Back to Journals » Infection and Drug Resistance » Volume 13
Bacteremia Caused by the Eggerthella lenta in a Previously Healthy 30-Year-Old Man with Acute Suppurative Appendicitis: A Case Report from China
Authors Jiang J, She B, Zheng R
Received 30 July 2020
Accepted for publication 26 September 2020
Published 19 October 2020 Volume 2020:13 Pages 3695—3698
DOI https://doi.org/10.2147/IDR.S274494
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Jie Jiang,1,2,* Bo She,3,4,* Rui Zheng1,2
1Department of Clinical Laboratory, The First People’s Hospital of Yunnan Province, Kunming, Yunnan, People’s Republic of China; 2Department of Clinical Laboratory, The Affiliated Hospital of Kunming University of Science and Technology, Kunming, Yunnan, People’s Republic of China; 3PET/CT Center, The First People’s Hospital of Yunnan Province, Kunming, Yunnan, People’s Republic of China; 4PET/CT Center, The Affiliated Hospital of Kunming University of Science and Technology, Kunming, Yunnan, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Rui Zheng Tel +86-0871-63638430
Email [email protected]
Abstract: Eggerthella lenta is part of the normal bacterial flora of the intestinal tract, but may cause life-threatening infections. E. lenta has been isolated from blood, abscesses, wounds, skin ulcers, and intra-abdominal infections. However, due to historical difficulties with laboratory identification by conventional biochemical methods, some cases of E. lenta infection have previously gone undiagnosed, especially in China where E. lenta infections are very rare. Recently, matrix-assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF MS), a new type of technology, has made it possible to identify E. lenta. We report a case of postoperative infection caused by E. lenta which was treated successfully using meropenem.
Keywords: Eggerthella lenta, MALDI-TOF, intra-abdominal abscess, bloodstream infection, anaerobic bacteria
Introduction
Eggerthella lenta was first identified in 1935 by Arnold Eggerth.1 It is sometimes vancomycin resistant and may represent the origin of vancomycin resistance.2 This genus of gram-positive, anaerobic bacilli belongs to the Eggerthellaceae family, which includes several genera of bacteria commonly found in the healthy human digestive tract.3 As a gut microbiota, E. lenta plays a key role in pharmacology. For example, it harbors a cytochrome-encoding operon which responsible for digoxin inactivation.4
Various infections caused by E. lenta have been reported including pelvic abscess and bloodstream infection.5 E. lenta bloodstream infections are increasingly recognized, often caused by intra-abdominal infections, and associated with high mortality.6 Previous study shows that E. lenta was susceptible to amoxicillin-clavulanate, cefoxitin, metronidazole, piperacillin-tazobactam, ertapenem, meropenem and partly susceptible to clindamycin, moxifloxacin, and penicillin.6,7 There is another report showed that chloramphenicol and vancomycin were the most reliable antibiotic treatment options for E. lenta.5
Anaerobic bacteria are difficult to culture and identify in clinical laboratories, so are often not recovered from infected sites. Recently, E. lenta has become recognized as an important cause of anaerobic bloodstream infections, which are associated with high mortality.6 Here, we report a case of bacteremia and acute suppurative appendicitis in a previously healthy 30-year-old man, which was caused by E. lenta. E. lenta was identified using matrix-assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF MS), an increasingly used new technology that has improved the detection of E. lenta in clinical laboratories.
Case Report
A 30-year-old man was transferred to our hospital from a local hospital in Wuding County in Yunnan Province, China, where he had initially presented with fever and had been diagnosed with acute purulent appendicitis. An appendectomy had been performed 7 days previously. On admission, he was febrile and had rigors. He had a temperature of 39.5°C, a heart rate of 99 beats/min, and a blood pressure of 108/78 mmHg. The rest of his physical examination was normal. On Days 2–4 after admission, his temperature ranged from 36.0°C to 40.0°C.
Laboratory investigations revealed an elevated white blood cell count of 17.70×109 cells/L (neutrophils: 86.4%) and an elevated procalcitonin (PCT) level of 25.91 ng/mL. Multiple blood samples were taken before antibiotics were initiated, and were processed for aerobic and anaerobic culture. A contrast-enhanced computerized tomography scan of his abdomen, performed on admission, revealed an ileocecal abscess (Figure 1), the size of abscess was 2.3 cmX2.9 cm. Two sets of blood cultures were incubated in the BacT/Alert 3D 240 blood culture system (bioMérieux, Marcy l’Étoile, France) for generally up to 5 days. Before the results of his blood culture were obtained, he was treated empirically with intravenous moxifloxacin on account of his recent appendectomy.
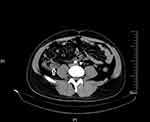 |
Figure 1 Abdominal contrast enhancement CT scan shows an abscess lesion of ileocecus (white arrow). |
On Day 3, gram-positive bacilli were identified in two sets of anaerobic blood cultures. These were inoculated onto Columbia blood agar base medium (Autobio, China) and incubated in 35◦C 5% CO2 and 35◦C anaerobic conditions, respectively. On Day 4, an isolate was identified as E. lenta using MALDI-TOF MS (bioMérieux, Marcy l’Étoile, France), E. coli ATCC 8739TM and K. aerogenes ATCC13048 used as Calibration Strain and Quality control Strain. Once the result became available, his treatment was changed to intravenous meropenem at a dose of 1 g every 8 h. Although percutaneous or surgical drainage is an option for treating intra-abdominal abscesses,8 our patient responded promptly to meropenem therapy, and his fever, PCT level, and leukocytosis resolved without surgical intervention. No microorganisms were identified in the follow-up blood cultures from Day 5. On Day 10, abdominal color Doppler ultrasonography was normal. After a further 6-day course of intravenous meropenem, he recovered and was discharged from hospital.
Discussion
Because of the fastidious nature of E. lenta, and historical difficulties with laboratory identification by conventional biochemical methods, some cases of E. lenta infection have previously gone undiagnosed, especially in China where cases of E. lenta infection are very rare. Recently, a new technology, matrix-assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF MS), has made it possible to accurately identify pathogens that are difficult to identify in clinical laboratories, including E. lenta.7 In this case, MALDI-TOF was a practical way to identify E. lenta at the species level.
The risk factors for infection caused by E. lenta include impaired immune function, recent chemotherapy, a gastrointestinal malignancy, diabetes mellitus, and chronic kidney disease, absence of fever at presentation and need for ICU stay are risk factors for 30-day mortality.7,9,10 Bacteremia due to this organism is always clinically significant, given its high mortality rate, and warrants a prompt search for a source. E. lenta can cause bacteremia due to abdominal or soft tissue sources. Patients are frequently bedridden and empiric piperacillin-tazobactam monotherapy is associated with a high mortality rate.6,10 E. lenta infections are associated with gastrointestinal infections, particularly perforated appendicitis.6 Our patient was 30 years old and was otherwise healthy, his peri-appendiceal abscess is the most likely source of his bacteremia and appendectomy is a risk factor for bacteremia.
Empiric therapy plays a crucial role in the treatment of anaerobic infections. A previous study showed that E. lenta isolates have variable susceptibility to moxifloxacin,7 However, after a 3-day course of moxifloxacin, our patient still had a fever, elevated PCT level (13.38 ng/mL), and leukocytosis (10.05×109 cells/L). Taking his culture results into account, his treatment was changed to meropenem and he had a good clinical response. There is currently a lack of consensus regarding the best antibiotic therapy for E. lenta. The good response to meropenem in our patient may have significance for the empirical use of meropenem in the treatment of bacteremia caused by E. lenta.
Conclusion
This case illustrates that E. lenta is an opportunistic pathogen and can cause of hospital-acquired infection such as appendicitis, and be a source of bloodstream infection. In this case, advances in clinical microbiology made it easier to identify E. lenta. The number of E. lenta infections diagnosed in China is likely to increase as a result of new technology, and greater attention should be paid to this infection.
Abbreviation
MALDI-TOF MS, matrix-assisted laser desorption ionization-time of flight mass spectrometry.
Data Sharing Statement
The datasets used and/or analyzed in this study are available from the corresponding author on reasonable request.
Ethics Statement
The clinical isolates in this study were specifically isolated for this research. Ethical approval was obtained from the Institutional Ethics Committee (The First People’s Hospital of Yunnan Province, Kunming, Yunnan, China). Written informed consent was received from each patient before sample collection that the study participants gave consent to publish.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
This research was funded by Yunnan Science and Technology Commission from Yunnan provincial Science and Technology Department and Kunming Medical University, grant number 2018FE001(−115) and Health commission of Yunnan province talent program, grant number H-2017027.
Disclosure
Jie Jiang and Bo She are co-first authors for this study. The authors declare no conflict of interest.
References
1. Eggerth AH. The gram-positive non-spore-bearing anaerobic bacilli of human feces. J Bacteriol. 1935;30(3):277–299. doi:10.1128/JB.30.3.277-299.1935
2. Stinear TP, Olden DC, Johnson PD, Davies JK, Grayson ML. Enterococcal vanB resistance locus in anaerobic bacteria in human faeces. Lancet. 2001;357(9259):855–856. doi:10.1016/S0140-6736(00)04206-9
3. Almeida A, Mitchell A, Boland M, et al. A new genomic blueprint of the human gut microbiota. Nature. 2019;568(7753):499–504. doi:10.1038/s41586-019-0965-1
4. Haiser HJ, Seim KL, Balskus EP, Turnbaugh PJ. Mechanistic insight into digoxin inactivation by Eggerthella lenta augments our understanding of its pharmacokinetics. Gut Microbes. 2014;5(2):233–238. doi:10.4161/gmic.27915
5. Bo J, Wang S, Bi Y, Ma S, Wang M, Du Z. Eggerthella lenta bloodstream infections: two cases and review of the literature. Future Microbiol. 2020;15(11):981–985. doi:10.2217/fmb-2019-0338
6. Ugarte-Torres A, Gillrie MR, Griener TP, Church DL. Eggerthella lenta bloodstream infections are associated with increased mortality following empiric piperacillin-tazobactam (TZP) monotherapy: a population-based cohort study. Clin Infect Dis. 2018;67(2):221–228. doi:10.1093/cid/ciy057
7. Gardiner BJ, Tai AY, Kotsanas D, et al. Clinical and microbiological characteristics of Eggerthella lenta bacteremia. J Clin Microbiol. 2015;53(2):626–635. doi:10.1128/JCM.02926-14
8. Thota VR, Dacha S, Natarajan A, Nerad J. Eggerthella lenta bacteremia in a Crohn’s disease patient after ileocecal resection. Future Microbiol. 2011;6(5):595–597. doi:10.2217/fmb.11.31
9. Wong D, Aoki F, Rubinstein E. Bacteremia caused by Eggerthella lenta in an elderly man with a gastrointestinal malignancy: a case report. Can J Infect Dis Med Microbiol. 2014;25(5):e85–86. doi:10.1155/2014/802481
10. Venugopal AA, Szpunar S, Johnson LB. Risk and prognostic factors among patients with bacteremia due to Eggerthella lenta. Anaerobe. 2012;18(4):475–478. doi:10.1016/j.anaerobe.2012.05.005
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
