. 1994;76(10):1507–1512. | |
Sumitomo N, Noritake K, Hattori T, et al. Experiment study on fracture fixation with low rigidity titanium alloy: plate fixation of tibia fracture model in rabbit. J Mater Sci Mater Med. 2008;19(4):1581–1586. | |
Navarro M, Michiardi A, Castano O, Planell JA. Biomaterials in orthopaedics. J R Soc Interface. 2008;5(27):1137–1158. | |
Golish SR, Mihalko WM. Principles of biomechanics and biomaterials in orthopaedic surgery. Instr Course Lect. 2011;60:575–581. | |
Bos RR, Boering G, Rozema FR, Leenslag JW. Resorbable poly(L-lactide) plates and screws for the fixation of zygomatic fractures. J Oral Maxillofac Surg. 1987;45(9):751–753. | |
Bergsma JE, de Bruijn WC, Rozema FR, Bos RR, Boering G. Late degradation tissue response to poly(L-lactide) bone plates and screws. Biomaterials. 1995;16(1):25–31. | |
Thordarson DB, Samuelson M, Shepherd LE, Merkle PF, Lee J. Bioabsorbable versus stainless steel screw fixation of the syndesmosis in pronation-lateral rotation ankle fractures: a prospective randomized trial. Foot Ankle Int. 2001;22(4):335–338. | |
Ni P, Fu S, Fan M, et al. Preparation of poly(ethylene glycol)/polylactide hybrid fibrous scaffolds for bone tissue engineering. Int J Nanomedicine. 2011;6:3065–3075. | |
Bergsma EJ, Rozema FR, Bos RR, de Bruijn WC. Foreign body reactions to resorbable poly(L-lactide) bone plates and screws used for the fixation of unstable zygomatic fractures. J Oral Maxillofac Surg. 1993;51(6):666–670. | |
Bos RR, Rozema FR, Boering G, et al. Degradation of and tissue reaction to biodegradable poly(L-lactide) for use as internal fixation of fractures: a study in rats. Biomaterials. 1991;12(1):32–36. | |
De Jong WH, Eelco Bergsma J, Robinson JE, Bos RR. Tissue response to partially in vitro predegraded poly-L-lactide implants. Biomaterials. 2005;26(14):1781–1791. | |
Wang X, Li Y, Wei J, de Groot K. Development of biomimetic nano-hydroxyapatite/poly(hexamethylene adipamide) composites. Biomaterials. 2002;23(24):4787–3791. | |
Wang H, Li Y, Zuo Y, Li J, Ma S, Cheng L. Biocompatibility and osteogenesis of biomimetic nano-hydroxyapatite/polyamide composite scaffolds for bone tissue engineering. Biomaterials. 2007;28(22):3338–3348. | |
Li J, Man Y, Zuo Y, et al. In vitro and in vivo evaluation of a nHA/PA66 composite membrane for guided bone regeneration. J Biomater Sci Polym Ed. June 16, 2010. [Epub ahead of print.] | |
Bonfield W. Composites for bone replacement. J Biomed Eng. 1988;10(6):522–526. | |
Bonfield W, Grynpas MD, Tully AE, Bowman J, Abram J. Hydroxyapatite reinforced polyethylene – a mechanically compatible implant material for bone replacement. Biomaterials. 1981;2(3):185–186. | |
Su B, Peng X, Jiang D, et al. In vitro and in vivo evaluations of nano-hydroxyapatite/polyamide 66/glass fibre (n-HA/PA66/GF) as a novel bioactive bone screw. PLoS One. 2013;8(7):e68342. | |
Yanez A, Cuadrado A, Carta JA, Garces G. Screw locking elements: a means to modify the flexibility of osteoporotic fracture fixation with DCPs without compromising system strength or stability. Med Eng Phys. 2012;34(6):717–724. | |
Meeson RL, Goodship AE, Arthurs GI. A biomechanical evaluation of a hybrid dynamic compression plate and a castless arthrodesis plate for pancarpal arthrodesis in dogs. Vet Surg. 2012;41(6):738–744. | |
Alameh M, Jean M, Dejesus D, Buschmann MD, Merzouki A. Chitosanase-based method for RNA isolation from cells transfected with chitosan/siRNA nanocomplexes for real-time RT-PCR in gene silencing. Int J Nanomedicine. 2010;5:473–481. | |
Yang X, Chen Q, Liu L, et al. Comparison of anterior cervical fusion by titanium mesh cage versus nano-hydroxyapatite/polyamide cage following single-level corpectomy. Int Orthop. 2013;37(12):2421–2427. | |
Zhao Z, Jiang D, Ou Y, Tang K, Luo X, Quan Z. A hollow cylindrical nano-hydroxyapatite/polyamide composite strut for cervical reconstruction after cervical corpectomy. J Clin Neurosci. 2012;19(4):536–540. | |
Erickson GM, Catanese J 3rd, Keaveny TM. Evolution of the biomechanical material properties of the femur. Anat Rec. 2002;268(2):115–124. | |
Sedlin ED, Hirsch C. Factors affecting the determination of the physical properties of femoral cortical bone. Acta Orthop Scand. 1966;37(1):29–48. | |
Ramakrishna S, Mayer J, Wintermantel E, Leong KW. Biomedical applications of polymer-composite materials: a review. Compos Sci Technol. 2001;61(9):1189–1224. | |
Marsell R, Einhorn TA. The biology of fracture healing. Injury. 2011;42(6):551–555. | |
Chen T, Zhou Y, Tan WS. Influence of lactic acid on the proliferation, metabolism, and differentiation of rabbit mesenchymal stem cells. Cell Biol Toxicol. 2009;25(6):573–586. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.