Back to Journals » International Journal of Nanomedicine » Volume 12
Biosensing of DNA oxidative damage: a model of using glucose meter for non-glucose biomarker detection
Authors Zhu X, Sarwar M, Yue Q, Chen C, Li CZ
Received 22 October 2016
Accepted for publication 2 December 2016
Published 2 February 2017 Volume 2017:12 Pages 979—987
DOI https://doi.org/10.2147/IJN.S125437
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Thomas Webster
Xuena Zhu,1 Mehenur Sarwar,1 Qiaoli Yue,2 Chunying Chen,3 Chen-Zhong Li1,4
1Nanobioengineering/Bioelectronics Laboratory, Department of Biomedical Engineering, Florida International University, Miami, FL, USA; 2Department of Chemistry, College of Chemistry and Chemical Engineering, Liao Chen University, Shandong, 3CAS Key Laboratory for Biomedical Effects of Nanomaterials and Nanosafety, National Center for Nanoscience and Technology, Beijing, 4Key Laboratory of Analytical Chemistry for Life Science of Shaanxi Province, School of Chemistry and Chemical Engineering, Shaanxi Normal University, Xi’an, People’s Republic of China
Abstract: Non-glucose biomarker-DNA oxidative damage biomarker 8-hydroxy-2'-deoxyguanosine (8-OHdG) has been successfully detected using a smartphone-enabled glucose meter. Through a series of immune reactions and enzymatic reactions on a solid lateral flow platform, 8-OHdG concentration has been converted to a relative amount of glucose, and therefore can be detected by conventional glucose meter directly. The device was able to detect 8-OHdG concentrations in phosphate buffer saline as low as 1.73 ng mL-1 with a dynamic range of 1–200 ng mL-1. Considering the inherent advantages of the personal glucose meter, the demonstration of this device, therefore, should provide new opportunities for the monitoring of a wide range of biomarkers and various target analytes in connection with different molecular recognition events.
Keywords: 8-OHdG, immunostrip, point-of-care, POCTs, biosensor, DNA oxidative damage
Introduction
In recent years, much effort has been devoted toward developing point-of-care (POC) devices.1–9 Among them, paper-based POC devices are a special category due to the advantages of being simple, rapid, on-site, and cost-effective; they have been widely used in home health care and medical testing,10–12 even environmental monitoring.13–15 The lateral flow immunochromatographic assay, also called lateral flow test, is one of the simplest and most popular formats of paper-based POC devices and can be used to detect specific substances, including small molecules,15–18 large proteins,19–21 and even whole pathogens,10 in a sample by using an immunological reaction. However, most of these tests are based on qualitative colorimetry, which is not sufficient in many cases. To overcome this limitation, a number of devices capable of quantitative analysis have been developed.
As we know, medical devices for POC are very costly to develop and manufacture and are difficult to commercialize. As a result, few have been commercialized successfully. The blood glucose meter (BGM) is one of the most successfully commercialized diagnostic devices on the market,22,23 and it is widely used by millions of diabetes patients to monitor their blood glucose levels every day because of its low cost, compact size, simple operation, and reliable quantitative results.2 The successful launch of US Food and Drug Administration (FDA)-approved smartphone-enabled BGMs, which enable the wireless transmission of reminders, test results, and data analysis through smartphone apps, will attract an even larger number of users.24 Generally, a glucose meter is capable of detecting glucose as the unique target. Recently, many groups have reported methods establishing a direct relationship between the concentrations of the targets in the samples and the glucose detected by a BGM, enabling a number of non-glucose targets to be detected quantitatively.2,25,26 However, most of these methods included much liquid handling, such as adding and mixing solution, which should be avoided as much as possible to ensure ease of use. Conversely, a lateral flow device, as mentioned previously, is supposed to replace most of the manual solution addition and mixing involved in the above-mentioned methods.
Here, we describe a novel design that combines the traditional lateral flow strip with a commercialized smartphone-enabled BGM (iBGStar® Blood Glucose Monitoring System) for portable and quantitative detection of a non-glucose target. The concept is demonstrated by using an oxidative DNA damage biomarker, 8-hydroxy-2′-deoxyguanosine (8-OHdG). The basic design of the device is adapted from our previously reported colorimetric visual detection platform17 based on a gold nanoparticles (AuNPs)-based competitive immunoassay. However, visual detection can provide only qualitative and semiquantitative results. Thus, to enable quantitative analysis, we establish a novel method that transforms the detection of the target to the detection of an enzyme invertase. The enzyme converts sucrose into glucose for BGM readout.
Materials and methods
Design and detection mechanism
As shown in Figure 1, the test strip is composed of five components, preloaded with different reagents and pasted onto a plastic backing plate. The sample pad pretreated with buffer (50 mM borate buffer, 1% bovine serum albumin [BSA], and 0.2% Tween 20) is where the strip contacts the liquid sample. Two conjugation pads, which are composed of glass fiber material, contain AuNPs-labeled mouse anti-8-hydroxyguanosine antibody (Ab; has equal binding affinity to 8-OHdG and 8-hydroxyguanosine) and 8-hydroxyguanosine–invertase conjugates, respectively. Next to the conjugation pad II is a flow membrane, which is made of nitrocellulose. There are two reaction zones on this membrane, which are called test line and control line. A goat anti-mouse immunoglobulin G (IgG) Ab is used as the test line capture reagent, whereas a goat anti-invertase Ab is used as the control line capture reagent. The sample moves along the strip due to capillary action and finally gets collected by the last part known as the absorption pad.
  | Figure 1 Design of a test strip for quantitative detection of 8-OHdG. |
To realize non-glucose target detection using a BGM, the relationship between target recognition and glucose generation must be established. In our design, as illustrated in Figure 1, the detection is based on two steps: the first step is that of competitive-type immunoreactions in the lateral flow strip (Figure 2A–D) and the second step contains an enzymatic reaction, which converts sucrose into detectable glucose for measurement by the smartphone-based BGMs (Figure 2E–G).
Upon target introduction (Figure 2A), the target moves along the strip due to capillary force and rehydrates conjugation pads I and II (Figure 2B). 8-OHdG in the sample solution and invertase-conjugated 8-hydroxyguanosine compete to bind to the Ab from the conjugation pad I. After that, the invertase that binds to AuNPs is captured by the test line (Figure 2C), whereas the free invertase is captured by the control line (Figure 2D). Ideally, the higher the concentration of 8-OHdG in the sample solution is the less invertase captured on the test line and the more invertase captured on the control line. Following the first step (~10 min), the test (3 mm) and control (3 mm) lines are cut by scissors and put on a hydrophobic plate (Figure 2E). Twenty microliters of 0.5 M sucrose solution is added on top of each line (Figure 2F). After a certain period of time, the invertase fixed on each line catalyzes the hydrolysis of sucrose to produce a specific amount of glucose for quantitative readout by the smartphone-based BGMs (Figure 2G).
Reagents
BSA, invertase from baker’s yeast (Saccharomyces cerevisiae), sucrose, gold chloride trihydrate, sodium borohydride (NaBH4), sodium periodate (NaIO4), ethylene glycol, potassium carbonate (K2CO3), boric acid, sodium chloride, and sodium phosphate were received from Sigma-Aldrich (St Louis, MO, USA). Trehalose, phosphate buffer saline (PBS, 1X, pH =7.4), and trisodium citrate dihydrate were purchased from VWR (West Chester, PA, USA). Tween 20 was obtained from Fisher Scientific (Fairlawn, NJ, USA). Plastic backing, nitrocellulose membrane, absorbing pad, and cellulose paper were acquired from Millipore (Billerica, MA, USA). 8-OHdG and 8-hydroxyguanosine were purchased from Cayman Chemical (Ann Arbor, MI, USA). Mouse monoclonal antibodies to 8-hydroxyguanosine, polyclonal goat anti-mouse IgG, and polyclonal goat anti-invertase IgG were purchased from Abcam (Cambridge, MA, USA). The iBGStar Blood Glucose Monitoring System and Blood Glucose Test Strips for iBGStar were obtained from Walgreens (Springfield, IL, USA).
Synthesis of AuNPs-labeled anti-8-hydroxyguanosine Ab conjugates for conjugation pad I
The AuNPs-labeled anti-8-hydroxyguanosine Ab conjugates were synthesized according to our previously published method.17 The Supplementary materials also include a detail instruction for the synthesis. For the conjugation, the AuNPs solution (~20 nm) was concentrated fivefold and the pH was adjusted to 8.5–9.0 with 0.1 M K2CO3 in advance for labeling. The Ab with optimal concentration (45 μL, 0.54 mg mL−1) was added to the AuNPs solution (560 μL, 5X) and stirred gently at room temperature for 1 h. The conjugate was stabilized by adding 65 μL 10% BSA in 20 mM sodium borate for a final concentration of 1% and incubated for another 20 min. Then the mixture was centrifuged for 15 min at 7,000 rcf. The supernatant was discarded, and the pellet was resuspended in 600 μL 1% BSA/PBS. Following the same centrifugation step, the supernatant was removed and the soft sediment of conjugates was finally resuspended in 600 μL conjugation buffer, containing 20 mM sodium phosphate, 0.25% Tween 20, 10% trehalose, and 5% BSA. The conjugate was stored at 4°C until required for use. Characterization of AuNP and its conjugation is documented in the Supplementary materials.
Preparation of 8-hydroxyguanosine–invertase conjugates for conjugation pad II
8-hydroxyguanosine (0.8 mg) was dissolved in 800 μL 1X PBS. The solution (1 mg mL−1) was mixed with NaIO4 (200 μL, 50 mM) and the mixture was incubated for 1 h in the dark. The reaction was stopped by adding ethylene glycol (2.5 μL) for 5 min. Then the mixture (500 μL) was mixed with invertase (2.5 mL, 29 g L−1, pH =9.5, adjusted by K2CO3 [50 g L−1]) by adding it dropwise under constant stirring and incubated for another 1 h. After that, NaBH4 (2 mL, 12 g L−1) was added and the mixture was incubated in the dark at 4°C overnight (12–16 h). Finally, the conjugates were dialyzed against 1X PBS and stored at −20°C.
Assembly of the lateral flow immuno strip
The goat anti-mouse IgG Ab (1 mg mL−1) was used as the test line capture reagent, whereas the goat anti-invertase IgG Ab (3.3 mg mL−1) was used as the control line capture reagent. These capture reagents were dispensed by the Linomat 5 dispenser onto a nitrocellulose membrane as the test and control lines. The sample pad was pretreated with buffer (50 mM borate buffer, 1% BSA, and 0.2% Tween 20), then AuNPs-labeled anti-8-hydroxyguanosine Ab (20 μL) and 8-hydroxyguanosine–invertase conjugates (20 μL) were dispensed by pipette onto glass fiber pads, which were called the conjugate pad I and II, respectively. After drying these membranes, the sample pad, conjugate pads, nitrocellulose membrane, and absorbent pad were pasted onto a plastic backing plate, which was already cut into 5-mm wide strips using a strip cutter. Then the strips were stored in a self-sealing plastic bag until use.
Results and discussion
Check the properties of 8-hydroxyguanosine–invertase conjugates
One of the most important components in the design is 8-hydroxyguanosine–invertase conjugates. This is synthesized according to a well-established method.27 The method requires several steps of chemical reactions that might affect the structure and function of the enzyme to a certain extent. Thus, two aspects of the functions of the conjugates were examined before being used for conjugation pad II: enzymatic activity (invertase part) for glucose production and antigenic activity (8-hydroxyguanosine part) for competitive immunoreactions.
The enzymatic activity of the conjugates (invertase part) was tested by mixing 10 μL of conjugates solution with 50 μL of 0.5 M sucrose solution. The BGM readout of the modified invertase was 134±5 mg dL−1 (n=3) after 2 min, while that of the original invertase was 148±7 mg dL−1 (n=3), indicating that the modified invertase retains a high enzymatic activity after the chemical modifications. The antigenic activity of the conjugates was tested through our previously published method.17 As illustrated in Figure 3, competitive type strips are designed for testing small molecules with single antigenic determinants (ie, 8-OHdG), which cannot bind to two antibodies simultaneously. Both 8-OHdG in the sample solution and 8-hydroxyguanosine on the test line compete to bind to the Ab from the conjugation pad. As a result, the intensity of the test line is inversely proportional to the concentration in the sample. Our previous publication has verified that 8-hydroxyguanosine–BSA conjugates maintain considerable Ab capturing capacity. In this study, we compare 8-hydroxyguanosine–invertase conjugates with 8-hydroxyguanosine–BSA conjugates in terms of this capacity (see test line in Figure 4). These two conjugates show no difference at both low (0 ng mL−1) and high (1,000 ng mL−1) 8-OHdG sample loading, demonstrating the feasibility of the function of 8-hydroxyguanosine in the 8-hydroxyguanosine–invertase conjugates for the following application.
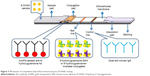  | Figure 3 Mechanism of competitive lateral flow immunoassay for 8-OHdG testing. |
Optimization of the amount of 8-hydroxyguanosine–invertase conjugates used on conjugation pad II and of the time for enzymatic reaction
The amount of 8-hydroxyguanosine–invertase conjugates used on conjugation pad II played an important role in the performance of this detection system. We tested five concentrations of conjugates (original 14.5 mg mL−1, diluted 5-fold, diluted 10-fold, diluted 20-fold, and diluted 30-fold) on conjugation pad II. Two concentration levels, PBS buffer (0 ng mL−1) and high concentration (50 ng mL−1), were prepared and applied to these five types of strips. After 10 min, the test (3 mm) and control (3 mm) lines were cut by scissors and put on a hydrophobic plate. Twenty microliters of 0.5 M sucrose solution was added on top of each line. After a certain period of time, the invertase fixed on each line catalyzes the hydrolysis of sucrose to produce a specific amount of glucose for quantitative BGM readout. At the same time, the catalytic reaction time was also optimized. The readouts of the BGM at 20 and 40 min time points were collected respectively, from which we found that 20 min is not enough for some low concentrations (diluted 10-fold, 20-fold, and 30-fold). However, the relative readout ratio defined as
|
was used to determine the efficiency of the strips to distinguish one concentration from another (Figure 5). As a result, 20-fold diluted conjugates and 40-min reaction time were chosen for the following experiment.
  | Figure 5 RRR for five concentrations of 8-hydroxyguanosine (8-OHdG) invertase conjugates used on the conjugation pad II. |
Quantitative detection of 8-OHdG by a BGM
Under the optimized conditions, a series of 8-OHdG solutions with concentrations of 0, 1, 10, 50, 100, 200, and 1,000 ng mL−1 in 1X PBS buffer were prepared and applied to the strips. After 10 min, the test (3 mm) and control (3 mm) lines were cut by scissors and put on a hydrophobic plate. Twenty microliters of 0.5 M sucrose solution was put on top of each line, and a smartphone-enabled BGM was used for readout after 40 min. As shown in Figure 6, the normalized readout ratio (NRR) defined as
|
decreased when the concentration increased, which is consistent with the theory of the competitive format of detection. From this figure, we can see that NRR has no obvious change when the concentration is >200 ng mL−1. However, elevated concentrations of 8-OHdG from 0 to 200 ng mL−1 lead to obviously decreased NRR. A dynamic range of 1–200 ng mL−1 can be obtained with R2=0.995, and as low as 1.73 ng mL−1 of 8-OHdG can be detected. Considering the cut-off value of 8-OHdG for healthy adults is 20–40 ng mL−1, our approach is, therefore, suitable for early diagnosis of 8-OHdG.
Conclusion
In conclusion, we have developed a simple approach for quantitative detection of 8-OHdG by using a low-cost strip and an easily accessible smartphone-based BGM. The cost for strips as demonstrated in this work is ~10 USD/pc, of which the majority comes from the purchase of 8-OHdG antibodies from commercial vendors. The device was able to detect 8-OHdG concentrations in PBS as low as 1.73 ng mL−1 with a dynamic range of 1–200 ng mL−1 in <1 h. The strip was easy to fabricate and convenient to operate, and the assay did not require any specialized skills or sophisticated instrument. In addition, because of the advantages of the BGM, the demonstration of this approach will provide new opportunities for the detection of other targets. Our current areas of focus are expanding the scope of the strip-BGM design to include other targets and even real biomedical samples testing, further simplifying the design to eliminate the need for target–invertase conjugates to be loaded on conjugation pad II enabling various types of target measurements and optimizing the experimental conditions to acquire a comparable sensitivity compared with other commercialized test kits. The results of these experiments will be reported in due course.
Acknowledgments
This research work is partially supported under grants NSF 1334417, NSFC 91543206, NSFC 21328501, and Shandong Taishan Scholar Project. We also thank Florida International University for Dissertation Year Fellowship (2014–2015) to X Zhu.
Disclosure
The authors report no conflicts of interest in this work.
References
Martinez AW, Phillips ST, Whitesides GM, Carrilho E. Diagnostics for the developing world: microfluidic paper-based analytical devices. Anal Chem. 2010;82(1):3–10. | ||
Xiang Y, Lu Y. Using personal glucose meters and functional DNA sensors to quantify a variety of analytical targets. Nat Chem. 2011;3(9): 697–703. | ||
Martinez AW, Phillips ST, Carrilho E, Thomas SW 3rd, Sindi H, Whitesides GM. Simple telemedicine for developing regions: camera phones and paper-based microfluidic devices for real-time, off-site diagnosis. Anal Chem. 2008;80(10):3699–3707. | ||
Wang S, Zhao X, Khimji I, et al. Integration of cell phone imaging with microchip ELISA to detect ovarian cancer HE4 biomarker in urine at the point-of-care. Lab Chip. 2011;11(20):3411–3418. | ||
Shen L, Hagen JA, Papautsky I. Point-of-care colorimetric detection with a smartphone. Lab Chip. 2012;12(21):4240–4243. | ||
Wang S, Inci F, Chaunzwa TL, et al. Portable microfluidic chip for detection of Escherichia coli in produce and blood. Int J Nanomedicine. 2012;7:2591–2600. | ||
Wang S, Esfahani M, Gurkan UA, Inci F, Kuritzkes DR, Demirci U. Efficient on-chip isolation of HIV subtypes. Lab Chip. 2012;12(8):1508–1515. | ||
Tennico YH, Hutanu D, Koesdjojo MT, Bartel CM, Remcho VT. On-chip aptamer-based sandwich assay for thrombin detection employing magnetic beads and quantum dots. Anal Chem. 2010;82(13):5591–5597. | ||
Foudeh AM, Fatanat Didar T, Veres T, Tabrizian M. Microfluidic designs and techniques using lab-on-a-chip devices for pathogen detection for point-of-care diagnostics. Lab Chip. 2012;12(18):3249–3266. | ||
Li CZ, Vandenberg K, Prabhulkar S, et al. Paper based point-of-care testing disc for multiplex whole cell bacteria analysis. Biosens Bioelectron. 2011;26(11):4342–4348. | ||
Pattarawarapan M, Nangola S, Cressey TR, Tayapiwatana C. Development of a one-step immunochromatographic strip test for the rapid detection of nevirapine (NVP), a commonly used antiretroviral drug for the treatment of HIV/AIDS. Talanta. 2007;71(1):462–470. | ||
Liu G, Mao X, Phillips JA, Xu H, Tan W, Zeng L. Aptamer-nanoparticle strip biosensor for sensitive detection of cancer cells. Anal Chem. 2009;81(24):10013–10018. | ||
Nie Z, Nijhuis CA, Gong J, et al. Electrochemical sensing in paper-based microfluidic devices. Lab Chip. 2010;10(4):477–483. | ||
Apilux A, Dungchai W, Siangproh W, Praphairaksit N, Henry CS, Chailapakul O. Lab-on-paper with dual electrochemical/colorimetric detection for simultaneous determination of gold and iron. Anal Chem. 2010;82(5):1727–1732. | ||
Wang L, Ma W, Chen W, et al. An aptamer-based chromatographic strip assay for sensitive toxin semi-quantitative detection. Biosens Bioelectron. 2011;26(6):3059–3062. | ||
Tang D, Sauceda JC, Lin Z, et al. Magnetic nanogold microspheres-based lateral-flow immunodipstick for rapid detection of aflatoxin B2 in food. Biosens Bioelectron. 2009;25(2):514–518. | ||
Zhu X, Hondroulis E, Liu W, Li CZ. Biosensing approaches for rapid genotoxicity and cytotoxicity assays upon nanomaterial exposure. Small. 2013;9(9–10):1821–1830. | ||
Zhu X, Shah P, Stoff S, Liu H, Li CZ. A paper electrode integrated lateral flow immunosensor for quantitative analysis of oxidative stress induced DNA damage. Analyst. 2014;139(11):2850–2857. | ||
Yang Q, Gong X, Song T, et al. Quantum dot-based immunochromatography test strip for rapid, quantitative and sensitive detection of alpha fetoprotein. Biosens Bioelectron. 2011;30(1):145–150. | ||
Lin YY, Wang J, Liu G, Wu H, Wai CM, Lin Y. A nanoparticle label/immunochromatographic electrochemical biosensor for rapid and sensitive detection of prostate-specific antigen. Biosens Bioelectron. 2008;23(11):1659–1665. | ||
Wang W, Wu WY, Wang W, Zhu JJ. Tree-shaped paper strip for semiquantitative colorimetric detection of protein with self-calibration. J Chromatogr A. 2010;1217(24):3896–3899. | ||
Heller A, Feldman B. Electrochemical glucose sensors and their applications in diabetes management. Chem Rev. 2008;108(7):2482–2505. | ||
Montagnana M, Caputo M, Giavarina D, Lippi G. Overview on self-monitoring of blood glucose. Clin Chim Acta. 2009;402(1–2):7–13. | ||
Carroll AE, Marrero DG, Downs SM. The HealthPia GlucoPack Diabetes phone: a usability study. Diabetes Technol Ther. 2007;9(2):158–164. | ||
Xiang Y, Lu Y. An invasive DNA approach toward a general method for portable quantification of metal ions using a personal glucose meter. Chem Commun (Camb). 2013;49(6):585–587. | ||
Su J, Xu J, Chen Y, Xiang Y, Yuan R, Chai Y. Personal glucose sensor for point-of-care early cancer diagnosis. Chem Commun (Camb). 2012;48(55):6909–6911. | ||
Senapathy P, Ali MA, Jacob MT. Mechanism of coupling periodate-oxidized nucleosides to proteins. FEBS Lett. 1985;190(2):337–341. |
Supplementary materials
Preparation of AuNPs
A well-established citrate reduction method was employed to generate AuNPs of various diameters. Different sizes of AuNPs produce different reflected color. AuNPs with a diameter of ~20 nm intrinsically give a better visibility on paper-based strip when accumulated on the test line, and therefore we used AuNPs with an average size ~20 nm in this work.
Briefly, the hydrogen tetrachloroaurate solution (50 mL, 0.01% ) was added to a Erlenmeyer flask (250 mL), stirred, and brought to the boil on a hotplate. Trisodium citrate solution (1 mL, 1%) was added rapidly to the boiling solution under constant stirring. Gradually, the color changed from pale yellow to wine red. After the color change ceased, the solution was boiled for another 10 min and stirred without heating for another 10 min to complete the reduction of the gold chloride. The size of the particles was characterized by the Zetasizer after the solution reached room temperature.
Characterization of AuNPs pre- and post-conjugation
The wine red-colored solution contains AuNPs with a size of ~20 nm (Zetasizer, Figure S1A) and its corresponding maximum absorbance is exhibited at 520 nm as shown in Figure S1B (ultraviolet–visible spectrophotometer). It is well known that the strong absorbance at 520 nm is attributed to the surface plasmon resonance of AuNPs and the maximum absorption peak will be shifted or disappeared if the nanoparticles aggregate or get further modifications.
As a quality control step post-conjugation, the absorbance spectrum was again measured and compared with that of AuNPs that do not get antibody modifications. A ~3 nm red shift was observed as an indication for successful conjugations (Figure S2).
  | Figure S2 Antibody conjugation of gold nanoparticles produces a red shift in the ultraviolet–visible spectrometer. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.






