Back to Journals » International Journal of Nephrology and Renovascular Disease » Volume 9
Axitinib in the treatment of renal cell carcinoma: patient selection and perspectives
Received 23 November 2015
Accepted for publication 29 January 2016
Published 29 March 2016 Volume 2016:9 Pages 65—72
DOI https://doi.org/10.2147/IJNRD.S83874
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Pravin Singhal
Vivek Narayan, Naomi Balzer Haas
Division of Hematology/Medical Oncology, Hospital of the University of Pennsylvania, Philadelphia, PA, USA
Background: Axitinib is a next-generation, selective tyrosine kinase inhibitor targeting the vascular endothelial growth factor receptors. It is approved for the treatment of metastatic renal cell carcinoma (mRCC) based on a demonstrated progression-free survival advantage over sorafenib in the second-line treatment setting. However, given the variety of available targeted therapies for mRCC, appropriate patient selection for the available therapies remains a significant clinical challenge.
Purpose: This review summarizes the available evidence on the clinical, toxicity, and pharmacologic considerations for determining appropriate patient selection for axitinib therapy. In addition, it describes recent data on the use of predictive biomarkers to guide clinical management. This paper consists of material obtained via PubMed and Medline literature searches through October 2015.
Conclusion: Axitinib has a well-established role in the management of mRCC. Consistent clinical efficacy has been demonstrated across prognostic risk groups and prior therapeutic exposures. Although axitinib is generally well tolerated, appropriate toxicity management is critical to maximizing drug delivery and optimizing treatment outcomes. Although incident hypertension has been associated with improved clinical outcomes on axitinib, there are currently no validated clinical or genetic predictive biomarkers to guide patient selection.
Keywords: axitinib, vascular endothelial growth factor receptor, renal cell carcinoma, predictive marker
Introduction
Kidney cancer is a common and lethal disease, with approximately 214,000 new cases and 140,000 disease-related deaths reported annually worldwide.1 Renal cell carcinoma (RCC) comprises greater than 90% of kidney cancer diagnoses, and metastatic renal cell carcinoma (mRCC) is resistant to conventional cytotoxic chemotherapies.2 For many years, cytokine-based therapies remained the standard approach for the management of mRCC, with only a small number of patients achieving a significant treatment response. However, since the early 2000s, a growing understanding of mRCC biology has led to the development of numerous therapies targeting the vascular endothelial growth factor (VEGF) and the mammalian target of rapamycin (mTOR) molecular pathways. Since 2005, sorafenib, sunitinib, temsirolimus, pazopanib, bevacizumab in combination with interferon, everolimus, and axitinib have been approved by the US Food and Drug Administration for the treatment of mRCC.3–9 The availability of such therapies has resulted in a doubling of median overall survival (OS) to approximately 2 years, and VEGF- and mTOR-directed therapies represent the current care standard for mRCC management.3–6
Although the variety of available targeted therapies has been a welcome boon for medical oncologists, there remains a lack of validated biomarkers to guide therapeutic decision making for the treatment of patients with mRCC. Indeed, current guidelines list all of the approved agents as potential options for first-line and/or subsequent therapy.10,11 In an effort to provide evidence-based guidance for optimal treatment selection, multiple studies have evaluated different sequential treatment strategies.12–16 However, in the absence of clear predictive markers to inform patient selection, therapeutic decision making continues to rely heavily on the available clinical trial data, the drug’s mechanism of action and expected toxicity profile, and the individual physician’s experience. In a recent retrospective review of treatment practices at US community oncology practices, the VEGF-mTOR-VEGF therapeutic sequence was most commonly utilized, but considerable practice variation was observed.17
Axitinib (AG-013736; Inlyta; Pfizer Inc, New York, NY, USA) is an orally administered, selective tyrosine kinase inhibitor (TKI) of the vascular endothelial growth factor receptor-1 (VEGFR-1), VEGFR-2, and VEGFR-3.18 As a second-generation VEGFR inhibitor, axitinib offers increased potency and specificity for the VEGFR when compared to earlier VEGFR inhibitors that demonstrated multikinase activity with off-target effects.19,20 In the pivotal Phase III randomized clinical trial (AXIS), axitinib demonstrated an improved progression-free survival (PFS) when compared to sorafenib in the second-line treatment setting.6 This was the first Phase III trial to utilize an active comparator and solidified the role of axitinib as a second-line treatment option for mRCC. When evaluated in the first-line setting, axitinib demonstrated clinical activity and safety, but no significant PFS benefit over sorafenib in a Phase III randomized comparison.21 Axitinib has been included among the therapeutic options for first-line mRCC treatment in consensus guidelines.10 However, given axitinib’s relatively recent approval, data from its use have not been included in most studies examining optimal drug-sequencing strategies.14,15,17 This review will focus on the important clinical and toxicity considerations for patient selection with the use of axitinib for the treatment of mRCC. In addition, it will review the evidence regarding potential predictive biomarkers under recent investigation.
Clinical considerations for axitinib patient selection
Prior drug exposure
Given the multiple available therapeutic agents and the considerable variation in tumor response, the nature of prior drug exposure is an important consideration for the selection of subsequent mRCC therapies.
Axitinib has demonstrated consistent clinical efficacy in patients with prior exposure to cytokine therapy. In an initial Phase II study of 52 patients with mRCC and disease progression following cytokine-based therapy, treatment with axitinib resulted in an objective response rate of 44.2% (95% confidence interval [CI], 30.5%–58.7%) and a median time to progression of 15.7 months (95% CI, 8.4–23.4 months).22 Long-term follow-up of this patient cohort demonstrated a 5-year OS rate of 20.6%.23 In the randomized Phase III AXIS trial, patients were prospectively stratified based on prior first-line therapy, and 251 patients (34.7%) had progressed after prior cytokine therapy.6 In this patient subgroup, axitinib demonstrated a significantly prolonged PFS over sorafenib (12.1 vs 6.5 months, hazard ratio [HR], 0.464, P<0.0001).6
When considering consecutive anti-VEGF TKIs as mRCC therapy, multiple retrospective studies have demonstrated that sequential anti-VEGF therapy remains a viable treatment approach with continued antitumor activity.24–26 Indeed, the Phase III INTORSECT trial, which randomized patients progressing on sunitinib to either the mTOR inhibitor temsirolimus or the VEGFR inhibitor sorafenib, revealed no significant difference in PFS and a median OS benefit favoring sorafenib.27 Of note, a purer trial design would have ideally compared everolimus and axitinib, as these are the mTOR and VEGFR inhibitors, respectively, that are supported by randomized Phase III evidence in the second-line setting.5,6 Nevertheless, these findings support the use of continued VEGF targeting despite disease progression on initial VEGF inhibition and clearly demonstrate the addiction of mRCC to VEGF signaling.
In the Phase III AXIS trial, 448 patients (62%) had progressed following first-line VEGF-directed therapy (sunitinib or bevacizumab).6 In the patient subgroup with prior sunitinib exposure, axitinib demonstrated a significant PFS improvement compared with sorafenib (4.8 vs 3.4 months, HR, 0.741, P=0.0107). Notably, the PFS on axitinib was considerably numerically longer in the cytokine-exposed subgroup relative to the sunitinib-exposed subgroup (12.1 vs 4.8 months), which likely reflects a lack of resistance to VEGF-targeted therapy among cytokine-exposed patients and some diminishing clinical benefit to serial VEGFR inhibition.6,28 Axitinib has also demonstrated clinical activity in the third-line setting in a sorafenib-refractory patient cohort.29 Finally, although small retrospective studies have supported the use of a sunitinib “rechallenge” in selected mRCC patients, limited data are available on the similar use of axitinib reexposure.30
In terms of the disease response to prior therapy, a prior response to sunitinib therapy was not associated with longer PFS or OS on second-line axitinib in a post hoc analysis of the AXIS trial data.31 This finding was consistent with other reported retrospective analyses.32 These findings may indicate that the use of second-line axitinib can be considered despite a lack of objective response to a first-line VEGFR-directed agent. However, a shorter time to progression on first-line sunitinib (≤25th percentile) was associated with a shorter OS on subsequent axitinib therapy, likely reflecting aggressive underlying disease biology and cross-resistance to VEGF-directed therapies.28 In fact, the results of some retrospective studies suggest the selection of an alternative therapeutic target (ie, mTOR) in patients demonstrating a primary refractory response to anti-VEGF therapy, although there are limited data on axitinib in this setting and this patient subgroup generally exhibits a poor prognosis regardless of subsequent therapy.24,33
mRCC prognostic group
Clinically defined prognostic risk groups have been established to guide counseling and decision making for patients with mRCC. The Memorial Sloan-Kettering Cancer Center (MSKCC) risk classification was originally developed and validated in cytokine-treated patients.34,35 This classification scheme includes diminished Karnofsky performance status, lactate dehydrogenase level >1.5 times the upper limit of normal (ULN), anemia, corrected serum calcium level > ULN, and time from initial diagnosis to initiation of therapy <1 year as adverse prognostic features.34,35 More recently, a second prognostic model (Heng risk group) was developed and validated in a patient cohort treated with anti-VEGF-targeted therapies.36 In addition to selected MSKCC risk criteria, this model included a neutrophil count > ULN and a platelet count > ULN as adverse prognostic features.36 Both models classify patients into good-, intermediate-, and poor-risk groups and are commonly used in clinical practice to predict the expected clinical course independent of specific treatment intervention.37
In the AXIS trial, subgroup analyses of PFS were performed based on both MSKCC and Heng risk classifications.6 These findings demonstrated at least a trend toward a PFS advantage favoring axitinib in all risk groups (Table 1). Therefore, although the AXIS trial validated the clear prognostic utility of the MSKCC risk criteria in the trial cohort, these clinical risk groupings have yet to provide predictive information for guiding appropriate treatment selection.28
  | Table 1 Axitinib clinical efficacy by prior therapy and prognostic group |
Toxicity and pharmacologic considerations for axitinib patient selection
Toxicity considerations
An important consideration for treatment selection in mRCC is the potential toxicities of therapy, especially within the context of patient comorbidities and prior treatment-related adverse events. In the AXIS trial, axitinib was generally well tolerated, with 14 patients (4%) discontinuing therapy due to treatment-related adverse events.6 This treatment discontinuation was most commonly due to fatigue (N=4) or transient ischemic attack (N=3). At least one axitinib dose reduction was required in 31% of patients, compared with 52% of patients treated with sorafenib.6 Hypertension, nausea, hypothyroidism, and dysphonia were relatively more common with axitinib when compared to sorafenib.28 The most common grade ≥3 adverse events (≥3% incidence) reported in prospective trials of axitinib for the treatment of mRCC, are listed in Table 2.
In a meta-analysis of 1,908 patients from ten Phase II and Phase III clinical trials evaluating axitinib (including the AXIS trial), the overall incidence of all-grade and high-grade hypertension in cancer patients was reported as 40.1% (95% CI, 30.9%–50.2%) and 13.3% (95% CI, 6.7%–24%), respectively.38 Notably, axitinib use was associated with a significantly elevated risk of all-grade (relative risk =3.00, 95% CI, 1.29–6.97, P=0.011) and high-grade hypertension (relative risk =1.71, 95% CI, 1.21–2.43, P=0.003). Furthermore, the risk of hypertension with axitinib appeared to be significantly higher than the risk reported in systematic reviews of other approved VEGFR TKIs (except for the risk of all-grade hypertension observed with the use of pazopanib, which had similar effect size).38 Similarly, a meta-analysis of Phase II and Phase III trials of several oral multikinase inhibitors (including four trials with axitinib) reported a significantly increased risk of all-grade hypertension with the use of these agents.39 However, no significantly increased risk was observed with axitinib relative to the other included agents.39
As treatment-induced hypertension may lead to serious end-organ effects and/or the interruption of effective cancer therapy, blood pressure should be monitored carefully, with the prompt initiation of antihypertensive medications as necessary. Although the exact mechanisms of VEGF-inhibitor-related hypertension remain unclear, it is hypothesized that an increase in systemic vascular resistance results from decreased nitric oxide production, vascular rarefaction, and the secretion of neurohormonal vasoconstrictive factors (including endothelin 1).40,41 Sufficient controlled clinical studies are currently lacking to inform the appropriate selection of antihypertensive agents for VEGF-inhibitor-induced hypertension. However, limited reports have indicated benefit from dihydropyridine calcium channel blockers (given their direct vasodilatory effects) and angiotensin-converting enzyme inhibitors (given their potential effects on vascular rarefaction and angiogenesis).42,43 In general, it is recommended to avoid nitrates and loop diuretics,42 but individual patient circumstances and comorbidities should be considered when selecting an antihypertensive regimen.
In addition, as many patients with mRCC exhibit impaired renal function as a result of prior nephrectomy or other medical comorbidities, the dosing and safety of mRCC therapies in patients with chronic kidney disease is an important consideration. A recently reported population pharmacokinetic study demonstrated that axitinib pharmacokinetics were similar regardless of baseline renal function and that the standard dose of axitinib was well tolerated in mRCC patients with impaired renal function.44 Although proteinuria is a less common toxicity of axitinib, it may result in treatment discontinuation. In a Japanese Phase II study of 64 patients on axitinib 5 mg twice daily, baseline proteinuria (≥1+) was associated with the development of significant proteinuria on treatment (≥2 g/24 h).45
In terms of thyroid dysfunction, a Japanese study observed that mRCC patients treated with axitinib developed incident thyroid dysfunction more commonly and within a shorter time frame when compared to those treated with sorafenib or sunitinib.46 However, these findings were limited by the small retrospective nature of the study. Nevertheless, these findings together highlight the important toxicity considerations and clinical monitoring necessary while using axitinib.
Pharmacologic considerations
Drug dose adjustment is an important consideration in the use of axitinib. In early pharmacokinetic studies, a linear correlation was found between the axitinib dose and both the maximal plasma concentration and the area under plasma concentration–time curve.47 While age over 60 years and Japanese ethnicity were associated with decreased drug clearance, the magnitude of effect was not sufficiently large to warrant predetermined dose adjustment.48 These dose-proportional pharmacokinetics of axitinib indicate that increased plasma exposure will result from axitinib dose uptitration.49 Furthermore, preliminary studies suggested that an increased plasma axitinib concentration was associated with prolonged PFS and OS in mRCC patients.23,48 It was, therefore, hypothesized that dose escalation from the standard starting dose of 5 mg twice daily may lead to improved clinical outcomes.
A randomized, double-blind Phase II clinical trial evaluated the safety and efficacy of axitinib dose escalation in the first-line treatment of mRCC.49 After a 4-week run-in period to identify patients with acceptable axitinib tolerance at the standard starting dose, subjects were randomized to masked, stepwise dose titration up to 10 mg twice daily or to placebo titration. Patients with axitinib dose titration experienced a higher objective response rate than those with placebo titration (54% vs 34%, one-sided P=0.019). However, a comparison of the median PFS between the axitinib-titration and the placebo-titration groups was not statistically different (14.5 vs 15.7 months, HR, 0.85, 95% CI, 0.54–1.35, P=0.24).49 The reason for this discrepancy between the improved response rate and the absence of a PFS advantage is currently unclear. However, it is possible that increased toxicity in certain patients with the initial dose titration (and resultant dose reduction) may account for these discrepant findings (as this would allow for an initial objective response, but subsequent mitigation of a potential PFS benefit).49
The findings from this dose-titration study emphasize that axitinib drug exposure (via dosing and/or schedule) is a very important determinant of clinical outcome. Therefore, the common flat-dosing schedules used for mRCC-targeted agents are likely inadequate for optimizing clinical outcome in individual patients. However, recent follow-up from the randomized dose-titration study indicates that pharmacokinetic measurements alone cannot be used to successfully individualize axitinib dosing and that a sufficient dose-optimization model is still needed.50
These findings also highlight the importance of adequate toxicity management to maximize drug delivery and hopefully optimize treatment outcomes. Fortunately, the toxicities associated with axitinib are generally predictable and manageable. In addition, consensus toxicity monitoring and management recommendations have been outlined, with an emphasis on the proactive management of hypertension, diarrhea, and fatigue.51,52 Needless to say, the potential benefits of increased dose delivery must be balanced against the potential toxicities in the individual patient. While clearly an important determinant of efficacy, axitinib pharmacokinetics are likely only one of many factors (including drug-, tumor-, and patient-related factors) contributing to observed individual clinical outcomes.
Exploration of predictive biomarkers
Clinical biomarkers
To date, there is a lack of validated clinical biomarkers to guide therapy selection for mRCC. However, this remains an area of active investigation.
Systemic hypertension has been evaluated as a potential predictive marker for improved outcome with VEGF-directed therapies. Hypertension has been identified as a common marker of clinical efficacy in patients treated with VEGF-directed therapies, including sunitinib and axitinib.53,54 In a subgroup analysis of the Phase III AXIS trial, the development of a systolic blood pressure ≥140 mmHg or a diastolic blood pressure ≥90 mmHg during treatment were independent predictors of OS in multivariable analyses.28 The appropriate medical management of hypertension does not appear to mitigate the potential beneficial effects observed with VEGF-targeted therapies.53 However, it has remained unclear whether the observed association between treatment-induced hypertension and clinical outcome reflects drug exposure levels or tumor/host factors resulting in enhanced response to VEGFR inhibition.37 In addition, dose-titration studies have indicated that blood pressure cannot yet be used to successfully individualize axitinib dosing to optimize outcome.50
While other organ effects such as hypothyroidism, cytopenias, and interstitial pneumonitis have also been investigated as potential predictive clinical biomarkers for multiple mRCC therapies, there is not yet an evidence to support their use with axitinib, given its relatively recent addition to the available mRCC therapies.
Genetic and molecular biomarkers
Axitinib and other VEGF-directed therapies target the tumor microenvironment and nonmalignant endothelial cells, as opposed to having a direct cytotoxic effect on tumor cells.55 As such, there has been interest in pharmacogenetic evaluations of germline single-nucleotide polymorphisms (SNPs) as reliable predictive biomarkers for clinical efficacy and/or adverse drug reactions.
As previously discussed, axitinib plasma exposure has been associated with improved clinical efficacy, and axitinib is known to display variable pharmacokinetics between individuals.48,56 However, a meta-analysis of eleven studies evaluating SNPs in genes encoding major enzymes involved in axitinib metabolism did not demonstrate any significant association with axitinib plasma exposure.56 In addition, an analysis of blood samples from 305 patients from the AXIS trial evaluated the association between germline SNPs and clinical outcomes.57 Fifteen germline SNPs in VEGF-A, VEGFR-1, VEGFR-2, and hypoxia-inducible factor-1α were included. The investigators found that no SNP was associated with significant incident hypertension. While VEGF-A rs699947 and rs833061 were associated with prolonged OS in axitinib-treated patients, this association was not significant on multivariable analyses.57
Recent advances in genomic sequencing have led to the comprehensive molecular characterization of RCC.58 This has allowed for the identification of recurrently mutated genes in RCC and the evaluation of their prognostic and predictive values. For example, recent interest has focused on the development of predictive tumor-based molecular signatures based on recurrently mutated RCC genes (including PBRM1, BAP1, SETD2, and KDM5C). While PBRM1 and BAP1 expression have been preliminarily associated with prognostic outcome, their predictive utility remains unclear.59 However, compelling data regarding the predictive utility of KDM5C mutation have recently been presented. In a prospective cohort of patients treated with sunitinib or everolimus, patients with mutated KDM5C demonstrated an improved response to sunitinib when compared to patients with the wild-type gene.60 A similar effect was not observed in the everolimus-treated cohort, therefore suggesting that KDM5C may represent a predictive efficacy biomarker for VEGF-directed therapies, such as sunitinib and axitinib.60 These compelling preliminary data are planned for validation in separate cohorts with a variety of VEGF-directed therapies. In addition, the optimal use of axitinib remains an area of active investigation, with ongoing clinical studies evaluating the therapy in a variety of combination strategies and disease settings (Table 3).
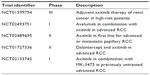  | Table 3 Selected ongoing axitinib clinical trials |
Conclusion
Axitinib is an important component of the mRCC treatment strategy. Indeed, axitinib was the first therapy to demonstrate a PFS benefit in the second-line setting over an active comparator. However, given the number and variety of available agents for the treatment of mRCC, predictive markers will be necessary to maximize benefit and minimize treatment-related toxicity. Furthermore, with recent Phase III evidence in support of additional effective mRCC therapies, including the programmed death-1 checkpoint inhibitor nivolumab61 and the multikinase inhibitor cabozantinib,62 the therapeutic options are rapidly increasing. Therefore, the discovery of new information from genetically informed prospective clinical trials to guide appropriate treatment selection for individual patients will remain a research imperative.
Disclosure
The authors report no conflicts of interest in this work.
References
GLOBOCAN 2012 v1.0. Cancer incidence and mortality worldwide: IARC CancerBase no 11 [updated 2012]. Available from: http://globocan.iarc.fr. Accessed January 30, 2016. | |
Znaor A, Lortet-Tieulent J, Laversanne M. International variation and trend in renal cell carcinoma incidence and mortality. Eur Urol. 2015;67:519–530. | |
Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356(2):115–124. | |
Sternberg CN, Davis ID, Mardiak J, et al. Pazopanib in locally advanced or metastatic renal cell carcinoma: results of a randomized phase III trial. J Clin Oncol. 2010;28(6):1061–1068. | |
Motzer RJ, Escudier B, Oudard S, et al. Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomised, placebo-controlled phase III trial. Lancet. 2008;372(9637):449–456. | |
Rini BI, Escudier B, Tomczak P, et al. Comparative effectiveness of axitinib versus sorafenib in advanced renal cell carcinoma (AXIS): a randomised phase 3 trial. Lancet. 2011;378(9807):1931–1939. | |
Hudes G, Carducci M, Tomczak P, et al. Temsirolimus, interferon α, or both for advanced renal-cell carcinoma. N Engl J Med. 2007;356(22):2271–2281. | |
Escudier B, Eisen T, Stadler WM, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med. 2007;356(2):125–134. | |
Escudier B, Pluzanska A, Koralewski P, et al. Bevacizumab plus interferon α-2a for treatment of metastatic renal cell carcinoma: a randomised, double-blind phase III trial. Lancet. 2007;370(9605):2103–2111. | |
National Comprehensive Cancer Network. NCCN Guidelines: Kidney Cancer. Version I. Fort Washington, PA: NCCN; 2016. | |
Escudier B, Porta C, Schmidinger M, et al. Renal cell carcinoma: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2014;25:49–56. | |
Stephan Michel M, Walter V, Maria de S, et al. SWITCH: a randomized sequential open-label study to evaluate efficacy and safety of sorafenib (SO)/sunitinib (SU) versus SU/SO in the treatment of metastatic renal cell cancer (mRCC). J Clin Oncol. 2014;32(Suppl 4):abstr 393. | |
Motzer R, Barrios CH, Kim TM, et al. Record-3: phase II randomized trial comparing sequential first-line everolimus (EVE) and second-line sunitinib (SUN) versus first-line SUN and second-line EVE in patients with metastatic renal cell carcinoma (mRCC). J Clin Oncol. 2013;31(Suppl):abstr 4504. | |
Heng DY, Signorovitch J, Swallow E, et al. Comparative effectiveness of second-line targeted therapies for metastatic renal cell carcinoma: a systematic review and meta-analysis of real-world observational. PLoS One. 2014;9(12):e114264. | |
Alimohamed N, Lee J, Srinivas S, et al. A population-based overview of sequences of targeted therapy in metastatic renal cell carcinoma. Clin Genitourin Cancer. 2014;12(4):e127–e131. | |
Escudier B, Gore M. Sequencing therapy in metastatic renal cell cancer. Semin Oncol. 2013;40(4):465–471. | |
Jonasch E, Signorovitch JE, Lin PL, et al. Treatment patterns in metastatic renal cell carcinoma: a retrospective review of medical records from US community oncology practices. Curr Med Res Opin. 2014;30(10):2041–2050. | |
Hu-Lowe DD, Zou HY, Grazzini ML, et al. Nonclinical antiangiogenesis and antitumor activities of axitinib (AG-013736), an oral, potent, and selective inhibitor of vascular endothelial growth factor receptor tyrosine kinases 1, 2, 3. Clin Cancer Res. 2008;14(22):7272–7283. | |
Sonpavde G, Hutson TE, Rini BI. Axitinib for renal cell carcinoma. Expert Opin Investig Drugs. 2008;17(5):741–748. | |
Akaza H, Fukuyama T. Axitinib for the treatment of advanced renal cell carcinoma. Expert Opin Pharmacother. 2014;15(2):283–297. | |
Hutson TE, Lesovoy V, Al-Shukri S, et al. Axitinib versus sorafenib as first-line therapy in patients with metastatic renal-cell carcinoma: a randomised open-label phase 3 trial. Lancet Oncol. 2013;14(13):1287–1294. | |
Rixe O, Bukowski RM, Michaelson MD, et al. Axitinib treatment in patients with cytokine-refractory metastatic renal-cell cancer: a phase II study. Lancet Oncol. 2007;8(11):975–984. | |
Rini BI, De La Motte Rouge T, Harzstark AL, et al. Five-year survival in patients with cytokine-refractory metastatic renal cell carcinoma treated with axitinib. Clin Genitourin Cancer. 2013;11(2):107–114. | |
Li J, Yang C, Wang S, et al. First-line treatment result influence second-line regimen selection in targeted therapy for metastatic renal cell carcinoma. Anticancer Res. 2014;34(10):5643–5647. | |
Vickers MM, Choueiri TK, Rogers M, et al. Clinical outcome in metastatic renal cell carcinoma patients after failure of initial vascular endothelial growth factor-targeted therapy. Urology. 2010;76(2):430–434. | |
Escudier B, Szczylik C, Porta C, Gore M. Treatment selection in metastatic renal cell carcinoma: expert consensus. Nat Rev Clin Oncol. 2012;9(6):327–337. | |
Hutson TE, Escudier B, Esteban E, et al. Randomized phase III trial of temsirolimus versus sorafenib as second-line therapy after sunitinib in patients with metastatic renal cell carcinoma. J Clin Oncol. 2014;32(8):760–767. | |
Motzer RJ, Escudier B, Tomczak P, et al. Axitinib versus sorafenib as second-line treatment for advanced renal cell carcinoma: overall survival analysis and updated results from a randomised phase 3 trial. Lancet Oncol. 2013;14(6):552–562. | |
Rini BI, Wilding G, Hudes G, et al. Phase II study of axitinib in sorafenib-refractory metastatic renal cell carcinoma. J Clin Oncol. 2009;27(27):4462–4468. | |
Zama IN, Hutson TE, Elson P, et al. Sunitinib rechallenge in metastatic renal cell carcinoma patients. Cancer. 2010;116(23):5400–5406. | |
Escudier B, Michaelson MD, Motzer RJ, et al. Axitinib versus sorafenib in advanced renal cell carcinoma: subanalyses by prior therapy from a randomised phase III trial. Br J Cancer. 2014;110(12):2821–2828. | |
Al-Marrawi MY, Rini BI, Harshman LC, et al. The association of clinical outcome to first-line VEGF-targeted therapy with clinical outcome to second-line VEGF-targeted therapy in metastatic renal cell carcinoma patients. Target Oncol. 2013;8(3):203–209. | |
Heng DY, MacKenzie MJ, Vaishampayan UN, et al. Primary anti-vascular endothelial growth factor (VEGF)-refractory metastatic renal cell carcinoma: clinical characteristics, risk factors, and subsequent therapy. Ann Oncol. 2012;23(6):1549–1555. | |
Motzer RJ, Mazumdar M, Bacik J, Berg W, Amsterdam A, Ferrara J. Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma. J Clin Oncol. 1999;17(8):2530–2540. | |
Motzer RJ, Bacik J, Murphy BA, Russo P, Mazumdar M. Interferon-alfa as a comparative treatment for clinical trials of new therapies against advanced renal cell carcinoma. J Clin Oncol. 2002;20(1):289–296. | |
Heng DY, Xie W, Regan MM, et al. Prognostic factors for overall survival in patients with metastatic renal cell carcinoma treated with vascular endothelial growth factor-targeted agents: results from a large, multicenter study. J Clin Oncol. 2009;27(34):5794–5799. | |
Michaelson MD, Stadler WM. Predictive markers in advanced renal cell carcinoma. Semin Oncol. 2013;40(4):459–464. | |
Qi W, He A, Shen Z, Yao Y. Incidence and risk of hypertension with a novel multi-targeted kinase inhibitor axitinib in cancer patients: a systematic review and meta-analysis. Br J Clin Pharmacol. 2013;76(3):348–357. | |
Abdel-Rahman O, Fouad M. Risk of cardiovascular toxicities in patients with solid tumors treated with sunitinib, axitinib, cediranib, or regorafenib: an updated systematic review and comparative meta-analysis. Crit Rev Oncol Hematol. 2014;92(3):194–207. | |
Hamnvik OR, Choueiri TK, Turchin A, et al. Clinical risk factors for the development of hypertension in patients treated with inhibitors of the VEGF signaling pathway. Cancer. 2015;121(2):311–319. | |
Izzedine H, Ederhy S, Goldwasser F, et al. Management of hypertension in angiogenesis inhibitor-treated patients. Ann Oncol. 2009;20(5):807–815. | |
Copur MS, Obermiller A. An algorithm for the management of hypertension in the setting of vascular endothelial growth factor signaling inhibition. Clin Colorectal Cancer. 2011;10(3):151–156. | |
Katsi V, Ioannis Z, Manolakou S, et al. Anti-VEGF anticancer drugs: mind the hypertension. Recent Pat Cardiovasc Drug Discov. 2014;9:63–72. | |
Chen Y, Rini BI, Motzer RJ, et al. Effect of renal impairment on the pharmacokinetics and safety of axitinib. Target Oncol. Epub September 24, 2015. | |
Tomita Y, Uemura H, Fujimoto H, et al. Key predictive factors of axitinib (AG-013736)-induced proteinuria and efficacy: a phase II study in Japanese patients with cytokine-refractory metastatic renal cell carcinoma. Eur J Cancer. 2011;47(17):2592–2602. | |
Daimon M, Kato T, Kaino W, et al. Thyroid dysfunction in patients treated with tyrosine kinase inhibitors, sunitinib, sorafenib and axitinib, for metastatic renal cell carcinoma. Jpn J Clin Oncol. 2012;42(8):742–747. | |
Rugo HS, Herbst RS, Liu G, et al. Phase I trial of the oral antiangiogenesis agent AG-013736 in patients with advanced solid tumors: pharmacokinetic and clinical results. J Clin Oncol. 2005;23(24):5474–5483. | |
Rini BI, Garrett M, Poland B, et al. Axitinib in metastatic renal cell carcinoma: results of a pharmacokinetic and pharmacodynamic analysis. J Clin Pharmacol. 2013;53(5):491–504. | |
Rini BI, Melichar B, Ueda T, et al. Axitinib with or without dose titration for first-line metastatic renal-cell carcinoma: a randomised double-blind phase 2 trial. Lancet Oncol. 2013;14(12):1233–1242. | |
Rini BI, Melichar B, Fishman M, et al. Axitinib dose titration: analyses of exposure, blood pressure and clinical response from a randomized phase II study in metastatic renal cell carcinoma. Ann Oncol. 2015;26(7):1372–1377. | |
Larkin J, Fishman M, Wood L, et al. Axitinib for the treatment of metastatic renal cell carcinoma: recommendations for therapy management to optimize outcomes. Am J Clin Oncol. 2014;37(4):397–403. | |
Bracarda S, Castellano D, Procopio G, et al. Axitinib safety in metastatic renal cell carcinoma: suggestions for daily clinical practice based on case studies. Expert Opin Drug Saf. 2014;13(4):497–510. | |
Rini BI, Schiller JH, Fruehauf JP, et al. Diastolic blood pressure as a biomarker of axitinib efficacy in solid tumors. Clin Cancer Res. 2011;17(11):3841–3849. | |
Rini BI, Cohen DP, Lu DR, et al. Hypertension as a biomarker of efficacy in patients with metastatic renal cell carcinoma treated with sunitinib. J Natl Cancer Inst. 2011;103(9):763–773. | |
Rodríguez-Antona C, García-Donas J. Constitutional genetic variants as predictors of antiangiogenic therapy outcome in renal cell carcinoma. Pharmacogenomics. 2012;13(14):1621–1633. | |
Brennan M, Williams JA, Chen Y, Tortorici M, Pithavala Y, Liu YC. Meta-analysis of contribution of genetic polymorphisms in drug-metabolizing enzymes or transporters to axitinib pharmacokinetics. Eur J Clin Pharmacol. 2012;68(5):645–655. | |
Escudier B, Rini BI, Motzer RJ, et al. Genotype correlations with blood pressure and efficacy from a randomized phase III trial of second-line axitinib versus sorafenib in metastatic renal cell carcinoma. Clin Genitourin Cancer. 2015;13(4):328–337.e3. | |
Cancer Genome Atlas Research Network. Comprehensive molecular characterization of clear cell renal cell carcinoma. Nature. 2013;499(7456):43–49. | |
Joseph R, Kapur P, Serie D, et al. Clear cell renal cell carcinoma subtypes identified by BAP1 and PBRM1 expression. J Urol. 2016; 195(1):180–187. | |
Hsieh J, Chen D, Wang P, Chen Y, Redzematovic A, Marker M. Identification of efficacy biomarkers in a large metastatic renal cell carcinoma (mRCC) cohort through next generation sequencing (NGS): results from RECORD-3. J Clin Oncol. 2015;33(Suppl):abstr 4509. | |
Motzer RJ, Escudier B, McDermott DF, et al. Nivolumab versus everolimus in advanced renal-cell carcinoma. N Engl J Med. 2015; 373(19):1803–1813. | |
Choueiri T, Escudier B, Powles T, et al. Cabozantinib versus everolimus in advanced renal-cell carcinoma. N Engl J Med. 2015; 373(19):1814–1823. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

