Back to Journals » Clinical Ophthalmology » Volume 13
Assessment of tear-evaporation rate in thyroid-gland patients
Authors Abusharaha A, Alturki AA, Alanazi SA, Fagehi R , Al-Johani N, El-Hiti GA , Masmali AM
Received 25 September 2018
Accepted for publication 31 October 2018
Published 7 January 2019 Volume 2019:13 Pages 131—135
DOI https://doi.org/10.2147/OPTH.S188614
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Ali Abusharaha,1 Abdulaziz A Alturki,1 Saud A Alanazi,1 Raied Fagehi,1 Naji Al-Johani,2 Gamal A El-Hiti,1 Ali M Masmali1
1Department of Optometry, College of Applied Medical Sciences, King Saud University, Riyadh 11433, Saudi Arabia; 2Department of Endocrinology and Thyroid Oncology, Comprehensive Cancer Center, King Fahad Medical City, Riyadh 11433, Saudi Arabia
Objective: To assess the tear-evaporation rate in thyroid-gland patients using a VapoMeter.
Methods: Twenty thyroid gland patients aged 18–43 years (mean 34.3±6.3 years) completed the study. Additionally, an age-matched control group of 20 patients aged 18–43 years (32.2±5.1 years) was enrolled in the study for comparison purposes. An Ocular Surface Disease Index dry-eye questionnaire was completed, followed by a test to determine the tear-evaporation rate using the VapoMeter. The test was performed three times per subject by the same examiner. Two readings were obtained each time.
Results: Significant differences (P<0.05) were found between mean Ocular Surface Disease Index and tear-evaporation-rate scores within the study and control groups. The average tear-evaporation rate was much higher in the study group (median 41.2 [IQR 41.4] g/m2·h) than the control group (15.7 [13.7] g/m2·h). Moreover, the average Ocular Surface Disease Index score for thyroid-gland patients was much higher (15.6 [23.4]) compared to the control group (5.5 [7.50]).
Conclusion: The tear-evaporation rate in thyroid-gland patients was found to be much higher than normal-eye subjects.
Keywords: thyroid gland, thyroid-associated ophthalmopathy, Graves’s ophthalmopathy, dry eye, tear-evaporation rate, evaporative dry eye
Introduction
The thyroid gland produces thyroxine, which regulates the body’s metabolism. Abnormalities in the thyroid gland affect hormone secretion, which leads to disease. The two most common thyroid-gland diseases are hypothyroidism and hyperthyroidism. Graves’s disease is a systemic autoimmune condition that is associated with the loss of the immune abilities of the thyrotropin receptor.1–3 Thyroid gland patients usually experience dry eye syndrome as a result of proptosis, lid retraction, and exposure.4 Several clinical signs and symptoms are common in thyroid-gland patients, such as exophthalmos, which leads to the expansion of eye soft tissue.5–7 Exophthalmos can lead to incomplete eyelid closure, and as a result increases the tear-evaporation rate (TER), as well as tear hyperosmolarity.8,9 Moreover, retraction of the upper eyelid is a common clinical sign in thyroid patients.10 In the Arab world, reports indicate that the prevalence of thyroid diseases is high.11–17 Levothyroxine, a medication that produces the thyroid hormone, is prescribed for hypothyroidism patients.18 On the other hand, thioamide drugs can be used to reduce thyroid-gland secretion in hyperthyroidism patients.18 Other medications, such as carbimazole and methimazole, can be used to treat hypothyroidism.19 Surgical intervention is sometimes needed for thyroid patients who cannot tolerate medication due to iodine allergy.
Dry-eye disease is a multifactorial condition associated with the disturbance of vision, ocular discomfort, and instability of tear film.20 The two main types of dry eye are known as aqueous tear-deficiency and evaporative dry eye.20 The most common factors that can lead to dry eye are high osmolarity of tears, accelerated TER, and ocular surface inflammation.20 Due to the complexity of tear film, no single test can be used to diagnose eye dryness, so several combined tests are needed to assess it. Some of these tests measure the tear-production rate, while others assess the quality of the tears, tear osmolarity, tear-ferning patterns, and TER. The Schirmer test, phenol red thread test, tear-meniscus height, tear breakup time, tear osmolarity, and tear-ferning tests are all commonly used to detect dryness of the eye.21–26 Studies have shown that half of all asymptomatic patients demonstrate clinical signs of dry eye.27 Therefore, it is important to examine dry-eye patients on a regular basis, in order to avoid discomfort, reduced vision, cornea damage, and all other symptoms that are associated with dry eye.
Thyroid-gland patients have increased width of the palpebral fissure and several eyelid alterations, which leads to tear-film instability, corneal exposure, increased TER, and tear osmolarity.28–31 Several studies have concluded that normal TER is 19–21 g/m2·h.32–35 On the other hand, the TER in thyroid gland patients was found to be 36–86 g/m2·h.36,37 In the current study, we evaluated the TER in thyroid-gland patients and compared the results with normal-eye subjects. As far as we are aware, this is the first report to evaluate TER in thyroid-gland patients using a VapoMeter. the VapoMeter is portable, light, practical, not sensitive to ambient airflows, can be used in a wide range (≥200 g/m2·h) and in all angles. Moreover, it does not need daily calibration and requires a short time (10 seconds) for the measurement of TER.
Methods
This observational, case-control, nonrandomized study included 20 thyroid-gland patients (five men, 15 women) aged 18–43 years (mean 34.3±6.3 years) with two types of thyroid-gland disease (nine hyperthyroidism and eleven hypothyroidism patients). Subjects were recruited from the Endocrinology Clinic at King Fahad Medical City, Riyadh. Additionally, an age-matched control group of 20 patients (six men, 14 women) aged 18–43 years (32.2±5.1 years) was enrolled in the study. Informed written consent of all participants was obtained prior to conducting this study. Ethical approval for the study was obtained from the College of Applied Medical Science Ethics Committee, King Fahad Medical City. All participants were treated according to the tenets of the Declaration of Helsinki. Exclusion criteria included subjects with a risk factor for eye dryness, such as those who had undergone recent ocular surgery or consumed medication, as well as smokers, pregnant women, and breastfeeding women. Moreover, any patients with diabetes, those with any abnormalities of the eyelids and lashes, contact-lens wearers, and patients who had had thyroid surgery or radioactive iodine therapy over the last 3 years were also excluded from the study.
An Ocular Surface Disease Index (OSDI) dry-eye questionnaire was completed, followed by the assessment of TER. TER was measured using a VapoMeter (Delfin Technologies, Kuopio, Finland). The test was performed three times, during which two readings were obtained in each test. The first reading with obtained with both eyes open and blinking normally, whereas the second test was conducted with both eyes closed and with a 2-minute gap between readings. The TER was calculated by subtracting the reading when both eyes were closed from the one obtained when both eyes were open, and averages were subsequently calculated for the three readings. The TER for normal eye conditions is considered to be <25 g/m2·h and >25 g/m2·h for evaporative dry-eye conditions.32–35 All tests were carried out by the same examiner at a temperature of 23°C and humidity <40%. The effect of alcohol pads (70% 2-propanol) on the VapoMeter was tested. There was no significant difference between the use of the device immediately or after ≥15 minutes of using the alcohol pad.
Data were collected using Excel (Microsoft Corporation, Redmond, WA, USA) and analyzed using SPSS version 22.0 (IBM Corporation, Armonk, NY, USA) for Windows. Data obtained were found to be abnormally distributed for both OSDI and TER scores. Therefore, the Mann–Whitney U test was used to analyze the data (P<0.05).
Results
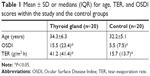
Mean ± SD for age and median (IQR) for OSDI questionnaire scores and TER measurements are shown in Table 1. OSDI scores ranged from 0 to 84 and from 0 to 27 for the study and control groups, respectively. There was a medium negative correlation (r=−0.30, P=0.05) between OSDI and TER scores.
  | Table 1 Mean ± SD or medians (IQR) for age, TER, and OSDI scores within the study and the control groups |
Side-by-side box plots for OSDI and TER scores for both study and control groups are shown in Figures 1 and 2, respectively. Figure 1 clearly shows that OSDI scores were much higher in thyroid-gland patients than the control group. For TER measurements (Figure 2), a significant difference was found between the median average for thyroid gland patients (41.2 [41.4]) and the control group (15.7 [13.7]). For thyroid-gland patients, the TER ranged from 12.0 to 182.3 g/m2·h and for the control group from 7.0 to 36.4 g/m2·h. TER measurements showed that 17 thyroid gland patients had higher readings than the normal level.
  | Figure 1 Side-by-side box plots for OSDI scores within study and control groups. |
  | Figure 2 Side-by-side box plots for TER scores within study and control groups. |
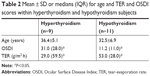
Thyroid-gland subjects (20 patients) who were examined represented patients suffering from two different types of disease: hyperthyroidism (n=9) and hypothyroidism (n=11). The median average for the TER was low in hyperthyroidism patients (29.0 [59.5]) compared with those patients suffering from hypothyroidism (53.0 [28.0]), as shown in Table 2. The median OSDI score was high in hyperthyroidism patients (31.0 [28.0]) compared with hypothyroidism patients (11.2 [11.0]).
Discussion
Thyroid-gland patients suffer from expansion of soft tissue in the eye, which is a condition known as exophthalmos.5–7 Exophthalmos leads to hyperosmolarity and a high TER, due to incomplete closure of the eyelids.8,9 Such phenomena can disturb the ocular tear film, leading to dry-eye symptoms. The normal TER is in the range of 19–21 g/m2·h.3–36 The current study has indicated a high TER in thyroid-gland patients compared to the control group, and the differences observed were deemed significant. It was found that TER was higher than the normal level in the majority (85%) of patients who suffered from thyroid-gland disease. Such high TER levels lead to dry-eye symptoms, possibly due to the disorderly function of the eyelids and reduced blink rate (intrinsic causes) as a result of corneal exposure.38 It is believed that disturbances within the lipid layer in the tear film are responsible for the high TER. The lipid polar phase within the tear film contains glycolipids and phospholipids, and thus works as a surfactant between the hydrophilic aqueous–mucin layer and the apolar lipid layer. Therefore, it supports the interaction between aqueous–mucin layers and acts as a barrier.39 The apolar lipid layer of the tear film contains cholesterol esters, wax, and triglycerides, and provides the interface between the air and tear film. Incidentally, a normal human tear-film lipid layer tends to reduce TER by ~90%–95%.40–42
An inverse relationship between the thickness of the lipid layer and the TER has been suggested.43 In addition, a correlation between thyroid-gland disease and dry-eye disease has been found.28 For example, results of the tear-breakup-time test and the Schirmer test that were carried out on 30 thyroid-gland patients indicated low average scores compared to normal-eye subjects. This could possibly be due to exophthalmos, eyelid retraction, and lagophthalmos.28 Positive vital staining with lissamine green was observed in 60% of the patients who participated in the study.28 Moreover, a significant correlation between thyroid eye disease and poor impression cytology was established using Nelson’s grading system.28 A more detailed study that includes a higher number of thyroid-gland patients is needed to understand better the mechanism by which dry-eye disease occurs within patients suffering from thyroid-gland disease. Moreover, it is essential to detect the thickness of the lipid layer of the tear film, Hertel exophthalmic values, upper- and lower-eyelid positions, and lagophthalmos in thyroid-gland patients.
Conclusion
The TER in thyroid-gland patients is much higher than normal-eye subjects. Clearly, thyroid-gland disease is a risk factor for eye dryness. Thyroid-gland patients may possibly suffer from an evaporative dry-eye condition due to disturbances within the apolar lipid layer. Lipid-rich eyedrops could thus be used to reduce the discomfort associated with eye dryness due to thyroid-gland disease.
Acknowledgment
This project was supported by King Saud University, Deanship of Scientific Research, Research Chairs.
Disclosure
The authors report no conflicts of interest in this work.
References
Smith TJ, Hegedüs L. Graves’ disease. N Engl J Med. 2016;375(16):1552–1565. | ||
Pujol-Borrell R, Giménez-Barcons M, Marín-Sánchez A, Colobran R. Genetics of Graves’ disease: special focus on the role of TSHR gene. Horm Metab Res. 2015;47(10):753–766. | ||
Wiersinga WM. Thyroid autoimmunity. Endocr Dev. 2014;26:139–157. | ||
Gupta A, Sadeghi PB, Akpek EK. Occult thyroid eye disease in patients presenting with dry eye symptoms. Am J Ophthalmol. 2009;147(5):919–923. | ||
Bahn RS. Graves’ ophthalmopathy. N Engl J Med. 2010;362(8):726–738. | ||
Bartalena L, Wiersinga WM, Pinchera A. Graves’ ophthalmopathy: state of the art and perspectives. J Endocrinol Invest. 2004;27(3):295–301. | ||
Abràmoff MD, Kalmann R, de Graaf ME, Stilma JS, Mourits MP. Rectus extraocular muscle paths and decompression surgery for Graves orbitopathy: mechanism of motility disturbances. Invest Ophthalmol Vis Sci. 2002;43(2):300–307. | ||
Gilbard JP, Farris RL. Ocular surface drying and tear film osmolarity in thyroid eye disease. Acta Ophthalmol (Copenh). 1983;61(1):108–116. | ||
Khurana AK, Sunder S, Ahluwalia BK, Malhotra KC. Tear film profile in graves’ ophthalmopathy. Acta Ophthalmol (Copenh). 1992;70(3):346–349. | ||
Cruz AA, Ribeiro SF, Garcia DM, Akaishi PM, Pinto CT. Graves upper eyelid retraction. Surv Ophthalmol. 2013;58(1):63–76. | ||
Ghawil M, Tonutti E, Abusrewil S, et al. Autoimmune thyroid disease in Libyan children and young adults with type 1 diabetes mellitus. Eur J Pediatr. 2011;170(8):983–987. | ||
Nouh AM, Eshnaf IAM, Basher MA. Prevalence of thyroid dysfunction and its effect on serum lipid profiles in a Murzok, Libya population. Thyroid Sci. 2008;3(10):1–6. | ||
Lamfon HA. Thyroid disorders in Makkah, Saudi Arabia. Ozean J Appl Sci. 2008;1(1):52–58. | ||
Sawka AM, Brierley JD, Tsang RW, et al. An updated systematic review and commentary examining the effectiveness of radioactive iodine remnant ablation in well-differentiated thyroid cancer. Endocrinol Metab Clin North Am. 2008;37(2):457–480. | ||
Abdulmughni YA, Al-Hureibi MA, Al-Hureibi KA, Ghafoor MA, Al-Wadan AH, Al-Hureibi YA. Thyroid cancer in Yemen. Saudi Med J. 2004;25(1):55–59. | ||
Akbar DH, Ahmed MM, Al-Mughales J. Thyroid dysfunction and thyroid autoimmunity in Saudi type 2 diabetics. Acta Diabetol. 2006;43(1):14–18. | ||
Nasheiti NA. Childhood hypothyroidism in Iraq: a retrospective study. Int J Endocrinol Metabol. 2005;3(3):136–139. | ||
Garber JR, Cobin RH, Gharib H, et al; American Association Of Clinical Endocrinologists And American Thyroid Association Taskforce On Hypothyroidism In Adults. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Thyroid. 2012;22(12):1200–1235. | ||
Fumarola A, Di Fiore A, Dainelli M, Grani G, Calvanese A. Medical treatment of hyperthyroidism: state of the art. Exp Clin Endocrinol Diabetes. 2010;118(10):678–684. | ||
The definition and classification of dry eye disease: report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007;5(2):75–92. | ||
Masmali A, Alqahtani TA, Alharbi A, El-Hiti GA. Comparative study of repeatability of phenol red thread test versus Schirmer test in normal adults in Saudi Arabia. Eye Contact Lens. 2014;40(3):127–131. | ||
Ibrahim OM, Dogru M, Takano Y, et al. Application of visante optical coherence tomography tear meniscus height measurement in the diagnosis of dry eye disease. Ophthalmology. 2010;117(10):1923–1929. | ||
Masmali A, Alrabiah S, Alharbi A, El-Hiti GA, Almubrad T. Investigation of tear osmolarity using the TearLabOsmolarity System in normal adults in Saudi Arabia. Eye Contact Lens. 2014;40(2):74–78. | ||
Cho P, Ho KY, Huang YC, Chui HY, Kwan MC. Comparison of noninvasive tear break-up time measurements from black and white background instruments. Optom Vis Sci. 2004;81(6):436–441. | ||
Masmali AM, Al-Bahlal JM, El-Hiti GA, et al. Repeatability and diurnal variation of tear ferning test. Eye Contact Lens. 2015;41(5):262–267. | ||
Masmali AM, Al-Qhtani S, Al-Gasham TM, El-Hiti GA, Purslow C, Murphy PJ. Application of a new grading scale for tear ferning in non-dry eye and dry eye subjects. Cont Lens Anterior Eye. 2015;38(1):39–43. | ||
Sullivan BD, Crews LA, Messmer EM, et al. Correlations between commonly used objective signs and symptoms for the diagnosis of dry eye disease: clinical implications. Acta Ophthalmol. 2014;92(2):161–166. | ||
Rizvi SAR, Rana V, Sheelu SS, Maheshwari V, Gupta Y. Dry eye evaluation in thyroid associated orbitopathy. Int J Ocul Oncol Oculoplasty. 2016;2(2):90–94. | ||
Sabita P, Ajit T, Narayan SD, Kumar SA, Niranjan A. Ocular manifestations in thyroid eye disorder: a cross-sectional study from Nepal. Int J Clin Med. 2016;7(12):814–823. | ||
Huh HD, Kim JH, Kim SJ, Yoo JM, Seo SW. The change of lacrimal gland volume in Korean patients with thyroid-associated ophthalmopathy. Korean J Ophthalmol. 2016;30(5):319–325. | ||
Novaes P, Diniz Grisolia AB, Smith TJ. Update on thyroid-associated Ophthalmopathy with a special emphasis on the ocular surface. Clin Diabetes Endocrinol. 2016;2:19. | ||
Tomlinson A, Doane MG, McFadyen A. Inputs and outputs of the lacrimal system: review of production and evaporative loss. Ocul Surf. 2009;7(4):186–198. | ||
McCulley JP, Shine WE, Aronowicz J, Oral D, Vargas J. Presumed hyposecretory/hyperevaporative KCS: tear characteristics. Trans Am Ophthalmol Soc. 2003;101:141–152; discussion 152–154. | ||
Tomlinson A, Pearce EI, Simmons PA, Blades K. Effect of oral contraceptives on tear physiology. Ophthalmic Physiol Opt. 2001;21(1):9–16. | ||
Khanal S, Tomlinson A, Diaper CJ. Tear physiology of aqueous deficiency and evaporative dry eye. Optom Vis Sci. 2009;86(11):1235–1240. | ||
Khanal S, Tomlinson A, Esakowitz L, et al. Changes in corneal sensitivity and tear physiology after phacoemulsification. Ophthalmic Physiol Opt. 2008;28(2):127–134. | ||
McCann LC, Tomlinson A, Pearce EI, Papa V. Effectiveness of artificial tears in the management of evaporative dry eye. Cornea. 2012;31(1):1–5. | ||
Yokoi N, Komuro A. Non-invasive methods of assessing the tear film. Exp Eye Res. 2004;78(3):399–407. | ||
McCulley JP, Shine WE. The lipid layer of tears: dependent on meibomian gland function. Exp Eye Res. 2004;78(3):361–365. | ||
Mathers WD, Lane JA. Meibomian gland lipids, evaporation, and tear film stability. Adv Exp Med Biol. 1998;438:349–360. | ||
Tsubota K, Yamada M. Tear evaporation from the ocular surface. Invest Ophthalmol Vis Sci. 1992;33(10):2942–2950. | ||
Hisatake K, Tanaka S, Aizawa Y. Evaporation rate of water in a vessel. J Appl Phys. 1993;73(11):7395–7401. | ||
Bron AJ, Tiffany JM. The contribution of meibomian disease to dry eye. Ocul Surf. 2004;2(2):149–165. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.