Back to Journals » Cancer Management and Research » Volume 11
Biliary tract cancers: current knowledge, clinical candidates and future challenges
Authors Tariq N, McNamara MG , Valle JW
Received 26 August 2018
Accepted for publication 13 December 2018
Published 29 March 2019 Volume 2019:11 Pages 2623—2642
DOI https://doi.org/10.2147/CMAR.S157092
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Ahmet Emre Eşkazan
Noor-ul-Ain Tariq,1,2 Mairéad G McNamara,1,2 Juan W Valle1,2
1Faculty of Biomedicine and Health Sciences, Division of Cancer Sciences, University of Manchester, Manchester M13 9NT, UK; 2Department of Medical Oncology, The Christie NHS Foundation Trust, Manchester M20 4BX, UK
Abstract: Biliary tract cancers (BTCs) are rare with poor prognosis. Due to the advent of genomic sequencing, new data have emerged regarding the molecular makeup of this disease. To add to the complexity, various subtypes also harbor a varied genetic composition. The commonly mutated genes associated with this cancer are KRAS, EGFR, IDH, FGFR and BAP1. Various clinical studies are looking at targeting these genetic mutations. Another therapeutic area of note is the potential for the use of immunotherapy in patients with BTC. Although BTC may be a result of chronic inflammation, this does not necessarily translate into increased immunogenicity. This literature review discusses the diverse molecular and immune-related pathways in patients with BTC and their potential therapeutic implications.
Keywords: biliary tract cancer, intrahepatic cholangiocarcinoma, extrahepatic cholangiocarcinoma, gallbladder cancer, genome sequencing, molecular targets, immunotherapy
Introduction
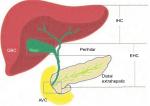
Biliary tract cancers (BTCs) constitute epithelial malignancies of the biliary tree and include the following: gallbladder cancer (GBC), ampulla of Vater cancer (AVC), (the extra-hepatic [EHC] and intra-hepatic [IHC] bile ducts). Historically, the term cholangiocarcinoma (CCA) encompasses EHC and IHC, excluding GBC and AVC.1 The anatomical subtypes of BTC are depicted in Figure 1.
BTC constitutes approximately 3% of all gastrointestinal malignancies2 and is the most common hepatobiliary cancer after hepatocellular carcinoma.3 Unfortunately, the mortality rate (3.58 per 100,000) is very high. This is comparable to the incidence rate (3.64 per 100,000) in England4 and equates to a 5-year survival of 2% in the metastatic setting.5,6 The global prevalence of BTC has risen by a factor of 22%, and 150,000 patients were diagnosed with BTC in 2015.7 Overall, there is a huge variation in incidence with certain areas depicting high prevalence (eg, Japan and South Korea). This can be accounted for by liver fluke (Opisthorchis viverrini [OV] and Clonorchiasis sinensis [CS]) infestation in zones (north-east Thailand and China), where CCA is more common.8,9 Areas with high prevalence of cholelithiasis correspond to a high prevalence of GBC, such as India and Chile.10–12 Geographical regions where the abovementioned risk factors are uncommon have less cases of BTC.11
Apart from the abovementioned risk factors, primary sclerosing cholangitis (PSC), primary biliary cirrhosis (PBC), cirrhosis due to other causes, hepatitis C and congenital malformations such as choledochal cysts and multiple biliary papillomatosis are also associated with an increased risk of developing BTC.13–15 Further to these, patients with germline mutations resulting in Lynch syndrome and BRCA1 and BRCA2 (breast cancer gene 1 and 2) genetic aberrations are also predisposed to BTC. There is a lifetime risk of 2% of developing BTC with Lynch syndrome and RR of 4.97% of developing CCA in carriers of BRCA2.16,17
Treatments for BTC are stratified according to the stage of the disease, where surgery remains the mainstay of cure in early stages, although this represents a small minority of patients (10%–40%).18 Recent data from the BILCAP study support the use of adjuvant capecitabine with an improvement in median overall survival (OS) from 36 (observation alone after surgery) to 53 months (HR 0.75, P=0.028 in the sensitivity analysis).19 For locoregional disease, treatments such as radio-embolization, chemoembolization and external beam radiotherapy can be considered, although due to preliminary evidence these techniques have not yet been adopted in standard practice. For the first-line treatment of advanced disease, the Phase III ABC-02 clinical trial confirmed the superiority of the combination of gemcitabine and cisplatin (GC) over single-agent gemcitabine. Reported median OS was 11.7 months vs 8.1 months, respectively (HR 0.64; 95% CI 0.52–0.80; P<0.001),20 and henceforth this has become a global standard of care for late-stage BTC. Although the modest survival benefit gained from this regimen has not yet been surpassed in a randomized Phase III trial, the combination of gemcitabine with an oral fluoropyrimidine S-1, in a Phase III study, reported a median OS of 15.1 months for the gemcitabine and S-1 arm vs 13.4 months in the GC arm (HR 0.95; 90% CI 0.78–1.15; P=0.046 for non-inferiority).21 This regimen may be considered as an alternative treatment for appropriate patients where comorbidities restrict the use of platinum agents. A Phase II clinical trial evaluating the combination of gemcitabine, cisplatin and nab-paclitaxel in the first-line setting in patients with advanced BTC has reported a superior median progression-free survival (PFS) than that associated historically with the standard GC regimen (11.4 months vs 8.0 months) in the preliminary results with a median OS of 19.2 months. This study (NCT02392637) is estimated to be completed in April 2019.22,23
There is no current defined standard-of-care regimen in the second-line setting in advanced BTC. The current ABC-06 randomized Phase III clinical study is analyzing the role of chemotherapy in this setting vs symptomatic management in patients who have received previous chemotherapy. This study is completed, and the results are expected (NCT01926236).24
The advent of genomic sequencing has led to better understanding of pathogenesis of cancers. Studies in BTC have revealed not only germline and somatic mutations but also genetic aberrations exclusive to anatomical subtypes of BTC. These include KRASTP53ErbB2 in EHC; IDH1/2FGFR1/2 and BAP1 in IHC; and TP53, ErbB2, PIK3CAERrbB1/EGFR in GBC.25–28 These findings may potentiate the development of the use of personalized medicine in this disease group.
Further to the use of genomics and personalized medicine aiming at indubitable targets in this cancer, the in-depth analysis of the immune microenvironment may uncover potential targetable pathways, as BTC has been associated with chronic inflammatory pathology.11
The aim of this review was to evaluate the various potential pathways implicated at the molecular level in the development and progression of BTC and also to address the immune microenvironment and its potential involvement. Localized therapy is beyond the remit of this review.
Methodology
A categorical review of electronic databases was performed, which included Embase, Medline, PubMed and clinicaltrails.gov. Full manuscripts as well as conference abstracts available in the English language and published up to July 2018 employing the following keywords were interrogated: biliary tract cancer, intrahepatic cholangiocarcinoma, extrahepatic cholangiocarcinoma, gall bladder cancer, genome sequencing, KRAS, BRAF, FGF, IDH, VEGF, EGFR, BAP1, molecular targets and immunotherapy.
Significant potential targetable pathways in patients with BTC
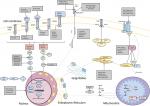
Figure 2 shows the important signaling pathways that may be targetable in patients with BTC.
Ras–Raf–mitogen-activated protein kinase–extracellular signal-regulated kinase (Ras–Raf–MEK–ERK) pathway
The Ras–Raf–MEK–ERK pathway, as shown in Figure 2, is one of the focal signaling pathways for the development of carcinogenesis of BTC,29,30 and one of its first links, KRAS, is a frequently mutated site31 in BTC. Studies in different geographical regions have shown a variance in the frequency of KRAS across the anatomical subtypes, demonstrating 67% in EHC, 45% in IHC and 84% in GBC by the Japanese group,32 42% in EHC, 22% in IHC and 11% in GBC by the Cambridge and MD Anderson groups33 and 22% in EHC and 9% in IHC by the Mayo clinic group.34 Further to differences in frequencies as per the anatomical variant, the KRAS mutation was evaluated in association with clinical outcomes in patients with BTC in different geographical regions by groups in Taiwan, India and USA.32–34
Table 1 summarizes the association of OS with the mutation status for KRAS in the various anatomical subtypes of BTC. The frequency of KRAS mutations ranges from 0% to 41% in these studies, and its presence is associated with worse OS in all the different analyses.27,35–38
Apart from KRAS, other links in this pathway have been evaluated. In Taiwan, the presence of mutations in EGFR, KRAS and BRAF genes was analyzed in relation to median OS. Although the rest of the mutations failed to reveal a significant association with OS, patients carrying EGFR mutations had a median OS of 6 months as compared to 16 months (P≤0.00001) in patients who did not have these mutations.39 A German study evaluated 69 patients with CCA, reporting the presence of BRAF mutations in 22% of the cases, but OS was not significantly correlated with its presence.40 Another study revealed the presence of BRAF mutations in 7.4% of patients with IHC with the OS for patients with wild-type (wt) tumors being 37.3 months as compared to 13.5 months in the population bearing the mutation.41
Potential therapeutic targets for BTC within the Ras–Raf–MEK–ERK pathway
Different novel treatments for targeting the Ras–Raf–MEK–ERK pathway have been analyzed in various studies, including therapeutic agents such as sorafenib, selumetinib, refametinib, trametinib and pazopanib.
Sorafenib is a multi-kinase inhibitor and angiogenesis blocker, which after showing activity in vitro42 was tested in patients with inoperable or advanced IHC in a pilot study reporting an OS of 5.7 months in these cases.43 Further to this, a Phase II study described an increase in toxicities without any benefit in survival outcomes by adding sorafenib to cisplatin and gemcitabine in patients with advanced BTC.44 Selumetinib is another molecule which targets MEK1/2 link by inhibition and has been tested in vitro and in xenografts, prepared from patients with CCA and GBC. It demonstrated activity through cell cycle arrest and delayed reinitiation of S-phase in the cell cycle.45 A Phase II study of selumetinib in monotherapy in patients with predominantly pretreated advanced BTC reported a median OS of 9.8 months.46 Another MEK inhibitor, refametinib and vemurafenib, which is a v-Raf murine sarcoma viral oncogene homolog B (BRAF) inhibitor, is being assessed in Phase II clinical studies (NCT01524978, NCT02346032).47–49 Trametinib, which is an MEK inhibitor, has been tested alongside a VEGFR TKI, pazopanib, resulting in dose-limiting toxicities in nearly all patients (96% [24/25]) in this study who had CCA, while the OS was only 6.4 months.50 Another negative study was closed to accrual after interim analysis, where patients with pretreated advanced BTC who received trametinib failed to show any responses.51
Relevance of the phosphatidylinositol-4,5-bisphosphate 3-kinase–AKT–mammalian target of rapamycin (PIK3–AKT–mTOR) pathway in patients with BTC
The PI3KA–AKT–mTOR pathway, as shown in Figure 2, is also known to be a pivotal link in carcinogenesis.52 Preclinical studies on human CCA cell lines, using MEK1/2 and PI3K inhibitors, showed that the CXC chemokine ligand-12/C-X-C motif chemokine receptor 4 (CXCL-12/CXC4) was blocked by these inhibitors. These receptors/ligands act as an activator for this pathway.53 Another key element of this pathway, mTOR, was assessed via an mTOR inhibitor, everolimus, in vitro showing dose-dependent inhibition of cell proliferation by this inhibitor.54 Further to this, a Phase II study investigated everolimus as a first-line treatment option in patients with advanced BTC, reporting a median OS of 9.5 months and was associated with everolimus resistance in patients with KRAS mutations (P=0.03), with a negative correlation seen between basal pAKT and tAKT with everolimus resistance (P=0.007), regardless of the KRAS status.55
Relevance of the FGF pathway in BTC
The FGF pathway as shown in Figure 2 directly and indirectly upregulates the MAPK and PI3KA pathway, and some studies have shown that fusions in this pathway in BTC have a positive correlation with OS, specifically in the CCA cohort. These include FGFR2-BICCI, FGFR2-AHCYL1, FGFR2-MGEA5, FGFR2-TACC3, FGFR2-KIAA1598 and FGFR2-CREB5.56–59 The frequency of FGFR genetic aberrations varied from 8% to 25% with some exclusiveness for the IHC subtype, where most studies reported a range of 13%–14%. These include mutations, insertions, deletions, gene fusions, and translocations, etc.56,58–62
Table 2 summarizes the variance in FGFR genetic alterations in the anatomical subtypes and its association with survival in patients with BTC. These studies show a wide range of frequency of FGFR genetic aberrations from 0% to 100% with a positive correlation with survival in carriers of the genetic aberration and a more indolent course of disease, resulting in better outcomes.26,27,56,58–62 Again, exclusiveness of the presence of FGFR genetic aberrations in the IHC subtype was noted.
Preclinical studies used a multi-receptor inhibitor (including FGFR), pazopanib, to target cell lines with mutated FGFR2, which resulted in cell cycle arrest63 after which ponatinib and/or pazopanib in two patients with IHC carrying the FGFR fusion gene who achieved partial responses.57 Another highly selective pan-FGFR inhibitor, BGJ398, was assessed in an umbrella study which included patients with CCA and resulted in stable disease in patients with FGFR2 fusions and mutations, whereas one patient who had a KRAS mutation progressed rapidly on this study drug.64 Recently, a Phase II study that analyzed the efficacy of BGJ398 in patients with advanced CCA harboring FGFR2 fusions or other FGFR molecular alterations that were refractory to standard-of-care chemotherapies was reported. It reported an overall response rate (ORR) of 14.8% and a disease control rate (DCR) of 75.4%; however, there was exclusiveness of response in the population harboring FGFR2 fusions only with a DCR of 83.3% in these patients. On the other hand, the patients (n=4) harboring FGFR3 amplifications did not show any response to BGJ398.65 A Phase II basket study analyzed ARQ 087 which is a pan-FGFR inhibitor in patients with CCA, adrenocortical carcinomas and other solid tumors with FGFR1-3 or KIT/PDGFR genetic aberrations. Of the 80 patients analyzed, nearly one-half (n=7) of the 16 patients who exhibited durable response had a genetic alteration in the FGFR pathway, highlighting the response exclusivity to FGFR pathway alterations. In the IHC subgroup, three of five patients with FGFR2 fusions had response (partial response + stable disease) to treatment. All the five patients with IHC, but without FGFR fusions/amplifications, progressed on treatment. In other solid tumors, patients with FGFR amplifications also had a response.66 The same agent, ARQ 087, was analyzed in another Phase I/II open-label study of patients with IHC who were carriers of FGFR2 genes, showing promising results, with a durable DCR in 67% in these patients, and nine patients were still having ongoing treatment at the time the study was published.67 A Phase I study on an FGFR 1–4 inhibitor, TAS-120, in 45 patients with refractory CCA and FGFR2 gene fusions and FGF genetic aberrations has recently reported overall disease control rate (DCR) of 79% with good overall tolerability of the therapeutic agent. A Phase II has been initiated.68 A Phase II study is evaluating an FGFR 1–3 inhibitor, INCB054828, in patients who are refractory to first line and have unresectable, advanced or metastatic CCA harboring the FGFR2 translocation, and it is estimated to be completed in December 2018.69
Relevance of the isocitrate dehydrogenase (IDH) pathway in patients with BTC
IDH acts as a key enzyme for the citric acid cycle,70 as shown in Figure 2, and thus far mutations in this enzyme, which may result in oncogenesis,71 have been exclusively linked to the IHC sub-variant of BTC.72 Studies have reported a frequency of 19%–36% in patients with IHC,61 where Borger et al72 reported it as not only a mutation exclusive to IHC subtype but also the most frequently mutated gene in this disease subtype.
A preclinical study analyzed response to 122 Food and drug administration (FDA)-approved drugs to 17 BTC cell lines including two IHC cell lines with mutations in IDH1, employing high-throughput drug screening to produce a unique drug-sensitivity profile for each individual cell line. This study showed high sensitivity of the IDH-mutant IHC cell lines to dasatinib and saracatinib; both of which are inhibitors of Src family of tyrosine kinases, whereas dasatinib also inhibits (segment of Abelson proto-oncogene and breakpoint cluster region) BCR/ABL family of tyrosine kinases. Interestingly, this response did not correlate with Src activity in the IDH mutant IHC cells, and neither cell lines with IDH mutation in other solid tumors showed such a striking sensitivity. Although this article provides helpful insight into this pathway, it needs to be verified in human studies.73 Another preclinical study employed high-throughput screening to evaluate cell inhibition with 484 small molecular targeting compounds on cell lines and organoids derived from patients with IHC and EHC. This study reported pathways of resistance through micro-RNA 21 (MIR21) to heat shock protein 90 (HSP90) inhibitors. All cell lines, irrespective of mutations, were sensitive to HSP90 inhibitors, but high levels of MIR21 conferred resistance to these molecules. Not only this study was able to identify a potential therapeutic agent but also a biomarker for the efficacy of these agents warrants further evaluation in studies.74
The IDH mutations in patients who had liver fluke infestation (OV and CV) leading to CCA were analyzed and found to be prevalent in cases of IHC which were not associated with OV. The non-OV-associated group had a higher prevalence of IDH mutations: 9.3% as compared to 2.8% in the OV-associated cases.75
Table 3 summarizes the frequency and correlation of survival in patients with BTC and IDH mutations.
After the promising results of the Phase I study of an IDH1 inhibitor, AG-120, in patients with previously treated advanced BTC which showed stable disease in 56% of patients76 with IHC and EHC carrying the IDH1 mutation, a Phase III randomized clinical trial (RCT), “ClarIDHy”, has been developed, comparing AG-120 with a placebo in patients with CCA who carry the mutation in IDH1.77 It is estimated to be completed in August 2020.
Relevance of the Wingless-related integration (Wnt) pathway in patients with BTC
The Wnt signaling cascade is a complex intracellular signaling pathway, as shown in Figure 2, and its dysfunctionality can lead to stimulation of genes, such as c-myc, c-jun, VEGF and cyclin D.78,79 A preclinical study reported increased expression of Wnt and its components in human CCA and IHC cell lines, whereby the blockage of the Wnt pathway resulted in increased apoptosis and cell cycle arrest.80 This pathway has also been studied in relation to liver fluke infestation and one of its components, ubiquitin E3 ligase ring finger 43 (RNF43), was found to be mutated in 9.3% of cases of CCA which were associated with OV, alongside a negative trend for survival in these patients (HR 7.775; P<0.001).81 However, despite the abovementioned findings, apart from a preclinical study evaluating an inhibitor of the Wnt pathway, ie, Dickkopf-1 (DKK1) in cells lines from various tumor sites, including BTC, which suppressed cell invasion and growth, especially in cell lines which had a high expression of DKK1 gene.82 Currently in this particular pathway there no current trails in BTC.
Relevance of the deoxyribonucleic acid damage response (DDR) pathway in patients with BTC
Functional BRCA1 and BRCA2 genes are essential for genomic stability and help the nuclei in resisting damage to deoxyribonucleic acid (DNA). These genes are one of the tumor suppressor genes, and defects in these have been associated with apoptosis and malignant cell transformation.83 The breast cancer linkage consortium reports an RR of developing CCA in carriers of BRCA2 mutations to be 4.97%.84 The combined data from six studies61,85–89 evaluating 142 patients with all four types of BTC stated a frequency of 2.41% for BRCA2 and 1.81% for BRCA1 genetic aberrations by the CCA Cancer Genome Atlas 2018,90 reporting a median survival of approximately 24 months (all stages). A retrospective study evaluated 18 patients with CCA who either carried germline (five cases) or somatic (13 cases) mutations reporting a median OS of 25 months in advanced stages (III and IV) and 40.27 months in early disease (stage I and II). These patients were also evaluated for response to treatments, where a platinum-based chemotherapy agent was compared to a poly-ADP ribose polymerase inhibitor (PARPi). The later showed a better outcome, and patients who were treated with PARPi were reported to have an OS going up to 64.76 months.16 A Phase I/II study, which is estimated to start in August 2018, is going to analyze the dose and side effects of liposomal irinotecan alongside a PARPi (rucaparib) in various cancers including BTC and is expected to be completed in 2021.91 Another Phase II trial is analyzing the ORR with a PARPi (niraparib) in patients with BAP1 and other DNA damage response pathway-deficient cancers including CCA and is expected to be completed in 2021.92
Clinical relevance of key targets identified in patients with BTC
After discussing individual potential implicated pathways, this review now evaluates some further mechanistic targets, including angiogenesis and their role in BTC.
Angiogenesis in tumors of patients with BTC
Although factors such as vascular endothelial growth factor (VEGF), FGF and EGF, which promote angiogenesis, have been identified in patients with BTC,93,94 BTCs are considered as hypovascularized tumors.95 This fact is further supported in patients with IHC, by the presence of low microvessel density (MVD) in tumors, which denotes areas of neovascularization.96 Another study reported a mean MVD of 30.5 vessels per ×200 optical field in a sample of 62 patients with GBC. Within this cohort, patients who had higher MVD had a worse median OS (2-year survival of 25%) as compared to patients with low MVD (2-year survival of 43%).97 The same group evaluated MVD in another sample of 60 patients with GBC and revealed an MVD of 20 per ×200 optical field.98 Another study on 118 patients with GBC confirmed a correlation of tumor stage and liver metastasis with MVD and classified MVD as an independent prognostic factor.99
Role of VEGF in patients with BTC
VEGF, which has been reported as a pivotal angiogenesis factor, was found to be highly expressed at 75.6% in a study of 33 surgically resected cases of CCA.100 A larger analysis of tumors from 236 patients for molecular profiling revealed the presence of the VEGF gene in 53.8% (n=57/106) of IHC and 59.2% (n=77/130) of EHC cases.101 A study that assessed 60 cases of patients with GBC by immunohistochemistry revealed a high VEGF expression in 27 cases and a low VEGF expression in 33 cases,98 where no significant association between VEGF expression and survival was found.98
Among the various VEGF inhibitors, bevacizumab and cediranib have been assessed in patients with BTC. A Phase II study that evaluated the addition of bevacizumab to gemcitabine and oxaliplatin reported a median OS of 14.2 months in patients with advanced IHC (n=22) and 8.5 months in patients with advanced GBC (n=10), whereas median OS was not given for patients with EHC (n=3).102 A Phase III study, ABC-03, evaluated the addition of cediranib (vs placebo) to GC chemotherapy in patients with advanced BTC. Although this study failed to reach its primary end point (improvement in PFS) or show a significant difference in OS, the response rate improved by 25% in the cediranib arm (P=0.0036).103 A Phase II study comparing ramucirumab (VEGFR antibody) vs merestinib (MET inhibitor) vs placebo, in combination with GC in patients with advanced or metastatic BTC as a first-line treatment, has completed accrual and is awaiting results.104
Role of EGFR in patients with BTC
The EGFR/HER2 receptor acts through targeting all the leading pathways including the Ras-Raf-MAP-ERK pathway, the PI3k-AKT-mTOR pathway, the phospholipase C, Ca2+/calmodulin-dependent kinase (CaMK/PKC), Janus-associated kinase (JAK) pathway and the STAT protein pathway105 which makes it a highly susceptible anti-tumorigenesis target. It was found to be present in 100% of IHC samples, 52.6% of EHC samples and 38.5% of GBC samples from treatment-naïve patients.106
Table 4 summaries the various EGFR antibodies such as erlotinib, cetuximab and panitumumab which have been analyzed in various combinations with gemcitabine in selective (KRAS wt) and nonselective patient groups with advanced (inoperable or metastatic) BTC in Phase II and III clinical studies. However, the largest Phase III study that analyzed samples from 268 patients who were diagnosed with all the four types of advanced BTC failed to show any difference in OS by the addition of erlotinib to gemcitabine and oxaliplatin.107
So far, with little or no clinical benefit, EGFR inhibitors are perhaps not the right therapeutic choice for patients with BTC until a predictive biomarker for EGFR inhibitors is developed, and trialing these targeted treatments in BTC is not advised. Although work in colorectal cancer examining the role of KRAS status in therapeutic decision-making has been validated,108 the abovementioned evidence fails to confirm the role of KRAS status or EGFR expression in the therapeutic management of BTC.
Role of BAP1 in patients with BTC
Germline mutations in the BAP1 gene have been associated with cancers of the uvea, kidney, skin and mesothelium,109 and it has been identified as a tumor suppressor gene.110 A study of 64 patients has reported mutations in this gene in 20% of patients with IHC and 6% of patients with GBC.61 A larger study reported the presence of BAP1 mutations in 26% of cases from a cohort of 211 patients with IHC.111
This gene was also analyzed in 209 patients in association with OV-associated cases of CCA, and a 10.5% frequency of BAP1 genes in non-OV-associated cases was reported in comparison to 2.8% in OV-associated cases.75
The presence of aberrations in the BAP1 gene was associated with short time to recurrence in postsurgical patients with CCA in a study that included 75 patients. It was also associated with shorter OS in patients with EHC (8.9 months vs 19.9 months, P=0.007), when compared to patients with EHC, who did not have the BAP1 gene mutation.26 Another study that reviewed 22 patients with CCA who bore mutations in the BAP1 gene reported a mean time to progression of 3.8 months in these cases, and a patient who had undergone curative resection presented with recurrence 8 weeks after surgery, illustrating the aggressive nature of this disease.112
Histone deacetylase inhibitors (HDACI) have been used to target BAP1 mutations and have shown preclinical activity with 30% inhibitory effect in the CCA cell lines113 and in combination with cisplatin led to cytotoxicity, inhibition of growth and increased cell apoptosis in another preclinical study using CCA cell lines.114
Differentiation of the proliferation and the inflammation class
Further to the abovementioned details, a multinational study has presented an interesting concept of defining two unique classes of BTC to help understand the tumor biology of BTC. This study assessed 149 samples (including all stages) of IHC from Milan, Barcelona and New York. The study analyzed genomic mutations using high-density single-nucleotide polymorphism array and gene expression profiles. It classified the samples into two broad categories: the “inflammation class” and the “proliferation class”. The first class, ie, “the inflammation class”, constituted 38% of the total samples and was found to have overexpression of cytokines and activation of STAT3. However, “the proliferation class”, accounting for 62% of the total samples, harbored activated oncogenic pathways with mutations expressed in MAPK, Ras pathways and KRAS, BRAF genes. A better median OS was associated with the inflammation class, ie, 47.2 months compared to 24.3 months in the proliferation class (P=0.048).115 This innovative concept needs further exploration to assess whether this work can be used translationally.
Table 5 summarizes the current trials in targeted treatments in BTC registered on clinicaltrials.gov (last accessed on August 25, 2018).
Potential for the use of immunotherapy in patients with BTC
BTCs have been associated with chronic inflammatory conditions and viral infections; therefore, there may be a role for immunomodulatory agents in this disease group. Understanding the underlying immune environment may yield a successful strategy to target this poor prognostic disease group.
The concept of immunosurveillance and immune editing has been supported by Dunn et al.116 They introduced the concept of elimination whereby the host immunity consisting of natural killer T cells (NKTC), natural killer cells (NKC), interferon γ (IFN γ; initiates immune reactions) led to cytotoxic death of cancer cells. This was followed by equilibrium whereby the immune environment and the cancer cells lived in harmony. Eventually, leading to escape, whereby the cancer cells that survived the equilibrium phase form tumorigenic growths. Research was conducted on the development of carcinoma in situ leading to fulminant tumors in a large sample size of 375 patients with BTC. This study reported an increase in the number of macrophages as the precancerous lesion developed into carcinoma, whereas B-lymphocytes, CD8+ T cells, CD4+ T cells, regulatory T cells, mast cells and NKC steadily declined as the cancer formed.117 The same cells that reduced in number as the cancer formed were associated with better prognosis, highlighting a robust immunosurveillance mechanism led by these cells.
Inflammatory markers using neutrophil-to-lymphocyte ratio (NLR) and derived neutrophil-to-lymphocyte ratio (dNLR) have been evaluated as prognostic biomarkers in BTC. Higher values of NLR ≥3.0 in patients with BTC were associated with a poor OS of 12 months as compared to patients who had lower values of NLR <3.0 with an OS of 21.6 months (adjusted HR =1.26, P=0.01).118 Similarly, high dNLR was associated with poor prognosis.119
Although the role of cancer vaccines has been evaluated in Phase I clinical studies in BTC, the modest benefit has not sparked enough interest to lead to further trials.120–122 Another important component of the immune environment is the cytokines that were analyzed in a cohort of 54 patients with inoperable or advanced BTC who had stable disease after first-line chemotherapy followed by chemoradiation. This was followed by IL2 and retinoic acid (RA) infusion. Although only a small number completed treatment (seven patients), the median OS was not reached when the trial was reported (at 27.5 months).123 Apart from vaccines, adoptive cell therapy (ACT) is also being evaluated in BTC, albeit in small case series or case reports. This involves using the patient’s own cells, which are adapted after extrapolating them from the host. The reformed cells are then again infused into the host after the depletion of lymph in patients. A single case study of a locally advanced patient with IHC treated with ACT was reported as being disease-free 3.5 years after surgery at the time of the case report.124 In another case report, a patient with metastatic CCA who received ACT followed by IL2 was reported to have stable disease for 13 months.125 A case series that reported on the combination of the use of vaccine and ACT in the adjuvant setting reported a better OS of 31.9 months in the patients who received this adjuvant treatment strategy as compared to a median OS of 17.4 months in patients (P=0.022) who underwent surgery alone.126
Immune check point inhibitors are currently being used across various poor prognostic tumor groups with good results; however, there is some association of programmed cell death protein ligand-1 (PD-L1), expression and effectiveness of these treatments.127,128 Expression of PD-L1 in BTC shows a wide range from 29% to 100%,129,130 and the full analysis of the KEYNOTE-028 study is still awaited. This Phase Ib trial is evaluating the effects of treatment with a monoclonal antibody against human immune cell check point programmed death 1 (PD-1), pembrolizumab in patients with previously pretreated advanced BTC who have PD-L1 expression.131 BTCs are infrequently associated with Lynch syndrome, a genetic disorder thatpredisposes to microsatellite instability (MSI) and mismatch repair deficiency (MMR).17 With the food and drug association (FDA) approval of pembrolizumab132 for MSI- and MMR-deficient tumors, in patients who have BTC associated with Lynch syndrome, there may be an option for treating them with immune check point inhibitors, where available..133
Table 6 summarizes the current immune-mediated trials in BTC registered on clinicaltrials.gov (last accessed on August 25, 2018).
  | Table 6 Current trials involving the immune system in BTC registered on clinicaltrials.gov Notes: Tariq NU, Vogel A, McNamara MG, Valle JW. Biliary tract cancer: implicated immune-mediated pathways and their associated potential targets. Oncol Res Treat. 2018;41(5):298–304. Copyright © 2018 Karger Publishers, Basel, Switzerland.203 Abbreviations: aBTC, advanced biliary tract cancer; aCCA, advanced cholangiocarcinoma; BTC, biliary tract cancer; ca, carcinoma; CapeOx, capecitabine and oxaliplatin; CCA, cholangiocarcinoma; chemo, chemotherapy; CTLA4, cytotoxic T-lymphocyte-associated antigen 4; GBC, gallbladder cancer; GC, gemcitabine and cisplatin; HCC, hepatocellular carcinoma; HER, human EGF receptor; mBTC, metastatic biliary tract cancer; MEK, mitogen-activated protein kinase; NKC, natural killer cell; PD-1, programmed death 1; PD-L1, programmed cell death ligand-1; TKI, tyrosine kinase inhibitor. |
Discussion
BTCs are rare cancers which are poorly understood and have few treatment options, low response rates and bad prognosis. The rarity and difficulty in getting good diagnostic samples pose hurdles to effective development of translational research. The various complex issues that govern this disease group include the following: identification of a driver mutation, heterogeneity that exists within the tumor, difficulty in getting repeat samples on recurrence and difference in the behavior of cell lines from real-life patients.
Available information on genomic and somatic mutations in patients with BTC has expanded, but this comes with its own limitations. Various techniques used for molecular profiling yield different results, and there is a lack of global standardization. Much work is needed to reduce the variance in the results obtained across the use of different methodologies.
Intra-tumoral heterogeneity has been an area of debate from as early as 1976, further supplemented by work done in clonal evolution in 1990 by Fearon and Vogelstein.134,135 Further research has been completed recently in this area, where central and peripheral samples from the same tumorigenic mass, from four patients with surgically resected IHC, were evaluated for private and common mutations. Therefore, private mutations were defined as exclusive mutations found in only one region of one tumor for one patient. In contrast, common mutations were, as the name suggests, the mutations which were found in most of the patients. Overall, 75% of patients exhibited private mutations in the center as well as the periphery, whereas one patient had a high percentage (58%) of private mutations in the periphery. The average mean percentage of private mutations was 12% across all samples in all patients.136 Although exciting, this heterogeneity limits the use of personalized medicine in everyday clinical practice. In lung adenocarcinomas, these private mutations or “neoantigens” have been shown to increase sensitivity to immune check point inhibitors, such as anti-cytotoxic T-lymphocyte-associated antigen 4 (anti-CTLA4) and PD-1 inhibitors with resulting improved outcomes.137
Another important facet of this cancer where limited research has been performed is the “tumor microenvironment” (TME). The dense collagenous stroma constitutes TME and contains important components such as cancer-associated fibroblasts (CAFs), α-smooth muscle actin (α-SMA+), which probably originate from activated hepatic stellate cells or hepatic portal fibroblasts.138 In a mouse study, it was reported that there was intrahepatic accumulation of extracellular matrix components, type III collagen and activated fibroblasts, which then resulted in CCA genesis and progression, in mice that were treated with carbon tetrachloride (CCL4).139 Indeed, studies in pancreatic ductal adenocarcinoma have previously shown a role of the stroma in tumor growth.140
Another point to note is that most of the molecular profiling studies include the analysis of surgical samples and therefore represent early-stage disease, which may not be a true representation of the patients who are seen in clinics.
The role of immunomodulating treatments in BTC is still an area of exploration, and none of the current immune investigational drugs have been approved in this disease group. The expression of PD-L1 is a predictive biomarker in other tumor sites, such as non-small-cell lung cancer (NSCLC), for the efficacy of these immunotherapies. Apart from this biomarker, another recently emerging predictor of response is the human microbiome, where certain bacterial species are associated with clinical efficacy of immunotherapies.141
The use of monotherapy vs combination treatment in advanced BTC is also an issue. Historically, clinical studies have used both novel agents as monotherapy as well as in combination with cytotoxic treatments. However, further research in combining treatments that potentiate cytotoxic effects and are at the same time tolerable is necessary. There is also a niche for developing prognostic and predictive biomarkers in BTC to better inform treatment choice.
Currently, there are still gaps in the understanding of the whole process that governs carcinogenesis and resistance to treatments in BTC, and future studies may be able to address this dilemma. In time, prospective studies may further identify novel therapies targeting this disease and lead to improvements in survival outcome.
Disclosure
Noor-ul-Ain Tariq received honoraria for lecturers, participation in writing guidelines and travel reimbursements in 2014 from Boehringer Ingelheim and received funding from the Timpson fellowship. Boehringer Ingelheim or Timpson have no influence over the contents of this review. Mairéad G McNamara was advisory board member of Ipsen, SHIRE, Celgene and Sirtex, received research support from NuCana BioMed Ltd. and SHIRE, received honoraria for participation in Speaker’s Bureau from Pfizer and Ipsen and received travel expenses from Bayer. Juan W Valle received travel grants from Celgene, Ipsen, Novartis, NuCana for more than 5 years, received honoraria for participation in Speakers’ Bureau for Abbott, Celgene, Ipsen, Novartis, Pfizer and Sirtex and provided consulting or advisory role for for Abbott, Agios, AstraZeneca, Baxalta, Bioven, Celgene, Delcath, Genoscience Pharma, Incyte, Ipsen, Keocyt, Lilly, Merck, MidaTech, Mundipharma, Novartis, NuCana, PCI Biotech, Pfizer, Pieris Pharmaceuticals and QED Pharmaceuticals. The authors report no other conflicts of interest in this work.
References
de Groen PC, Gores GJ, Larusso NF, Gunderson LL, Nagorney DM. Biliary tract cancers. N Engl J Med Overseas Ed. 1999;341(18):1368–1378. | ||
Charbel H, Al-Kawas FH. Cholangiocarcinoma: epidemiology, risk factors, pathogenesis, and diagnosis. Curr Gastroenterol Rep. 2011;13(2):182–187. | ||
Hennedige TP, Neo WT, Venkatesh SK. Imaging of malignancies of the biliary tract-an update. Cancer Imaging. 2014;14(1):1470–7330. | ||
England Public Health. National Cancer Intelligence Network: Rare and Less Common Cancers, Incidence and Mortality in England, London; 2015. | ||
Cancer.gov. Surveillance epidemiology and end results program; 2015. Seer Data. Available from: https://seer.cancer.gov/statfacts/html/livibd.html. Accessed February 20, 2019. | ||
In:Seer Data%U. Available from: https://seer.cancer.gov/csr/1975_2014/results_merged/sect_1901_overview.pdf#search=biliary+tract+cancer+incidence%~Seer.Cancer.gov. | ||
Vos T, Allen C, Arora M, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388(10053):1545–1602. | ||
Parkin DM, Srivatanakul P, Khlat M. Liver cancer in Thailand. I. A case-control study of cholangiocarcinoma. Int J Cancer. 1991;48(3):323–328. | ||
Khan SA, Toledano MB, Taylor-Robinson SD. Epidemiology, risk factors, and pathogenesis of cholangiocarcinoma. HPB. 2008;10(2):77–82. | ||
Randi G, Malvezzi M, Levi F, et al. Epidemiology of biliary tract cancers: an update. Ann Oncol. 2009;20(1):146–159. | ||
Khan ZR, Neugut AI, Ahsan H, Chabot JA. Risk factors for biliary tract cancers. Am J Gastroenterol. 1999;94(1):149–152. | ||
Kirstein MM, Vogel A. Epidemiology and risk factors of cholangiocarcinoma. Visc Med. 2016;32(6):395–400. | ||
Khan SA, Toledano MB, Taylor-Robinson SD. Epidemiology, risk factors, and pathogenesis of cholangiocarcinoma. HPB. 2008;10(2):77–82. | ||
Lee SS, Kim MH, Lee SK, et al. Clinicopathologic review of 58 patients with biliary papillomatosis. Cancer. 2004;100(4):783–793. | ||
Chapman RW. Risk factors for biliary tract carcinogenesis. Ann Oncol. 1999;10(Suppl 4):S308–S311. | ||
Golan T, Raitses-Gurevich M, Kelley RK, et al. Overall survival and clinical characteristics of BRCA-associated cholangiocarcinoma: a multicenter retrospective study. Oncologist. 2017;22(7):804–810. | ||
Shigeyasu K, Tanakaya K, Nagasaka T, et al. Early detection of metachronous bile duct cancer in Lynch syndrome: report of a case. Surg Today. 2014;44(10):1975–1981. | ||
Cidon EU. Resectable cholangiocarcinoma: reviewing the role of adjuvant strategies. Clin Med Insights Oncol. 2016;10:CMO.S32821. | ||
Primrose JN, Fox R, Palmer DH, et al. Adjuvant capecitabine for biliary tract cancer: The BILCAP randomized study. American Society of Clinical Oncology. Vol. 35; 2017:4006–4183. | ||
Valle J, Wasan H, Palmer DH, et al. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N Engl J Med. 2010;362(14):1273–1281. | ||
Morizane C, Okusaka T, Mizusawa J, et al. Randomized phase III study of gemcitabine plus S-1 combination therapy versus gemcitabine plus cisplatin combination therapy in advanced biliary tract cancer: a Japan Clinical Oncology Group study (JCOG1113, FUGA-BT). J Clin Oncol. 2018;36(4_suppl):205. | ||
Shroff RT, Borad MJ, Xiao L, et al. A phase II trial of gemcitabine (G), cisplatin (C), and nab-paclitaxel (N) in advanced biliary tract cancers (aBTCs). J Clin Oncol. 2017;35(15_suppl):4018. | ||
Shroff RT, Xiao L, Kaseb AO. A phase II trial of gemcitabine (G), cisplatin (C), and nab-paclitaxel (N) in advanced biliary tract cancers (aBTCs): Updated survival analysis. J Clin Oncol. 2018;36(4_suppl):350. | ||
The Christie NHS Foundation Trust, UK Cancer Research [homepage on the Internet]. Active symptom control alone or with mFOLFOX chemotherapy for locally advanced/metastatic biliary tract cancers; 2014. Available from: https://ClinicalTrials.gov/show/NCT01926236. Accessed January 3, 2019. | ||
Jain A, Javle M. Molecular profiling of biliary tract cancer: a target rich disease molecular profiling of biliary tract cancer: a target rich disease. J Gastrointest Cancer. 2016;7(5):797–803. | ||
Churi CR, Shroff R, Wang Y, et al. Mutation profiling in cholangiocarcinoma: prognostic and therapeutic implications. PLoS One. 2014;9(12):e115383–116203. | ||
Javle M, Bekaii-Saab T, Jain A, et al. Biliary cancer: utility of next-generation sequencing for clinical management. Cancer. 2016;122(24):3838–3847. | ||
Nakamura H, Arai Y, Totoki Y, et al. Genomic spectra of biliary tract cancer. Nat Genet. 2015;47(9):1003–1010. | ||
Sohal DP, Shrotriya S, Abazeed M, Cruise M, Khorana A. Molecular characteristics of biliary tract cancer. Crit Rev Oncol Hematol. 2016;107:111–118. | ||
Hezel AF, Deshpande V, Zhu AX. Genetics of biliary tract cancers and emerging targeted therapies. J Clin Oncol. 2010;28(21):3531–3540. | ||
Bridgewater JA, Goodman KA, Kalyan A, Mulcahy MF. Biliary tract cancer: epidemiology, radiotherapy and molecular profiling. Am Soc Clin Oncol Educ Book. 2016;35:e194–203. | ||
Ohashi K, Tstsumi M, Nakajima Y, Nakano H, Konishi Y. Ki-ras point mutations and proliferation activity in biliary tract carcinomas. Br J Cancer. 1996;74(6):930–935. | ||
Ross JS, Wang K, Catenacci DVT, et al. Comprehensive genomic profiling of biliary tract cancers to reveal tumor-specific differences and genomic alterations. J of Clin Oncol. 2015;33(3 Suppl):231. | ||
Voss JS, Holtegaard LM, Kerr SE, et al. Molecular profiling of cholangiocarcinoma shows potential for targeted therapy treatment decisions. Hum Pathol. 2013;44(7):1216–1222. | ||
Malats N, Porta M, Piñol JL, Corominas JM, Rifà J, Real FX. Ki-ras mutations as a prognostic factor in extrahepatic bile system cancer. PANK-ras I project investigators. J Clin Oncol. 1995;13(7):1679–1686. | ||
Chen T-C, Jan Y-Y, Yeh T-S. K-ras mutation is strongly associated with perineural invasion and represents an independent prognostic factor of intrahepatic cholangiocarcinoma after hepatectomy. Ann Surg Oncol. 2012;19(S3):675–681. | ||
Kazmi HR, Chandra A, Nigam J, Noushif M, Parmar D, Gupta V. Prognostic significance of K- ras codon 12 mutation in patients with resected gallbladder cancer. Dig Surg. 2013;30(3):233–239. | ||
Mafficini A, Amato E, Cataldo I, et al. Ampulla of vater carcinoma: sequencing analysis identifies TP53 status as a novel independent prognostic factor and potentially actionable ERBB, PI3K, and WNT pathways gene mutations. Ann Surg. 2018;267(1):149–156. | ||
Chang YT, Chang MC, Huang KW, Tung CC, Hsu C, Wong JM. Clinicopathological and prognostic significances of EGFR, KRAS and BRAF mutations in biliary tract carcinomas in Taiwan. J Gastroenterol Hepatol. 2014;29(5):1119–1125. | ||
Tannapfel A, Sommerer F, Benicke M, et al. Mutations of the BRAF gene in cholangiocarcinoma but not in hepatocellular carcinoma. Gut. 2003;52(5):706–712. | ||
Robertson S, Hyder O, Dodson R, et al. The frequency of KRAS and BRAF mutations in intrahepatic cholangiocarcinomas and their correlation with clinical outcome. Hum Pathol. 2013;44(12):2768–2773. | ||
Huether A, Höpfner M, Baradari V, Schuppan D, Scherübl H. Sorafenib alone or as combination therapy for growth control of cholangiocarcinoma. Biochem Pharmacol. 2007;73(9):1308–1317. | ||
Luo X, Jia W, Huang Z, et al. Effectiveness and safety of sorafenib in the treatment of unresectable and advanced intrahepatic cholangiocarcinoma: a pilot study. Oncotarget. 2017;8(10):17246. | ||
Lee JK, Capanu M, O’Reilly EM, et al. A phase II study of gemcitabine and cisplatin plus sorafenib in patients with advanced biliary adenocarcinomas. Br J Cancer. 2013;109(4):915–919. | ||
Xu J, Knox JJ, Ibrahimov E, et al. Sequence dependence of MEK inhibitor AZD6244 combined with gemcitabine for the treatment of biliary cancer. Clin Cancer Res. 2013;19(1):118–127. | ||
Bekaii-Saab T, Phelps MA, Li X, et al. Multi-institutional phase II study of selumetinib in patients with metastatic biliary cancers. J Clin Oncol. 2011;29(17):2357–2363. | ||
Roche H-L [homepage on the Internet]. A Study of Zelboraf (Vemurafenib) in Patients with BRAF V600 mutation-positive cancers; 2012. Available from: https://ClinicalTrials.gov/show/NCT01524978. Accessed January 3, 2019. | ||
University Health Network, Toronto [homepage on the Internet]. A study of different dosing schedules of selumetinib with Cisplatin/Gemcitabine (CIS/GEM) versus CIS/GEM alone in biliary cancer; 2014. Available from: https://ClinicalTrials.gov/show/NCT02151084. Accessed January 3, 2019. | ||
Samsung Medical Center [homepage on the Internet]. Phase II study of Refametinib, a MEK inhibitor, as second-line treatment in advanced biliary tract adenocarcinoma; 2015. Available from: https://ClinicalTrials.gov/show/NCT02346032. Accessed January 3, 2019. | ||
Shroff RT, Yarchoan M, O’Connor A, et al. The oral VEGF receptor tyrosine kinase inhibitor pazopanib in combination with the MEK inhibitor trametinib in advanced cholangiocarcinoma. Br J Cancer. 2017;116(11):1402–1407. | ||
Kim RD, McDonough SL, El-Khoueiry AB, et al. SWOG S1310: Randomized phase II trial of single agent MEK inhibitor trametinib vs. 5-fluorouracil or capecitabine in refractory advanced biliary cancer. J Clinl Oncol. 2017;35(15_suppl):4016. | ||
Cully M, You H, Levine AJ, Mak TW. Beyond PTEN mutations: the PI3K pathway as an integrator of multiple inputs during tumorigenesis. Nat Rev Cancer. 2006;6(3):184–192. | ||
Leelawat K, Leelawat S, Narong S, Hongeng S. Roles of the MEK1/2 and AKT pathways in CXCL12/CXCR4 induced cholangiocarcinoma cell invasion. World J Gastroenterol. 2007;13(10):1561. | ||
Moolthiya P, Tohtong R, Keeratichamroen S, Leelawat K. Role of mTOR inhibitor in cholangiocarcinoma cell progression. Oncol Lett. 2014;7(3):854–860. | ||
Yeung YH, Chionh FJM, Price TJ. Phase II study of everolimus monotherapy as first-line treatment in advanced biliary tract cancer: RADichol. J Clinl Oncol. 2014;32(15_suppl):4101. | ||
Arai Y, Totoki Y, Hosoda F, et al. Fibroblast growth factor receptor 2 tyrosine kinase fusions define a unique molecular subtype of cholangiocarcinoma. Hepatology. 2014;59(4):1427–1434. | ||
Borad MJ, Champion MD, Egan JB, et al. Integrated genomic characterization reveals novel, therapeutically relevant drug targets in FGFR and EGFR pathways in sporadic intrahepatic cholangiocarcinoma. PLoS Genet. 2014;10(2):e1004135. | ||
Wu YM, Su F, Kalyana-Sundaram S, et al. Identification of targetable FGFR gene fusions in diverse cancers. Cancer Discov. 2013;3(6):636–647. | ||
Zheng Z, Liebers M, Zhelyazkova B, et al. Anchored multiplex PCR for targeted next-generation sequencing. Nat Med. 2014;20(12):1479–1484. | ||
Graham RP, Barr Fritcher EG, Pestova E, et al. Fibroblast growth factor receptor 2 translocations in intrahepatic cholangiocarcinoma. Hum Pathol. 2014;45(8):1630–1638. | ||
Jiao Y, Pawlik TM, Anders RA, et al. Exome sequencing identifies frequent inactivating mutations in BAP1, ARID1A and PBRM1 in intrahepatic cholangiocarcinomas. Nat Genet. 2013;45(12):1470–1473. | ||
Ross JS, Wang K, Gay L, et al. New routes to targeted therapy of intrahepatic cholangiocarcinomas revealed by next-generation sequencing. The Oncologist. 2014;19(3):235–242. | ||
Kim ST, Jang HL, Lee SJ, et al. Pazopanib, a novel multitargeted kinase inhibitor, shows potent in vitro antitumor activity in gastric cancer cell lines with FGFR2 amplification. Mol Cancer Ther. 2014;13(11):2527–2536. | ||
Nogova L, Sequist LV, Perez Garcia JM, et al. Evaluation of BGJ398, a fibroblast growth factor receptor 1-3 kinase inhibitor, in patients with advanced solid tumors harboring genetic alterations in fibroblast growth factor receptors: Results of a global phase I, dose-escalation and dose-expansion study. J Clin Oncol. 2017;35(2):157–165. | ||
Javle M, Lowery M, Shroff RT, et al. Phase II study of BGJ398 in patients With FGFR-altered advanced cholangiocarcinoma. J Clin Oncol. 2018;36(3):276–282. | ||
Papadopoulos KP, El-Rayes BF, Tolcher AW, et al. A Phase 1 study of ARQ 087, an oral pan-FGFR inhibitor in patients with advanced solid tumours. Br J Cancer. 2017;117(11):1592–1599. | ||
Mazzaferro V, El-Rayes BF, Cotsoglou C. ARQ 087, an oral pan-fibroblast growth factor receptor (FGFR) inhibitor, in patients (pts) with advanced intrahepatic cholangiocarcinoma (iCCA) with FGFR2 genetic aberrations. Am Soc Clin Oncol. 2017;35(15_suppl):4017. | ||
Meric-Bernstam F, Arkenau H, Tran B, et al. O-001Efficacy of TAS-120, an irreversible fibroblast growth factor receptor (FGFR) inhibitor, in cholangiocarcinoma patients with FGFR pathway alterations who were previously treated with chemotherapy and other FGFR inhibitors. Ann Oncol. 2018;29(suppl_5):mdy149. | ||
Borad MJ, Davis SL, Lowery MA, Lihou CF, Abou-Alfa GK, Phase A-AGK. Phase 2, open-label, multicenter study of the efficacy and safety of INCB054828 in patients (pts) with advanced, metastatic, or surgically unresectable cholangiocarcinoma (CCA) with inadequate response to prior therapy. J Clin Oncol. 2017;35(15_suppl):TPS4145. | ||
Sulkowski PL, Corso CD, Robinson ND, et al. 2-Hydroxyglutarate produced by neomorphic IDH mutations suppresses homologous recombination and induces PARP inhibitor sensitivity. Sci Transl Med. 2017;9(375):eaal2463. | ||
Saha SK, Parachoniak CA, Ghanta KS, et al. Mutant IDH inhibits HNF-4α to block hepatocyte differentiation and promote biliary cancer. Nature. 2014;513(7516):110–114. | ||
Borger DR, Tanabe KK, Fan KC, et al. Frequent mutation of isocitrate dehydrogenase (IDH)1 and IDH2 in cholangiocarcinoma identified through broad-based tumor genotyping. Oncologist. 2012;17(1):72–79. | ||
Saha SK, Gordan JD, Kleinstiver BP, et al. Isocitrate dehydrogenase mutations confer dasatinib hypersensitivity and SRC dependence in intrahepatic cholangiocarcinoma. Cancer Discov. 2016;6(7):727–739. | ||
Lampis A, Carotenuto P, Vlachogiannis G, et al. MIR21 drives resistance to heat shock protein 90 inhibition in cholangiocarcinoma. Gastroenterology. 2018;154(4):e1065:1066–1079. | ||
Chan-On W, Nairismägi ML, Ong CK, et al. Exome sequencing identifies distinct mutational patterns in liver fluke-related and non-infection-related bile duct cancers. Nat Genet. 2013;45(12):1474–1478. | ||
Lowery MA, Abou-Alfa GK, Burris HA, et al. Phase I study of AG-120, an IDH1 mutant enzyme inhibitor: results from the cholangiocarcinoma dose escalation and expansion cohorts. J Clin Oncol. 2017;35(15_suppl):4015. | ||
Lowery MA, Abou-Alfa GK, Valle JW, et al. ClarIDHy: A phase 3, multicenter, randomized, double-blind study of AG-120 vs placebo in patients with an advanced cholangiocarcinoma with an IDH1 mutation. J Clin Oncol. 2017;35(15_suppl):TPS4142. | ||
Hao H-X, Jiang X, Cong F. Control of Wnt receptor turnover by R-spondin-ZNRF3/RNF43 signaling Module and its dysregulation in cancer. Cancers. 2016;8(6):54. | ||
Liu J, Pan S, Hsieh MH, et al. Targeting Wnt-driven cancer through the inhibition of Porcupine by LGK974. Proc Natl Acad Sci U S A. 2013;110(50):20224–20229. | ||
Zhang KS, Zhou Q, Wang YF, Liang LJ. Inhibition of Wnt signaling induces cell apoptosis and suppresses cell proliferation in cholangiocarcinoma cells. Oncol Rep. 2013;30(3):1430–1438. | ||
Ong CK, Subimerb C, Pairojkul C, et al. Exome sequencing of liver fluke-associated cholangiocarcinoma. Nat Genet. 2012;44(6):690–693. | ||
Sato N, Yamabuki T, Takano A, et al. Wnt inhibitor Dickkopf-1 as a target for passive cancer immunotherapy. Cancer Res. 2010;70(13):5326–5336. | ||
Liu Y, West SC. Distinct functions of BRCA1 and BRCA2 in double-strand break repair. Breast Cancer Res. 2002;4(1):9. | ||
Breast Cancer Linkage Consortium. Cancer risks in BRCA2 mutation carriers. J Natl Cancer Inst. 1999;91(15):1310–1316. | ||
Ong CK, Subimerb C, Pairojkul C, et al. Exome sequencing of liver fluke-associated cholangiocarcinoma. Nat Genet. 2012;44(6):690–693. | ||
National Cancer Institute; The Cancer Genome Atlas [webpage on the Internet]. 2018 Contains harmonized cancer genomic data sets; 2018. Available from: https://tcga-data.nci.nih.gov/docs/publications/tcga/. Accessed August 25, 2018. | ||
National Cancer Institute; PanCanAltas Publications [webpage on the Internet]. 2018 initiative aims to answer big, overarching questions about cancer by examining the full set of tumors characterized in the robust TCGA dataset; 2018. Available from: https://gdc.cancer.gov/about-data/publications/pancanatlas. Accessed January 3, 2019. | ||
Chan-On W, Nairismägi M-L, Ong CK, et al. Exome sequencing identifies distinct mutational patterns in liver fluke–related and non-infection-related bile duct cancers. Nature Genetics. 2013;45(12):1474–1478. | ||
Li M, Zhang Z, Li X, et al. Whole-exome and targeted gene sequencing of gallbladder carcinoma identifies recurrent mutations in the ErbB pathway. Nat Genet. 2014;46(8):872–876. | ||
Oncology CfM; cBioPortal for Cancer genomic [webpage on the Internet]. Combined study contains data from 6 studies (Cholangiocarcinoma (National University of Singapore, Nat Genet 2012) Cholangiocarcinoma (TCGA, PanCancer Atlas) Cholangiocarcinoma (TCGA, Provisional) Cholangiocarcinoma (National Cancer Centre of Singapore, Nat Genet 2013) Intrahepatic Cholangiocarcinoma (Johns Hopkins University, Nat Genet 2013) Gallbladder Carcinoma (Shanghai, Nat Genet 2014);2018. Available from: http://www.cbioportal.org/study?id=chol_nccs_2013%2Cchol_nus_2012%2Cchol_tcga_pa_can_atlas_2018%2Cchol_tcga%2Cchol_jhu_2013%2Cgbc_shanghai_2014#summary. Accessed August 25, 2018. | ||
Academic, Community Cancer Research United; National Cancer Institute. Liposomal Irinotecan, Fluorouracil, Leucovorin Calcium, and Rucaparib in Treating Patients With Metastatic Pancreatic, Colorectal, Gastroesophageal, or Biliary Cancer; 2021. Available from: https://clinicaltrials.gov/ct2/show/NCT03337087. Accessed February 20, 2019. | ||
A Trial of Niraparib in BAP1 and Other DNA Damage Response (DDR) Deficient Neoplasms (UF-STO-ETI-001). Available from: https://clinicaltrials.gov/ct2/show/NCT03207347. Accessed February 20, 019. | ||
Leyva-Illades D, McMillin M, Quinn M, Demorrow S. Cholangiocarcinoma pathogenesis: role of the tumor microenvironment. Transl Gastrointest Cancer. 2012;1(1):71. | ||
Moeini A, Sia D, Bardeesy N, Mazzaferro V, Llovet JM. Molecular pathogenesis and targeted therapies for intrahepatic cholangiocarcinoma. Clin Cancer Res. 2015:1078–1432. | ||
Kawahara N, Ono M, Taguchi K, et al. Enhanced expression of thrombospondin-1 and hypovascularity in human cholangiocarcinoma. Hepatology. 1998;28(6):1512–1517. | ||
Bunsiripaiboon P, Sornmayura P, Wilasrusmee C, Lertsithichai P. The prognostic significance of microvessel density in intrahepatic cholangiocarcinoma. J Med Assoc Thai. 2010;93(1):66. | ||
Giatromanolaki A, Sivridis E, Koukourakis MI, Polychronidis A, Simopoulos C. Prognostic role of angiogenesis in operable carcinoma of the gallbladder. Am J Clin Oncol. 2002;25(1):38–41. | ||
Giatromanolaki A, Koukourakis MI, Simopoulos C, Polychronidis A, Sivridis E. Vascular endothelial growth factor (VEGF) expression in operable gallbladder carcinomas. Eur J Surg Oncol. 2003;29(10):879–883. | ||
Chen Y, Chen Y, Yu G, Ding H. Lymphangiogenic and angiogenic microvessel density in gallbladder carcinoma. Hepato-gastroenterology. 2011;58(105):20–25. | ||
Tang D, Nagano H, Yamamoto H, et al. Angiogenesis in cholangiocellular carcinoma: Expression of vascular endothelial growth factor, angiopoietin-1/2, thrombospondin-1 and clinicopathological significance. Oncology Reports. 2006;15(3):5251021–5251335. | ||
Yoshikawa D, Ojima H, Iwasaki M, et al. Clinicopathological and prognostic significance of EGFR, VEGF, and HER2 expression in cholangiocarcinoma. Br J Cancer. 2008;98(2):418–425. | ||
Zhu AX, Meyerhardt JA, Blaszkowsky LS, et al. Efficacy and safety of gemcitabine, oxaliplatin, and bevacizumab in advanced biliary-tract cancers and correlation of changes in 18-fluorodeoxyglucose PET with clinical outcome: a phase 2 study. Lancet Oncol. 2010;11(1):481470–542045. | ||
Valle JW, Wasan H, Lopes A, et al. Cediranib or placebo in combination with cisplatin and gemcitabine chemotherapy for patients with advanced biliary tract cancer (ABC-03): a randomised phase 2 trial. The Lancet Oncol. 2015;16(8):967–978. | ||
Sama AR, Denlinger CS, Vogel A, et al. Gemcitabine and cisplatin plus ramucirumab or merestinib or placebo in first-line treatment for advanced or metastatic biliary tract cancer: a double-blind, randomized phase II trial. J Clin Oncol. 2017;35(4_suppl):TPS509. | ||
Yarden Y, Sliwkowski MX. Untangling the ErbB signalling network. Nat Rev Mol Cell Biol. 2001;2(2):127–137. | ||
Pignochino Y, Sarotto I, Peraldo-Neia C, et al. Targeting EGFR/HER2 pathways enhances the antiproliferative effect of gemcitabine in biliary tract and gallbladder carcinomas. BMC Cancer. 2010;10(1):6311471–6312407. | ||
Lee J, Park SH, Chang HM, et al. Gemcitabine and oxaliplatin with or without erlotinib in advanced biliary-tract cancer: a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 2012;13(2):181–188. | ||
Amado RG, Wolf M, Peeters M, et al. Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer. J Clin Oncol. 2008;26(10):1626–1634. | ||
Murali R, Wiesner T, Scolyer RA. Tumours associated with BAP1 mutations. Pathology. 2013;45(2):116–126. | ||
Battaglia A. The importance of multidisciplinary approach in early detection of BAP1 tumor predisposition syndrome: clinical management and risk assessment. Clin Med Insights Oncol. 2014;8:37–47. | ||
Andrici J, Goeppert B, Sioson L, et al. Loss of BAP1 expression occurs frequently in intrahepatic cholangiocarcinoma. Medicine. 2016;95(2):e2491. | ||
Al-Shamsi HO, Anand D, Shroff RT, et al. BRCA-associated protein 1 mutant cholangiocarcinoma: an aggressive disease subtype. J Gastrointest Oncol. 2016;7(4):556–561. | ||
Iwahashi S, Ishibashi H, Utsunomiya T, et al. Effect of histone deacetylase inhibitor in combination with 5-fluorouracil on pancreas cancer and cholangiocarcinoma cell lines. J Med Investig. 2011;58(1,2):106–109. | ||
Asgar MD, Senawong G, Sripa B, Senawong T. Synergistic anticancer effects of cisplatin and histone deacetylase inhibitors (SAHA and TSA) on cholangiocarcinoma cell lines. Int J Oncol. 2016;48(1):409–420. | ||
Sia D, Tovar V, Moeini A, Llovet JM. Intrahepatic cholangiocarcinoma: pathogenesis and rationale for molecular therapies. Oncogene. 2013;32(41):4861–4870. | ||
Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD. Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol. 2002;3(11):991–998. | ||
Goeppert B, Frauenschuh L, Zucknick M, et al. Prognostic impact of tumour-infiltrating immune cells on biliary tract cancer. Br J Cancer. 2013;109(10):2665–2674. | ||
Mcnamara MG, Templeton AJ, Maganti M, et al. Neutrophil/lymphocyte ratio as a prognostic factor in biliary tract cancer. Eur J Cancer. 2014;50(9):1581–1589. | ||
Grenader T, Nash S, Plotkin Y, et al. Derived neutrophil lymphocyte ratio may predict benefit from cisplatin in the advanced biliary cancer: the ABC-02 and BT-22 studies. Ann Oncol. 2015;26(9):1910–1916. | ||
Kaida M, Morita-Hoshi Y, Soeda A, et al. Phase 1 trial of Wilms tumor 1 (WT1) peptide vaccine and gemcitabine combination therapy in patients with advanced pancreatic or biliary tract cancer. J Immunother. 2011;34(1):92–99. | ||
Higashi M, Yonezawa S, Ho JJ, et al. Expression of MUC1 and MUC2 mucin antigens in intrahepatic bile duct tumors: its relationship with a new morphological classification of cholangiocarcinoma. Hepatology. 1999;30(6):1347–1355. | ||
Yamamoto K, Ueno T, Kawaoka T, et al. MUC1 peptide vaccination in patients with advanced pancreas or biliary tract cancer. Anticancer Res. 2005;25(5):3575–3579. | ||
Recchia F, Sica G, Candeloro G. Multicenter phase II study of sequential chemotherapy, radiotherapy, and immunotherapy in locally advanced pancreatic (Pa) and biliary tree (Bt) adenocarcinoma (ADK). J Clin Oncol. 2009;27(15S):3047–3047. | ||
Higuchi R, Yamamoto M, Hatori T, Shimizu K, Imai K, Takasaki K. Intrahepatic cholangiocarcinoma with lymph node metastasis successfully treated by immunotherapy with CD3-activated T cells and dendritic cells after surgery: report of a case. Surg Today. 2006;36(6):559–562. | ||
Tran E, Turcotte S, Gros A, et al. Cancer immunotherapy based on mutation-specific CD4+ T cells in a patient with epithelial cancer. Science. 2014;344(6184):641–645. | ||
Shimizu K, Kotera Y, Aruga A, Takeshita N, Takasaki K, Yamamoto M. Clinical utilization of postoperative dendritic cell vaccine plus activated T-cell transfer in patients with intrahepatic cholangiocarcinoma. J Hepatobiliary Pancreat Sci. 2012;19(2):171–178. | ||
Thompson RH, Gillett MD, Cheville JC, et al. Costimulatory B7-H1 in renal cell carcinoma patients: Indicator of tumor aggressiveness and potential therapeutic target. Proc Natl Acad Sci U S A. 2004;101(49):17174–17179. | ||
Okazaki T, Honjo T. PD-1 and PD-1 ligands: from discovery to clinical application. Int Immunol. 2007;19(7):813–824. | ||
Sabbatino F, Villani V, Yearley JH, et al. PD-L1 and HLA class I antigen expression and clinical course of the disease in intrahepatic cholangiocarcinoma. Clin Cancer Res. 2016;22(2):470–478. | ||
Ye Y, Zhou L, Xie X, Jiang G, Xie H, Zheng S. Interaction of B7-H1 on intrahepatic cholangiocarcinoma cells with PD-1 on tumor-infiltrating T cells as a mechanism of immune evasion. J Surg Oncol. 2009;100(6):500–504. | ||
Bang YJ, Doi T, Braud FD, et al. 525 Safety and efficacy of pembrolizumab (MK-3475) in patients (pts) with advanced biliary tract cancer: Interim results of KEYNOTE-028. Eur J Cancer. 2015;51:S112. | ||
NAEP Release [webpage on the Internet]. FDA approves first cancer treatment for any solid tumor with a specific genetic feature; 2017. FDA approval of cancer treatment(pembrolizumab) for solid tumour with specific genetic feature; 2018. Available from: https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm560167.htm. Accessed January 3, 2019. | ||
FDA News Release [webpage on the Internet]. FDA approval of cancer treatment for solid tumour with specific genetic feature. Available from: https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm560167.htm. Accessed January 3, 2019. | ||
Nowell PC. The clonal evolution of tumor cell populations. Science. 1976;194(4260):23–28. | ||
Fearon ER, Vogelstein B. A genetic model for colorectal tumorigenesis. Cell. 1990;61(5):759–767. | ||
Walter D, Döring C, Feldhahn M, et al. Intratumoral heterogeneity of intrahepatic cholangiocarcinoma. Oncotarget. 2017;8(9):14957. | ||
Mcgranahan N, Furness AJ, Rosenthal R, et al. Clonal neoantigens elicit T cell immunoreactivity and sensitivity to immune checkpoint blockade. Science. 2016;351(6280):1463–1469. | ||
Sirica AE, Campbell DJ, Dumur CI. Cancer-associated fibroblasts in intrahepatic cholangiocarcinoma. Curr Opin Gastroenterol. 2011;27(3):276–284. | ||
Farazi PA, Zeisberg M, Glickman J, Zhang Y, Kalluri R, Depinho RA. Chronic bile duct injury associated with fibrotic matrix microenvironment provokes cholangiocarcinoma in p53-deficient mice. Cancer Res. 2006;66(13):6622–6627. | ||
Olive KP, Jacobetz MA, Davidson CJ, et al. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science. 2009;324(5933):1457–1461. | ||
Matson V, Fessler J, Bao R, et al. The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients. Science. 2018;359(6371):104–108. | ||
Javle M, Bekaii-Saab T, Jain A, et al. Biliary cancer: utility of next-generation sequencing for clinical management. Cancer. 2016;122(24):3838–3847. | ||
Goyal L, Govindan A, Sheth RA, et al. Prognosis and clinicopathologic features of patients with advanced stage isocitrate dehydrogenase (IDH) mutant and IDH wild-type intrahepatic cholangiocarcinoma. Oncologist. 2015;20(9):1019–1027. | ||
Wang P, Dong Q, Zhang C, et al. Mutations in isocitrate dehydrogenase 1 and 2 occur frequently in intrahepatic cholangiocarcinomas and share hypermethylation targets with glioblastomas. Oncogene. 2013;32(25):3091–3100. | ||
Philip PA, Mahoney MR, Allmer C, et al. Phase II study of erlotinib in patients with advanced biliary cancer. J Clin Oncol. 2006;24(19):3069–3074. | ||
Rubovszky G, Láng I, Ganofszky E, et al. Cetuximab, gemcitabine and capecitabine in patients with inoperable biliary tract cancer: a phase 2 study. Eur J Cancer. 2013;49(18):3806–3812. | ||
Chen JS, Hsu C, Chiang NJ, et al. A KRAS mutation status-stratified randomized phase II trial of gemcitabine and oxaliplatin alone or in combination with cetuximab in advanced biliary tract cancer. Ann Oncol. 2015;26(5):943–949. | ||
Malka D, Cervera P, Foulon S, et al. Gemcitabine and oxaliplatin with or without cetuximab in advanced biliary-tract cancer (BINGO): a randomised, open-label, non-comparative phase 2 trial. Lancet Oncol. 2014;15(8):819–828. | ||
Jensen LH, Lindebjerg J, Ploen J, Hansen TF, Jakobsen A. Phase II marker-driven trial of panitumumab and chemotherapy in KRAS wild-type biliary tract cancer. Ann Oncol. 2012;23(9):2341–2346. | ||
Vogel A, Kasper S, Weichert W, et al. Panitumumab in combination with gemcitabine/cisplatin (GemCis) for patients with advanced kRAS WT biliary tract cancer: A randomized phase II trial of the Arbeitsgemeinschaft Internistische Onkologie (AIO). I American Society of Clinical Oncology: 0732-183X; 2015. | ||
Hezel AF, Noel MS, Allen JN, et al. Phase II study of gemcitabine, oxaliplatin in combination with panitumumab in KRAS wild-type unresectable or metastatic biliary tract and gallbladder cancer. Br J Cancer. 2014;111(3):430–436. | ||
Sun Yat-sen University; Jiangsu HengRui Medicine Co. Ltd [homepage on the Internet]. Apatinib as Second Line Therapy in Patients With Advanced Refractory Biliary Tract Cancers; 2017. Available from: https://ClinicalTrials.gov/show/NCT03144856. Accessed January 3, 2019. | ||
Eisai Co. Ltd.; Eisai Inc [homepage on the Internet]. Study of Lenvatinib (E7080) in Unresectable Biliary Tract Cancer Who Failed Gemcitabine-based Combination Chemotherapy; 2015. Available from: https://ClinicalTrials.gov/show/NCT02579616. Accessed January 3, 2019. | ||
Seoul National University Hospital [homepage on the Internet]. MEK162 in Combination With Capecitabine in Advanced Biliary Tract Cancer; 2016. Available from: https://ClinicalTrials.gov/show/NCT02773459. Accessed January 3, 2019. | ||
Aslan Pharmaceuticals [homepage on the Internet]. Varlitinib in Combination With Capecitabine for Advanced or Metastatic Biliary Tract Cancer; 2017. Available from: https://ClinicalTrials.gov/show/NCT03093870. Accessed January 3, 2019. | ||
Aslan Pharmaceuticals [homepage on the Internet]. Study of Varlitinib Plus Capecitabine in Patients With Advanced or Metastatic Biliary Tract Cancer; 2017. Available from: https://ClinicalTrials.gov/show/NCT03129074. Accessed January 3, 2019. | ||
Samsung Medical Center [homepage on the Internet]. Pemetrexed in combination with Erlotinib as a salvage treatment in patients with metastatic biliary tract cancer (BTC) who failed gemcitabine containing chemotherapy: a phase II single arm prospective study; 2017. Available from: https://ClinicalTrials.gov/show/NCT03110484. Accessed January 3, 2019. | ||
Aslan Pharmaceuticals [homepage on the Internet]. Varlitinib in combination with gemcitabine and cisplatin for treatment naïve advanced or metastatic BTC; 2016. Available from: https://ClinicalTrials.gov/show/NCT02992340. | ||
Eli Lilly and Company [homepage on the Internet]. A Study of Ramucirumab (LY3009806) or Merestinib (LY2801653) in Advanced or Metastatic Biliary Tract Cancer; 2016. Available from: https://ClinicalTrials.gov/show/NCT02711553. | ||
Hutchison Medipharma Limited [homepage on the Internet]. Study of Sulfatinib as Second-line Treatment in Patients With Biliary Tract Carcinoma; 2016. Available from: https://ClinicalTrials.gov/show/NCT02966821. Accessed January 3, 2019. | ||
M.D. Anderson Cancer Center; Eli Lilly and Company [homepage on the Internet]. Ramucirumab for advanced pre-treated biliary cancers; 2015. Available from: https://ClinicalTrials.gov/show/NCT02520141. Accessed January 3, 2019. | ||
Shanghai Jiao Tong University School of Medicine; Xinhua Hospital, Shanghai Jiao Tong University School of Medicine; Ruijin Hospital, RenJi Hospital, Eastern Hepatobiliary Surgery Hospital, Huashan Hospital [homepage on the Internet]. Molecularly target therapy with GEMOX in advanced or recurrent extrahepatic cholangiocarcinoma and gallbladder carcinoma; 2016. Available from: https://ClinicalTrials.gov/show/NCT02836847. Accessed January 3, 2019. | ||
Eli Lilly and Company, Merck Sharp & Dohme Corp [homepage on the Internet]. A Study of Ramucirumab Plus Pembrolizumab in Participants With Gastric or GEJ Adenocarcinoma, NSCLC, Transitional Cell Carcinoma of the Urothelium, or Biliary Tract Cancer; 2015. Available from: https://ClinicalTrials.gov/show/NCT02443324. Accessed January 3, 2019. | ||
Institut du Cancer de Montpellier – Val d’Aurelle [homepage on the Internet]. Activity of Regorafenib in combination with chemotherapy in patients with advanced biliary tract cancer; 2014. Available from: https://ClinicalTrials.gov/show/NCT02386397. Accessed January 3, 2019. | ||
University of Nebraska; National Cancer Institute (NCI); Adherex Technologies, Inc [homepage on the Internet]. ADH-1, Gemcitabine hydrochloride and cisplatin in treating patients with metastatic pancreatic or biliary tract cancer that cannot be removed by surgery; 2013. Available from: https://ClinicalTrials.gov/show/NCT01825603. Accessed January 3, 2019. | ||
Asian Pharmaceuticals [homepage on the Internet]. A study of varlitinib in Japanese subjects with advanced or metastatic solid tumours; 2017. Available from: https://ClinicalTrials.gov/show/NCT03082053. Accessed January 3, 2019. | ||
Abramson Cancer Center of the University of Pennsylvania. Study of gemcitabine, irinotecan and panitumumab in patients with advanced and metastatic biliary tract adenocarcinoma; 2009. Available from: https://ClinicalTrials.gov/show/NCT00948935. Accessed January 3, 2019. | ||
Mayo Clinic, National Cancer Institute (NCI) [homepage on the Internet]. Ponatinib hydrochloride in treating patients with advanced biliary cancer with FGFR2 Fusions; 2014. Available from: https://ClinicalTrials.gov/show/NCT02265341. Accessed January 3, 2019. | ||
University of Washington; National Cancer Institute (NCI) [homepage on the Internet]. Afatinib Dimaleate and Capecitabine in Treating Patients With Advanced Refractory Solid Tumors, Pancreatic Cancer or Biliary Cancer; 2015. Available from: https://ClinicalTrials.gov/show/NCT02451553. Accessed January 3, 2019. | ||
RenJi Hospital [homepage on the Internet]. The Effect of Individualized Precision Therapy Programs in Patients With BTC; 2016. Available from: https://ClinicalTrials.gov/show/NCT02943031. Accessed January 3, 2019. | ||
Shen Lin; Peking University [homepage on the Internet]. Anti-HER2 Therapy in Patients of HER2 Positive Metastatic Carcinoma of Digestive System; 2017. Available from: https://ClinicalTrials.gov/show/NCT03185988. Accessed January 3, 2019. | ||
National Cancer Institute (NCI) [homepage on the Internet]. Trametinib or Combination Chemotherapy in Treating Patients With Refractory or Advanced Biliary or Gallbladder Cancer or That Cannot Be Removed by Surgery; 2014. Available from: https://ClinicalTrials.gov/show/NCT02042443. Accessed January 3, 2019. | ||
AstraZeneca [homepage on the Internet]. A study to assess the safety, tolerability and anti-tumour activity of ascending doses of selumetinib in combination with MEDI4736 and selumetinib in combination with MEDI4736 and tremelimumab in patients with advanced solid tumours; 2015. Available from: https://ClinicalTrials.gov/show/NCT02586987. Accessed January 3, 2019. | ||
Senhwa Biosciences, Inc [homepage on the Internet]. Study of CX-4945 in combination with gemcitabine and cisplatin for frontline treatment of cholangiocarcinoma; 2014. Available from: https://ClinicalTrials.gov/show/NCT02128282. Accessed January 3, 2019. | ||
University of Pittsburgh; Bayer [homepage on the Internet]. A phase 2 trial of regorafenib as a single agent in advanced and metastatic biliary tract carcinoma/cholangiocarcinoma patients who have failed first-line chemotherapy; 2014. Available from: https://ClinicalTrials.gov/show/NCT02053376. Accessed January 3, 2019. | ||
Peking Union Medical College Hospital; 3D Medicines [homepage on the Internet]. Precise Treatment in Hepatobiliary Cancers (PTHBC); 2015. Available from: https://ClinicalTrials.gov/show/NCT02715089. Accessed January 3, 2019. | ||
Mazzaferro V, El-Rayes BF, Cotsoglou C. ARQ 087, an oral pan-fibroblast growth factor receptor (FGFR) inhibitor, in patients (pts) with advanced intrahepatic cholangiocarcinoma (iCCA) with FGFR2 genetic aberrations%! ARQ 4087, an oral pan-fibroblast growth factor receptor (FGFR) inhibitor, in patients (pts) with advanced intrahepatic cholangiocarcinoma (iCCA) with FGFR4012 genetic aberrations. J Clin Oncol. 2017;35(15_suppl):4017–4017. | ||
Hellenic Cooperative Oncology Group; GlaxoSmithKline [homepage on the Internet]. Clinical Trial to Investigate the Efficacy of Treatment With Gemcitabine/Pazopanib in Patients With Biliary Tree Cancer; 2013. Available from: https://ClinicalTrials.gov/show/NCT01855724. Accessed January 3, 2019. | ||
H. Lee Moffitt Cancer Center and Research Institute; Institute R Bayer [homepage on the Internet]. Copanlisib (BAY 80-6946) in Combination With Gemcitabine and Cisplatin in Advanced Cholangiocarcinoma; 2016. Available from: https://ClinicalTrials.gov/show/NCT02631590. Accessed January 3, 2019. | ||
Loxo Oncology, Inc [homepage on the Internet]. Study of LOXO-101 (Larotrectinib) in Subjects With NTRK Fusion Positive Solid Tumors (NAVIGATE); 2015. Available from: https://ClinicalTrials.gov/show/NCT02576431. Accessed January 3, 2019. | ||
Eli Lilly and Company [homepage on the Internet]. A Study of Merestinib (LY2801653) in Japanese Participants With Advanced or Metastatic Cancer; 2017. Available from: https://ClinicalTrials.gov/show/NCT03027284. Accessed January 3, 2019. | ||
Wake Forest University Health Sciences; National Cancer Institute (NCI) [homepage on the Internet]. CPI-613 in Treating Patients With Advanced or Metastatic Bile Duct Cancer That Cannot Be Removed By Surgery; 2013. Available from: https://ClinicalTrials.gov/show/NCT01766219. Accessed January 3, 2019. | ||
University of Southern California; National Cancer Institute (NCI); Vasgene Therapeutics, Inc [homepage on the Internet]. Recombinant EphB4-HSA Fusion Protein With Standard Chemotherapy Regimens in Treating Patients With Advanced or Metastatic Solid Tumors; 2015. Available from: https://ClinicalTrials.gov/show/NCT02495896. Accessed January 3, 2019. | ||
Seoul National University Hospital; Merck Serono International SA. Study of TH-302 Monotherapy as Second-line Treatment in Advanced Biliary Tract Cancer; 2016. Available from: https://ichgcp.net/clinical-trials-registry/NCT02433639. Accessed February 20, 2019. | ||
Shen Lin; Peking University. Anti-HER2 Therapy in Patients of HER2 Positive Metastatic Carcinoma of Digestive System; 2021. Available from: https://clinicaltrials.gov/ct2/show/NCT03185988. Accessed February 20, 2019. | ||
H. Lee Moffitt Cancer Center and Research Institute; Research I B. Single Agent Regorafenib in Refractory Advanced Biliary Cancers; 2019. Available from: https://clinicaltrials.gov/ct2/show/NCT02115542. Accessed February 20, 2019. | ||
Aslan Pharmaceuticals. Varlitinib in Combination With Capecitabine for Advanced or Metastatic Biliary Tract Cancer; 2019. Available from: https://clinicaltrials.gov/ct2/show/NCT03093870. Accessed February 20, 2019. | ||
Peking Union Medical College Hospital; 3D Medicines. Precise Treatment in Hepatobiliary Cancers (PTHBC); 2017. | ||
H. Lee Moffitt Cancer Center and Research Institute; Research I B. Copanlisib (BAY 80-6946) in Combination With Gemcitabine and Cisplatin in Advanced Cholangiocarcinoma; 2019. Available from: https://clinicaltrials.gov/ct2/show/NCT02631590. Accessed February 20, 2019. | ||
H. Lee Moffitt Cancer Center and Research Institute; Bristol-Myers Squibb [homepage on the Internet]. Study of Nivolumab in Patients With Advanced Refractory Biliary Tract Cancers; 2016. Available from: https://ClinicalTrials.gov/show/NCT02829918. Accessed January 3, 2019. | ||
Samsung Medical Center [homepage on the Internet]. Study of Pembrolizumab in Metastatic Biliary Tract Cancer as Second-line Treatment After Failing to at Least One Cytotoxic Chemotherapy Regimen: Integration of Genomic Analysis to Identify Predictive Molecular Subtypes; 2017. Available from: https://ClinicalTrials.gov/show/NCT03110328. Accessed January 3, 2019. | ||
Seoul National University Hospital [homepage on the Internet]. Durvalumab(MEDI4736)/Tremelimumab in Combination With Gemcitabine/Cisplatin in Chemotherapy-naïve Biliary Tract Cancer; 2017. Available from: https://ClinicalTrials.gov/show/NCT03046862. Accessed January 3, 2019. | ||
University of Michigan Cancer Center [homepage on the Internet]. Study of Nivolumab in Combination With Gemcitabine/Cisplatin or Ipilimumab for Patients With Advanced Unresectable Biliary Tract Cancer; 2017. Available from: https://ClinicalTrials.gov/show/NCT03101566. Accessed January 3, 2019. | ||
Second Military Medical University [homepage on the Internet]. Immunotherapy Using Precision T Cells Specific to Personalized Neo-antigen for the Treatment of Advanced Malignant Tumor of Biliary Tract; 2015. Available from: https://ClinicalTrials.gov/show/NCT02632019. Accessed January 3, 2019. | ||
AstraZeneca [homepage on the Internet]. A Phase I, Open-Label, Multicentre Study to Evaluate the Safety, Tolerability and Pharmacokinetics of MEDI4736 in Patients With Advanced Solid Tumours; 2013. Available from: https://ClinicalTrials.gov/show/NCT01938612. Accessed January 3, 2019. | ||
Merck Sharp & Dohme Corp. [homepage on the Internet] Study of Pembrolizumab (MK-3475) in Participants With Advanced Solid Tumors (MK-3475-158/KEYNOTE-158); 2015. Available from: https://ClinicalTrials.gov/show/NCT02628067. Accessed January 3, 2019. | ||
National Cancer Institute (NCI); National Institutes of Health Clinical Center [homepage on the Internet]. A Pilot Study of Combined Immune Checkpoint Inhibition in Combination With Ablative Therapies in Subjects With Hepatocellular Carcinoma (HCC) or Biliary Tract Carcinomas (BTC); 2016. Available from: https://ClinicalTrials.gov/show/NCT02821754. Accessed January 3, 2019. | ||
National Cancer Institute (NCI); National Institutes of Health Clinical Center [homepage on the Internet]. Tremelimumab With Chemoembolization or Ablation for Liver Cancer; 2013. Available from: https://ClinicalTrials.gov/show/NCT01853618. Accessed January 3, 2019. | ||
The First Affiliated Hospital with Nanjing Medical University; Miao Y; Abingen Ltd, Nanjing [homepage on the Internet]. T Cell Mediated Adaptive Therapy for Her2-positive Neoplasms of Digestive System; 2016. Available from: https://ClinicalTrials.gov/show/NCT02662348. Accessed January 3, 2019. | ||
National Cancer Institute (NCI); National Institutes of Health Clinical Center. Pembrolizumab, a monoclonal antibody against PD-1, in combination with capecitabine and oxaliplatin (CAPOX) in people with advanced biliary tract carcinoma (BTC); 2019. Availabel from: https://clinicaltrials.gov/ct2/show/NCT03111732. Accessed February 20, 2019. | ||
EORTC. Pembrolizumab in Biliary Tract Cancer. NCT03260712, ed; 2020. Available from: https://clinicaltrials.gov/ct2/show/NCT03260712. Accessed February 20, 2019. | ||
Yonsei University. Phase 1 clinical trial to evaluate the safety of allogeneic NK Cell (“SMT-NK”) cell therapy in advanced biliary tract cancer; 2018. Available from: https://ichgcp.net/clinical-trials-registry/NCT03358849. Accessed February 20, 2019. | ||
Tariq NU, Vogel A, McNamara MG, Valle JW. Biliary tract cancer: implicated immune-mediated pathways and their associated potential targets. Oncol Res Treat. 2018;41(5):298–304. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.