Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Asymmetry of Subthalamic Neuronal Firing Rate and Oscillatory Characteristics in Parkinson’s Disease
Authors Zhao XM, Zhuang P, Li Y, Zhang Y, Li J, Wang Y, Li J
Received 3 September 2019
Accepted for publication 11 January 2020
Published 28 January 2020 Volume 2020:16 Pages 313—323
DOI https://doi.org/10.2147/NDT.S229513
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jun Chen
This paper has been retracted.
Xue-Min Zhao, 1 Ping Zhuang, 1– 3 Yong-jie Li, 1 Yu-qing Zhang, 1 Jian-yu Li, 1 Yun-peng Wang, 1 Ji-ping Li 1
1Beijing Institute of Functional Neurosurgery, Xuanwu Hospital, Capital Medical University, Beijing, People’s Republic of China; 2Center of Parkinson’s Disease, Beijing Institute for Brain Disorders, Beijing, People’s Republic of China; 3Key Laboratory of Neurodegenerative Diseases (Capital Medical University), Ministry of Education, Beijing, People’s Republic of China
Correspondence: Ping Zhuang
Beijing Institute of Functional Neurosurgery, Xuanwu Hospital, Capital Medical University, No. 45 Changchun Street, Xicheng District, Beijing 100053, People’s Republic of China
Tel +86 10 83198887
Fax +86 10 83199238
Email [email protected]
Purpose: The aim of this study was to compare the neuronal firing rate and oscillatory activity of the subthalamic nucleus (STN) between the more affected (MA) and the less affected (LA) hemispheres in Parkinson’s disease (PD).
Patients and Methods: We recorded and analyzed the intra-operative microelectrode recordings (MER) from the STN of 24 PD subjects. Lateralized Unified Parkinson’s Disease Rating Scale (UPDRS) III sub-scores (item 20– 26) were calculated. The STN corresponding to the MA side was designated as the MA STN while the other side as the LA STN. Single unit characteristics including interspike intervals were identified and spectral analyses were assessed. Further, the mean spontaneous firing rate (MSFR) of neurons was calculated. The correlations between clinical symptoms and neuronal activity were analyzed.
Results: The firing rate in the MA and LA sides were 43.18 ± 0.74 Hz and 36.94 ± 1.32 Hz, respectively, with an increase of 16.9% in the MA group. The number of neurons that oscillated in the Tremor-Frequency Band (TFB), β-Frequency Band (βFB), and the non-oscillatory cells in the MA group were 43, 115, and 62 versus 78, 68, and 54 in the LA group, respectively. The proportions of the three types of neurons were different between both groups. The firing rate of the STN neurons and the UPDRS III sub-scores were positively correlated. Additionally, we observed a positive correlation between the percentage of βFB oscillatory neurons and bradykinesia score.
Conclusion: The firing rate of the STN in the MA hemisphere is higher than in the LA side, following disease progression and there seems to be an increase in firing rate. The βFB oscillatory neurons are at a larger proportion in the MA group while there were larger percentage of TFB oscillatory cells in the LA group. The proportion of βFB oscillatory neurons is selectively correlated with the severity of bradykinesia.
Keywords: Parkinson’s disease, subthalamic nucleus, intra-operative microelectrode recordings, tremor-frequency band, β-frequency band, βFB
Introduction
Parkinson’s disease (PD) is a progressive degenerative disorder of the human nervous system, characterized by impairments in movement initiation (akinesia) and reductions in the amplitude and velocity of voluntary movements (bradykinesia), accompanied by muscular rigidity and tremor at rest.1,2 PD tends to start on one side of the body and gradually spread to the opposite side, presenting asymmetrical symptoms in the course of the disease.3
The pathogenesis of PD is not yet clear. Damage in the subnuclei of the substantia nigra pars compacta (SNc) is considered the most important hallmark of PD, leading to dopamine deficiency in striatum. According to the hypothesis of the basal ganglia “rate model” proposed by Albin and Delong,4,5 the loss of dopaminergic input to the striatum causes cascade of functional modification to the basal ganglia circuitry. In brief, the main output nucleus, the globus pallidus internal (GPi), gets rid of the inhibitive pulse through the direct pathway and receives greater excitatory inputs through the indirect pathway. The overall result is the decreased thalamic activity and then the cortical excitability is attenuated. Histopathological studies have shown that 50%–70% of substantia nigra neurons had undergone apoptosis by the time motor symptoms appear.6,7
The asymmetric loss of dopaminergic neurons in bilateral substantia nigra may be one of the causes of asymmetrical symptoms on both sides.3,6 Over half of PD patients show obvious differences in UPDRS III scores of the left and right limbs.8 Studies using fluorine-18-labeled fluorodopa (18F-dopa) PET and single-photon emission CT (SPECT) imply that the amount of tracer uptake decreased obviously in the striatum corresponding to the more affected (MA) side.9,10
Dopaminergic depletion may also result in changes in neuronal firing activities of the basal ganglia. The symptoms of PD are associated with increased frequency, abnormal firing pattern, and excessive synchronization of oscillatory activities in the basal ganglia, including the STN.11 Direct evidence was absent to illustrate that the firing rate of the STN neurons in PD patients maybe higher than that of the normal human. Increased neuronal firing rate in the STN has been found in animal models of PD and supports the “rate model” of the basal ganglia.5,12 Increased firing rate in the group of patients with advanced symptoms than the group of patients with early symptoms has been previously shown.13 There is still a lack of studies comparing the firing rate in both hemispheres of individual subjects. Therefore, the first hypothesis of our study was that the firing rate on the MA side might be higher than the LA side.
In recent days, the changes in firing patterns and oscillations are considered to play a crucial role in the process of PD.12,14,15 When it comes to oscillatory activity, the β band oscillatory neurons are the most well studied, and based on the evidence, βFB activity has been associated with bradykinesia and rigidity.16 TFB associated oscillation has been correlated with limb tremor.17 As the disease progresses, tremor tends to be less severe while rigidity and bradykinesia may become the dominant problem.18–20 Intra-operative local field potentials (LFPs) from the subthalamic nucleus (STN) indicated that the α/β band oscillations were greater in the MA hemisphere.21 However, it is still unknown whether the firing patterns are different between the MA side and the LA side on the single-unit activity level in human subjects with PD. Therefore, the second hypothesis is βFB oscillatory activities increase with disease progression whereas TFB oscillation is attenuated. In the current study, with the advantage of microelectrode recordings during stereotactic operation, we aimed to shed light on these puzzling doubts. These findings may have important implications for neural signals that can be used to drive adaptive deep brain stimulation (aDBS) in PD.
Materials and Methods
Human Subjects and Clinical Assessment
Twenty-four patients (16 males, eight females, 58.3 ± 1.82 years) with idiopathic PD who were undergoing stereotactic surgery for implantation of bilateral STN DBS electrodes were included in this study. They had a mean duration of PD for 10.5 ± 1.19 years at the time of surgery. The total levodopa equivalent daily dosage (LEDD) was 780 ± 75.5 mg. Motor severity was defined with the aid of part III – United Parkinson Disease Rating Scale (UPDRS). All patients were assessed before and after an acute levodopa (L-dopa) challenge 1 week prior to the surgical date. Lateralized UPDRS III sub-scores (item 20–26: rest tremor, postural/action tremor, rigidity, and limb bradykinesia) were also calculated. The bradykinesia score included scores for finger tapping, hand movement, pronation-supination, and leg agility. The MA side was determined for each subject as the side with higher UPDRS III sub-scores in the medication off state. Subsequently, the other side was considered the LA side. If equal sub-scores appear on both sides, then the side on which the symptom started is considered as belonging to the MA group. The STN corresponding to the MA side was defined as the MA STN while the other side as the LA STN. The sub-scores on tremor, rigidity, and bradykinesia on both sides were also calculated. During surgery, the patients were awake and were not on dopaminergic medications for >12 h from the last oral dose of anti-parkinsonian medications. Demographic details of the patients are given in Table 1. Detailed UPDRS III sub-scores are shown in Table 2. This study was carried out in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of Xuanwu Hospital, Capital Medical University. All patients provided written informed consent.
 |
Table 1 Clinical Information of Patients |
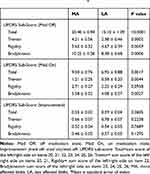 |
Table 2 UPDRS Sub-Score Summarya |
Surgical Procedure
Detailed surgical procedure has been presented in our previous study.17,22 In brief, all patients underwent pre-operative contrast enhanced volumetric T1 and T2-weighted magnetic resonance imaging (MRI) of the brain. On the day of surgery, Cosman–Roberts–Wells (CRW) frame (Radionics, Burlington, MA, USA) were applied to the skull under local anesthesia, and a full head computed tomography (CT) scan was obtained using 1-mm slice thickness. CT and MRI images were fused using Medtronic StealthStation FrameLink software. The targets of the STN were calculated based on a stereotactic atlas. The coordinates of the target point of the STN were 12 mm lateral, 1 mm posterior, and 4 mm inferior to the mid-commissural point. The recording tracks were made in a trajectory with a 12° angle lateral from the vertical line and approximately 60° from the intercommissural line. A full-head CT scan was obtained using 1-mm slice thickness 1 day after surgery and fused with MRI images for target planning before surgery. Lead location in the STN was clearly indicated and the trajectory length of microelectrode recording was confirmed indirectly.
Microelectrode Recording
As described in previous studies,16,23 microelectrode recordings were performed during targeting using the Microguide system Pro (AlphaOmega Engineering, Nazareth, Israel) with patients at rest as a general step in the targeting procedure. Briefly, a tungsten microelectrode with tip size 10–20 μm and resistances from 0.3 to 0.5 MΩ at 1000 Hz (Alpha Omega Engineering, Nazareth, Israel) was used. Recordings started 10 mm upward the target and advanced at 0.5 mm intervals. The dorsal border of STN was identified by the increased amplitude of background activity and higher frequency neuronal discharge, reflecting the summed activity of large densely packed neurons in the nucleus. When the electrode passed through the ventral margin of the STN, the background noise decreased suddenly. Three channels of electromyograms (EMGs) were simultaneously recorded from the extensor carpi radialis (ECR), flexor carpi radialis (FCR), and the tibialis anterior (TA) muscles on the contralateral limbs using surface electrodes. All signals recorded from the microelectrode were amplified (×20,000) and filtered (with a bandpass of 200 Hz–10 kHz). The sample rate was 12,019 Hz. EMG signals were amplified (×5000), filtered with a bandpass of 20–1,000 Hz, and sampled at 3 kHz.
Signal Processing
As previously described,22 all neuronal and EMG data were analyzed using Spike 2 (Cambridge Electronic Design, Cambridge, UK). Only spikes having a signal-to-noise ratio greater than 2:1 were used. For further analysis, we only chose recordings that were ≥10 s in duration and were free of artifacts and not obtained during periods with voluntary movements. Further, single-unit analysis was used. The neuronal waveform and shape were first identified by visual assessment. Action potentials were picked up using amplitude and waveform criteria through spike sorting function in Spike 2 for further analysis. The inter-spike interval (ISI) was obtained from the spike train and the mean spontaneous firing rate (MSFR) and coefficient of variation (CV) of ISI were calculated. The root-mean-square (RMS) was used to present the amplitude of the neurons. Oscillatory characteristics were evaluated using the power spectrum density (PSD) of the units. The raw data were rectified, DC removed with a time constant of 0.5 s, and down sampled to 3 kHz. Frequencies between 48 Hz and 52 Hz known as the white noise were removed before further analysis. The PSD was processed with the Welch method in a Hanning window, the FFT size was set as 4096, yielding a frequency resolution of 0.7336 Hz. Significant oscillation frequencies were defined as those that exceed a threshold of five standard deviations above the mean power in the 30–70 Hz band.24 The frequency with the maximal power of all frequencies that exceeded the threshold was regarded as the main oscillation frequency. If no frequency exceeded the threshold, then the neuron was considered non-oscillatory cell. In our study, the tremor frequency band (TFB) neurons oscillated at 3–7 Hz while β frequency band (βFB) neurons oscillated at 8–35 Hz.
Statistical Analysis
Results were statistically analyzed in GraphPad Prism (version 7.0, GraphPad Software Inc., San Diego, CA) and Origin (version 8.5, OriginLab Corporation, Northampton, MA, USA) software and presented as mean ± SEM. For the paired data of the two groups, the normally distributed was compared with Student’s t-tests while Wilcoxon matched-pairs signed-rank test was used if the distribution was not normal. When normal distribution is not followed, Wilcoxon matched-pairs signed-rank test is performed. For unpaired data, the Mann–Whitney U-test (rank-sum test) was performed. Chi-square analysis was used to compare the percentage of the three types of neurons between the two groups. Fisher’s exact test was performed to compare the dominant hand distribution in the two groups. Two-way ANOVA and post hoc Bonferroni t-test were used to compare the parameters of the three types of neurons in the two groups. Linear regression and correlation analysis were applied for the relevance between clinical symptoms and neuronal activities. For all statistical tests, a p-value <0.05 was considered statistically significant.
Results
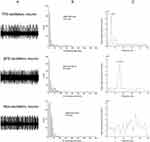
A total of 420 cells were analyzed in 24 PD patients. Figure 1 illustrates representative examples of most STN neurons which consist of either TFB oscillatory neuron, βFB oscillatory neuron, or non-oscillatory neurons. Of these neurons, 220 neurons were identified from 24 sides of STN in the MA group and 200 neurons from the LA group. The length of trajectory was 5.08 ± 0.18 mm versus 4.81 ± 0.16 mm in the MA group vs LA group, when the length of trajectory along the STN were compared. No significant difference in the trajectory length in both groups was found (p>0.05). It is important to note that the neurons studied were all considered from the corresponding locations on both hemispheres. We also plot the neurons within the STN, as shown in Figure 2. We found that the majority of oscillatory neurons were localized in the dorsal two-thirds of the STN. All but four patients included in our study were right-handed. There were 15 dominant hands belonging to the MA group while 9 belonging to the LA group. The number of dominant hands in the MA group and the LA group were similar (p>0.05).
Mean Spontaneous Firing Rate in the MA Group Was Higher Than in the LA Group
From the 220 cells in the MA group, we observed that the MSFR was 43.18 ± 0.74 Hz, which was significantly higher than 36.94 ± 1.32 Hz in 200 neurons in the LA group (p < 0.0001). We observed about 16.9% increase in MSFR in the MA group than in the LA group. Furthermore, the mean firing rate of TFB cells was 41.85 ± 1.48 Hz in MA group vs 36.23 ± 1.06 Hz in LA group (p<0.01), mean firing rate of βFB oscillatory neurons was 43.88 ± 0.8422 Hz in MA group vs 37.81 ± 1.22 Hz in LA group (p<0.0001), and non-oscillatory neurons had a mean firing rate of 42.55 ± 1.18 Hz in MA group vs 36.21 ± 1.42 Hz in LA group (p<0.001). In our further analysis, we found no difference in the firing rate among these three types of neurons. Figure 3 demonstrates the mean firing rate and the distribution of the firing rate in the MA and LA groups and firing rate of the TFB oscillatory neurons, βFB oscillatory neurons, and non-oscillatory neurons separately.
The Percentages of Three Types of Neurons are Different in Both Groups
Of the 220 neurons checked in the MA group, 43 (19.55%) were TFB neurons at tremor frequency 4.9 ± 0.2 Hz; 115 (52.27%) were βFB oscillatory neurons with periodic bursting at frequency of 19.4 ± 0.9 Hz; 62 (28.18%) were non-oscillatory neurons with irregular activity lacking oscillation frequency exceeding the threshold. Of the 200 neurons identified from the LA group, 78 (39%) neurons were TFB oscillatory neurons at a frequency of 4.6 ± 0.2 Hz; 68 (34%) were βFB oscillatory neurons with frequency of 19.1 ± 0.9 Hz; 54 (27%) were non-oscillatory neurons. Chi-square analysis indicated that there was a marginal difference of proportion among the TFB cells, βFB cells, and non-oscillatory cells in the MA and LA group (p < 0.0001, Figure 4). There was a significant difference of proportion among the TFB oscillatory neurons (19.55% vs 39.00%, p<0.001) and proportion of βFB oscillatory neurons (52.27% vs 34.00%; p < 0.001) between MA and LA group. No significant difference of proportion was observed among the non-oscillatory neurons in between the MA and LA sides (27.00% vs 28.18%; p > 0.05). The percentages of the three types of neurons was observed to be different between two groups. There were more TFB cells in the LA group while there were more βFB cells in the MA group, whereas non-oscillatory cells were almost the same on both sides.
No Significant Difference Was Observed on Tremor Score Percentage
The lateralized UPDRS III sub-score in the MA group was 20.40 ± 0.94 and was significantly higher than in the LA group (15.10 ± 1.09) (p < 0.0001). The sub-score of tremor (4.21 ± 0.56 vs 5.63 ± 0.32), rigidity (10.25 ± 0.58 vs 2.38 ± 0.46), and bradykinesia (4.67 ± 0.39, vs 8.38 ± 0.68) was calculated for the MA group vs LA group in the medication off state (p < 0.01, p < 0.01, p < 0.01), respectively. The percentage of tremor score based on the lateralized UPDRS III sub-scores were 16.5 ± 3.1% and 20.0 ± 2.5% in the MA and LA group. No significant difference was found (p > 0.05). Hence, it is evident that there was no difference between the symptom types observed in the two groups.
Selective Correlations Existed in Clinical Symptoms and Neuronal Activities
We examined the correlation between parkinsonian motor symptoms (tremor, rigidity and bradykinesia), as measured by the preoperative hemi-body UPDRS III sub-scores, and the neuronal activity parameters of STN. The UPDRS III sub-scores and the firing rates were found to be significantly correlated (r=0.5261, p<0.001, Figure 5A), especially between the firing frequency and the bradykinesia scores (r=0.4544, p<0.01, Figure 5B), and the higher firing rate following higher sub-scores. While tremor scores were not significantly correlated, the proportions of βFB oscillatory neurons were positively correlated with bradykinesia scores (r=0.2945, p<0.05, Figure 5C), when measured between the percentages of three types neurons of STN and their sub-scores, respectively.
Discussion
From the microelectrode recordings in the cohort of our study, we came to two main conclusions. First, the firing rate in the MA hemisphere was higher than in the LA sides. Second, the βFB oscillations were evident in the MA hemispheres corresponding to the MA sides, whereas TFB oscillatory activities were more evident on the LA sides. These findings were important to provide supplementary evidence to the classical basal ganglia rate model and supported the pattern and oscillation model. Hence, it is helpful for us to better understand the pathophysiological mechanism of PD. To elaborate, with our first set of evidence it was clear that the firing rate in the MA group (43.18 ± 0.74 Hz) was higher than in the LA group (36.94 ± 1.32 Hz) with an increase of 16.9%, when we analyzed the correlation of severity of symptoms and firing rate, we found a significant correlation between them, and bradykinesia was also positively correlated with the firing rate, which indicated that there was an increase in firing rate with disease progression. Rarely, studies have focused on understanding the firing rate of both sides in one individual. Previously, when a study compared the firing rate in the group with an early stage of PD vs group with an advanced stage, divided according to the H-Y stage criteria, significantly higher frequency was found in the advance group than in the early group (28.7 vs 36.3 Hz).13 Though no data on a healthy human was obtained, the relative frequency may also explain the phenomenon of symptom asymmetry. Furthermore, the significant correlation between clinical motor defects and neuronal firing can also indicate disease progression. Previously, many studies on primates have referred to the firing rate of STN. Studies in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)–treated monkeys exhibited an increase in mean firing rate from 19 ±10 Hz to 26 ±15 Hz after given a sufficient dose of MPTP, which was used to create parkinsonian symptoms.12 Studied on PD patients also found changes in firing rate with the status of the disease. The frequency usually occurred in ranges from 25 to 75 Hz in the off-medication state25–28 and it decreased after L-dopa administration or DBS.29,30
Another study observed that there was more neuronal degeneration in the SNc of the MA side.10 According to the basic model of PD in basal ganglia, degeneration leads to disinhibition of the STN, thus generating the cardinal signs of PD.5,31 Theoretically, the firing rate may be greater in the MA hemisphere. Our results can somewhat be a supplement to this model that the firing rate may be different on the basis of disease severity. Almost every patient in our study showed the tendency of higher firing rate on the initially affected STN. Only one patient with a total disease history over 20 years showed very similar frequencies on both hemispheres. We also analyzed the firing rate of the three different types of neurons separately and observed that the TFB cells, βFB cells, and non-oscillatory cells showed higher frequency in the MA group than in the LA group. But there was no significant difference among the three type of neurons. However, it is unclear if there was a difference in the discharge rate of the three cells. Levy et al27 reported there was no significant difference in the firing rates of tremor cells and cells with high-frequency oscillatory activity. While Rodriguez et al32 reported that the mean firing rate for tremor cells was lower than other neurons. Moran et al24 found the firing rate of non-oscillatory neurons was lower than the oscillating groups.
Second, oscillation characteristics are very important criteria that need to be discussed. The most important consequence of our study was that we found significant differences in the oscillatory activities in the MA and LA groups. The MA group had a higher percentage of βFB cells (52.27% vs 34.00%) with a lower percentage of TFB cells (19.55% vs 39.00%). Another crucial hypothesis about the basal ganglia was that the change in firing pattern of neurons was considered to be a key pathophysiological mechanism in PD. To date, the βFB activity has been the most well-studied frequency band. The βFB activity has been obvious in the STN of PD patients in the medication off state and encountered a significant decrease in the medication on state. It has also been observed to be decreased when receiving high-frequency stimulation delivered by DBS or performing active movements.33–36 Some studies have also shown that there was some deterioration in bradykinesia and rigidity if low-frequency stimulation at 10 and 20 Hz is given to the PD patients.37–39
Our results not only verify the high proportion of βFB cells in PD patients but also confirm that this proportion increases with disease progression and the βFB cells are especially correlated with the severity of bradykinesia. Strong correlations of β oscillations with rigidity and bradykinesia at rest have been shown.40,41 In our previous study, βFB has also been directly correlated with rigidity.16 In other studies, a direct relationship between basal ganglia oscillations and tremor has been found.22,42 The TFB oscillations are tremor coherent in more than 50% of STN neurons.43 These tremor frequency oscillatory neurons in the basal ganglia have been frequently correlated with limb tremor rhythm.17 We observed that the percentage of sub-score on rigidity or tremor was similar in both groups. This could potentially mean that these symptoms, such as rigidity or tremor, may not be the contributing factor for the difference in the βFB activity changes. The decreased proportion of TFB cells may be explained by the dominance of βFB cells while no significant change in non-oscillatory cells was observed. Though coherence exists between tremor frequency band neurons and limb muscular activities, the proportion of tremor cells did not increase significantly. A potential explanation was that the tremor sub-score did not clearly worsen with time.20 In addition, the cerebello-thalamo-cortical network seems to be more important with parkinsonian tremor.17 Chen et al also found that the beta band oscillations were associated with motor impairment and they found a strong negative correlation between their complexities, and still no such correlation was observed with tremor.41 Sharott et al compared more detailed activity parameters of STN neurons and obtained results similar to our observations. They confirmed that none of the parameters correlated with tremor.44 A possibility is the neuronal activity changes could happen ahead of a particular symptom. Symptom presentation may be due to some degree of accumulation of neuronal activities. Previous studies on LFP have showed similar outcomes. They found that 13–20 Hz frequency band oscillations showed greater effect in the MA hemisphere.21 A study on early-stage and advanced-stage PD patients failed to find the difference on neuronal oscillation activities, which may be due to the limited sample size used in this study.13 DBS or L-dopa administration can decrease the power of βFB oscillations,33,35,36 and direct evidence has shown the reduction in the subthalamic 8–35 Hz oscillatory activity correlates with clinical improvement in PD.45
Our data also showed significant differences on the lateralized UPDRS sub-score and symptom duration in the MA and LA groups, and no differences were observed on the number of dominant hand in both groups. The length of neuronal trajectories between both the hemispheres were very similar and the number of cells recorded showed no significant difference. The distribution of neurons in the STN was in accordance with our previous studies,16,22 with the majority of oscillatory neurons located in the dorsal two-third parts of the STN. Comparing the MA and LA sides of the brain in the individual PD patient could reduce the influence of other interfering factors.
Conclusion
Our results provided new evidence related to neuronal activity and disease progression in PD. We have shown in this study that the firing rate in the MA-STN was higher than in the LA side, and we have also identified that the higher rate significantly correlated with higher UPDRS sub-score. The βFB oscillatory neurons were found in a larger proportion in the MA group while there was a larger percentage of TFB cells in the LA group. These results reinforced the hypothesis of the basal ganglia circuit and emphasize the importance of βFB oscillatory in the generation of Parkinsonian motor deficits.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (81371256, 81171061, and 81361128012) and the Ministry of Education of the Republic of China (PXM2019-026283-000002). We thank International Science Editing for editing this manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Hoehn MM, Yahr MD. Parkinsonism: onset, progression and mortality. Neurology. 1967;17(5):427–442. doi:10.1212/WNL.17.5.427
2. Postuma RB, Berg D, Stern M, et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov Disord. 2015;30(12):1591–1601. doi:10.1002/mds.26424
3. Toth C, Rajput M, Rajput AH. Anomalies of asymmetry of clinical signs in parkinsonism. Mov Disord. 2004;19(2):151–157. doi:10.1002/mds.v19:2
4. RL A, AB Y, JB P. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989;12(10):366–375. doi:10.1016/0166-2236(89)90074-X
5. DeLong MR. Primate models of movement disorders of basal ganglia origin. Trends Neurosci. 1990;13(7):281–285. doi:10.1016/0166-2236(90)90110-V
6. Kempster PA, Gibb WR, Stern GM, Lees AJ. Asymmetry of substantia nigra neuronal loss in Parkinson’s disease and its relevance to the mechanism of levodopa related motor fluctuations. J Neurol Neurosurg Psychiatry. 1989;52(1):72–76. doi:10.1136/jnnp.52.1.72
7. Bernheimer H, Birkmayer W, Hornykiewicz O, Jellinger K, Seitelberger F. Brain dopamine and the syndromes of Parkinson and Huntington. Clinical, morphological and neurochemical correlations. J Neurol Sci. 1973;20(4):415–455. doi:10.1016/0022-510X(73)90175-5
8. Lee CS, Schulzer M, Mak E, Hammerstad JP, Calne S, Calne DB. Patterns of asymmetry do not change over the course of idiopathic parkinsonism: implications for pathogenesis. Neurology. 1995;45(3 Pt 1):435–439. doi:10.1212/WNL.45.3.435
9. Snow BJ, Tooyama I, McGeer EG, et al. Human positron emission tomographic [18F] fluorodopa studies correlate with dopamine cell counts and levels. Ann Neurol. 1993;34(3):324–330. doi:10.1002/(ISSN)1531-8249
10. Tatsch K, Schwarz J, Mozley PD, et al. Relationship between clinical features of Parkinson’s disease and presynaptic dopamine transporter binding assessed with [123I] IPT and single-photon emission tomography. Eur J Nucl Med. 1997;24(4):415–421. doi:10.1007/BF00881814
11. Cassidy M, Brown P. Spectral phase estimates in the setting of multidirectional coupling. J Neurosci Methods. 2003;127(1):95–103. doi:10.1016/S0165-0270(03)00129-8
12. Bergman H, Wichmann T, Karmon B, DeLong M. The primate subthalamic nucleus. II. Neuronal activity in the MPTP model of parkinsonism. J Neurophysiol. 1994;72(2):507–520. doi:10.1152/jn.1994.72.2.507
13. Remple MS, Bradenham CH, Kao CC, Charles PD, Neimat JS, Konrad PE. Subthalamic nucleus neuronal firing rate increases with Parkinson’s disease progression. Mov Disord. 2011;26(9):1657–1662. doi:10.1002/mds.23708
14. Wichmann T, Soares J. Neuronal firing before and after burst discharges in the monkey basal ganglia is predictably patterned in the normal state and altered in parkinsonism. J Neurophysiol. 2006;95(4):2120–2133. doi:10.1152/jn.01013.2005
15. Miller WC, DeLong MR. Altered tonic activity of neurons in the globus pallidus and subthalamic nucleus in the primate MPTP model of parkinsonism. In: Carpenter MB, Jayaraman A, editors. The Basal Ganglia II: Structure and Function—Current Concepts. Boston, MA: Springer US; 1987:415–427.
16. Feng H, Zhuang P, Hallett M, Zhang Y, Li J, Li Y. Characteristics of subthalamic oscillatory activity in parkinsonian akinetic-rigid type and mixed type. Int J Neurosci. 2016;126(9):819–828. doi:10.3109/00207454.2015.1074225
17. Du G, Zhuang P, Hallett M, Zhang Y, Li J, Li Y. Properties of oscillatory neuronal activity in the basal ganglia and thalamus in patients with Parkinson’s disease. Transl Neurodegener. 2018;7:17. doi:10.1186/s40035-018-0123-y
18. Coelho M, Marti MJ, Tolosa E, et al. Late-stage Parkinson’s disease: the Barcelona and Lisbon cohort. J Neurol. 2010;257(9):1524–1532. doi:10.1007/s00415-010-5566-8
19. Alves G, Larsen JP, Emre M, Wentzel-Larsen T, Aarsland D. Changes in motor subtype and risk for incident dementia in Parkinson’s disease. Mov Disord. 2006;21(8):1123–1130. doi:10.1002/(ISSN)1531-8257
20. Louis ED, Tang MX, Cote L, Alfaro B, Mejia H, Marder K. Progression of parkinsonian signs in Parkinson disease. Arch Neurol. 1999;56(3):334–337. doi:10.1001/archneur.56.3.334
21. Shreve LA, Velisar A, Malekmohammadi M, et al. Subthalamic oscillations and phase amplitude coupling are greater in the more affected hemisphere in Parkinson’s disease. Clin Neurophysiol. 2017;128(1):128–137. doi:10.1016/j.clinph.2016.10.095
22. Guo S, Zhuang P, Hallett M, et al. Subthalamic deep brain stimulation for Parkinson’s disease: correlation between locations of oscillatory activity and optimal site of stimulation. Parkinsonism Relat Disord. 2013;19(1):109–114. doi:10.1016/j.parkreldis.2012.08.005
23. Li X, Zhuang P, Hallett M, Zhang Y, Li J, Li Y. Subthalamic oscillatory activity in parkinsonian patients with off-period dystonia. Acta Neurol Scand. 2016;134(5):327–338. doi:10.1111/ane.2016.134.issue-5
24. Moran A, Bergman H, Israel Z, Bar-Gad I. Subthalamic nucleus functional organization revealed by parkinsonian neuronal oscillations and synchrony. Brain. 2008;131(Pt 12):3395–3409. doi:10.1093/brain/awn270
25. Hutchison WD, Allan RJ, Opitz H, et al. Neurophysiological identification of the subthalamic nucleus in surgery for Parkinson’s disease. Ann Neurol. 1998;44(4):622–628. doi:10.1002/(ISSN)1531-8249
26. Bejjani BP, Dormont D, Pidoux B, et al. Bilateral subthalamic stimulation for Parkinson’s disease by using three-dimensional stereotactic magnetic resonance imaging and electrophysiological guidance. J Neurosurg. 2000;92(4):615–625. doi:10.3171/jns.2000.92.4.0615
27. Levy R, Hutchison WD, Lozano AM, Dostrovsky JO. High-frequency synchronization of neuronal activity in the subthalamic nucleus of parkinsonian patients with limb tremor. J Neurosci. 2000;20(20):7766–7775. doi:10.1523/JNEUROSCI.20-20-07766.2000
28. Chopek JW, Hultborn H, Brownstone RM. Multistable properties of human subthalamic nucleus neurons in Parkinson’s disease. Proc Natl Acad Sci. 2019;116(48):24326–24333.
29. Levy R, Ashby P, Hutchison WD, Lang AE, Lozano AM, Dostrovsky JO. Dependence of subthalamic nucleus oscillations on movement and dopamine in Parkinson’s disease. Brain. 2002;125(Pt 6):1196–1209. doi:10.1093/brain/awf128
30. Filali M, Hutchison WD, Palter VN, Lozano AM, Dostrovsky JO. Stimulation-induced inhibition of neuronal firing in human subthalamic nucleus. Exp Brain Res. 2004;156(3):274–281. doi:10.1007/s00221-003-1784-y
31. DeLong MR, Wichmann T. Circuits and circuit disorders of the basal ganglia. Arch Neurol. 2007;64(1):20–24. doi:10.1001/archneur.64.1.20
32. Rodriguez MC, Guridi OJ, Alvarez L, et al. The subthalamic nucleus and tremor in Parkinson’s disease. Mov Disord. 1998;13(Suppl 3):111–118. doi:10.1002/mds.870131320
33. Brown P, Oliviero A, Mazzone P, Insola A, Tonali P, Di Lazzaro V. Dopamine dependency of oscillations between subthalamic nucleus and pallidum in Parkinson’s disease. J Neurosci. 2001;21(3):1033–1038. doi:10.1523/JNEUROSCI.21-03-01033.2001
34. Cassidy M, Mazzone P, Oliviero A, et al. Movement‐related changes in synchronization in the human basal ganglia. Brain. 2002;125(6):1235–1246. doi:10.1093/brain/awf135
35. Brown P, Mazzone P, Oliviero A, et al. Effects of stimulation of the subthalamic area on oscillatory pallidal activity in Parkinson’s disease. Exp Neurol. 2004;188(2):480–490. doi:10.1016/j.expneurol.2004.05.009
36. Little S, Brown P. What brain signals are suitable for feedback control of deep brain stimulation in Parkinson’s disease? Ann N Y Acad Sci. 2012;1265:9–24. doi:10.1111/j.1749-6632.2012.06650.x
37. Timmermann L, Florin E. Parkinson’s disease and pathological oscillatory activity: is the beta band the bad guy? - New lessons learned from low-frequency deep brain stimulation. Exp Neurol. 2012;233(1):123–125. doi:10.1016/j.expneurol.2011.10.022
38. Chen CC, Litvak V, Gilbertson T, et al. Excessive synchronization of basal ganglia neurons at 20 Hz slows movement in Parkinson’s disease. Exp Neurol. 2007;205(1):214–221. doi:10.1016/j.expneurol.2007.01.027
39. Timmermann L, Wojtecki L, Gross J, et al. Ten-Hertz stimulation of subthalamic nucleus deteriorates motor symptoms in Parkinson’s disease. Mov Disord. 2004;19(11):1328–1333. doi:10.1002/(ISSN)1531-8257
40. Little S, Pogosyan A, Kuhn AA, Brown P. Beta band stability over time correlates with Parkinsonian rigidity and bradykinesia. Exp Neurol. 2012;236(2):383–388. doi:10.1016/j.expneurol.2012.04.024
41. Chen CC, Hsu YT, Chan HL, et al. Complexity of subthalamic 13–35 Hz oscillatory activity directly correlates with clinical impairment in patients with Parkinson’s disease. Exp Neurol. 2010;224(1):234–240. doi:10.1016/j.expneurol.2010.03.015
42. Hutchison WD, Lozano AM, Tasker RR, Lang AE, Dostrovsky JO. Identification and characterization of neurons with tremor-frequency activity in human globus pallidus. Exp Brain Res. 1997;113(3):557–563. doi:10.1007/PL00005606
43. Amtage F, Henschel K, Schelter B, et al. Tremor-correlated neuronal activity in the subthalamic nucleus of Parkinsonian patients. Neurosci Lett. 2008;442(3):195–199. doi:10.1016/j.neulet.2008.06.087
44. Sharott A, Gulberti A, Zittel S, et al. Activity parameters of subthalamic nucleus neurons selectively predict motor symptom severity in Parkinson’s disease. J Neurosci. 2014;34(18):6273–6285. doi:10.1523/JNEUROSCI.1803-13.2014
45. Kuhn AA, Kupsch A, Schneider GH, Brown P. Reduction in subthalamic 8–35 Hz oscillatory activity correlates with clinical improvement in Parkinson’s disease. Eur J Neurosci. 2006;23(7):1956–1960. doi:10.1111/j.1460-9568.2006.04717.x
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.