Back to Journals » Clinical Interventions in Aging » Volume 9
Assessment of cognitive impairment in patients with Parkinson's disease: prevalence and risk factors
Authors Wang Q, Zhang Z, Li L, Wen H, Xu Q
Received 29 April 2013
Accepted for publication 4 August 2013
Published 12 February 2014 Volume 2014:9 Pages 275—281
DOI https://doi.org/10.2147/CIA.S47367
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Qiumei Wang,1 Zhenxin Zhang,2 Ling Li,2 Hongbo Wen,2 Qun Xu3,4
1Department of Geriatrics, 2Department of Neurology, 3School of Basic Medicine, Peking Union Medical College Hospital, 4Department of Epidemiology and Biostatistics, Institute of Basic Medical Sciences, Chinese Academy of Medical Sciences, Beijing, People's Republic of China
Background: Although Parkinson's disease (PD) is clinically characterized by motor symptoms, cognitive impairment is one of the most disabling non-motor symptoms. Despite it attracting increasing attention worldwide, less is known about its prevalence in the Chinese population. The objective of this study was to assess cognitive impairment and related risk factors in Chinese PD patients.
Methods: We collected the demographic, diagnostic, and treatment information of 901 PD patients from 42 centers throughout the People's Republic of China, then administered a battery of neuropsychological tests, to assess motor, cognitive, and neuropsychiatric symptoms.
Results: Overall, 193 of 901 (21.4%) PD patients met the criteria for dementia (PD-D), and 206 (22.8%) met the criteria for mild cognitive impairment (PD-MCI). Visuospatial dysfunction and attention/executive impairment predominated. Increased severity of cognitive impairment was associated with greater motor impairment. Patients with psychiatric symptoms, such as depression and hallucinations, were more likely to have dementia. Potentially, the younger-aged and more educated are shown less cognitive impairment, but age at onset, and levodopa equivalent dose, were not associated with the presence of cognitive dysfunction.
Conclusion: The prevalence and profile of cognitive impairment in Chinese PD patients, as well as the risk factors, are similar as those reported for other races, but the frequency of nonamnestic cognitive domains differs.
Keywords: cognitive impairment, risk factor, prevalence, Parkinson's disease
Introduction
Cognitive impairment is one of the most common and disabling non-motor symptoms in Parkinson’s disease (PD), yet it is not routinely treated in clinical practice. The prevalence of PD-related cognitive impairment varies greatly across studies. Moreover, although overt dementia in PD patients (PD-D) has been extensively investigated,1 and specific diagnostic criteria have been developed,2 there is no established diagnosis criteria for subtler impairment in PD patients.3
Our understanding of cognitive impairment and dementia in the Chinese PD population is particularly limited. Two small-scale studies,4,5 and reviews of the literature on all non-motor symptoms,6 have revealed that Chinese PD patients suffer from non-motor symptoms, but there appear to be some differences, compared with prevalence rates and risk factors for these symptoms within Western populations.6 Notably, there is no epidemiological data on cognitive impairment from a large sample of Chinese patients. Therefore, we recruited a series of PD patients, to investigate the frequency and profiles of cognitive impairment, and to assess possible predictive risk factors, within this population.
Patients and methods
Patients and study instruments
A total of 901 PD patients were recruited from 42 university-affiliated hospitals throughout seven cities in The People’s Republic of China from December 2005 to January 2008. All relevant information from patients in each hospital was recorded. Patients were then examined using a standardized interview documents on demographic, diagnostic, and treatment information, along with a three-part assessment battery. This study was approved by the institutional review boards of the relevant hospitals, and written informed consent was obtained from every patient selected.
The neuropsychological examination comprised three parts. Section A consisted of patient demographics, including age, gender, education, occupation, and living situation. Section B documented diagnostic and treatment information, and data regarding the onset and symptoms of PD. Section C comprised four scales for rating symptoms. The motor and related symptoms battery contained the Hoehn and Yahr Scale (HY),7 Part I (Mentation, Behavior, and Mood), Part II (Activities of Daily Living), and Part IV (Complications) of the Unified PD Rating Scale (UPDRS).8 The cognitive battery contained the Chinese version of the Mini Mental State Exam (C-MMSE) (30 maximum) and the Montreal Cognitive Assessment (MoCA) (30 maximum). Additionally, all patients were assessed using standard cognitive tests: the Fuld Object Memory Evaluation (0–20 points), to assess the memory domain, the Fuld Verbal Fluency test (FVFT), to evaluate verbal ability, the revised Wechsler Intelligence Scale for Children (WISC-R) Block Design (62 maximum), to assess visuospatial function, the revised Wechsler Adult Intelligence Scale (WAIS-R) Digit Span (22 points), to evaluate attention, and the seventeen-item Hamilton Depression Rating Scale with 17 items (Hdrs-17) and Neuropsychiatric Inventory (NPI), to evaluate behavior and mood. Cut-offs for determination of cognitive impairment were based on individual education levels.
We included only cases of PD with clinical onset before the starting date of the survey. We employed a standardized diagnostic appraisal for all patients, which followed the UK Parkinson’s Disease Society Brain Bank Criteria for idiopathic PD. A principal investigator reconfirmed all assessments, after regional supervisors had made diagnoses using all available information from each center. The diagnosis of probable PD with dementia (PD-D) was made using criteria recommended by the Movement Disorder Society.2 PD patients with mild cognitive impairment (PD-MCI) were not clinically demented, but were impaired in any one of four cognitive domains: attention, executive function, visuospatial function, and free-recall memory.
Data analyses
Data are expressed as means with standard deviations (SD) for normal distributions. We used chi-square tests for nominal variables, and Fisher’s Exact tests for quantitative variables. Logistic regression analyses were performed to evaluate categorical predictor variables for PD-D and PD-MCI. Odds ratios (OR) and 95% confidence intervals (CI) were reported. Differences were considered statistically significant at P<0.05. Analyses were performed using SAS version 8.0 statistical software (SAS Institute Inc., Cary, NC, USA).
Results
Patient characteristics
A total of 901 consecutive patients met the UK Parkinson’s Disease Society Brain Bank Criteria for idiopathic PD; a total of 18 patients were excluded because of symptoms suggesting atypical parkinsonism (n=12), and overlapping enrollment with different centers (n=6). There were 562 (62.4%) men. The average age was 65.4 ± 10.7 years (Table 1). The average duration of PD in this sample was 5.5 ± 4.5 years. According to the HY scale, 19.2%, 62.2%, and 18.6% were at Stages I, II, and III or higher, respectively.
  | Table 1 Patient characteristics |
Cognitive impairment prevalence in PD
Among the 901 patients, 21.4% (n=193) met the PD-D criteria recommended by the Movement Disorder Society, and 22.8% (n=206) met the criteria for PD-MCI. Overall, 44.3% of PD patients had some degree of cognitive impairment (Table 2). The cognitive assessment scores for patients with PD-D, PD-MCI, and PD with no cognitive impairment (PD-NoCI) are shown in Table 3. PD-D and PD-MCI patients performed significantly worse in nearly all the domains, compared with PD-NoCI patients. C-MMSE and MoCA global cognitive scores were significantly lower for both PD-MCI and PD-D patients. Based on the proposed cut-off value of 26 (or less) to identify cognitive impairment using both MoCA and MMSE,10 we found that MoCA could detect 86.5% of PD-D and 84.5% of PD-MCI, whereas MMSE identified just 65.3% of PD-D and 33.6% of PD-MCI (Table 4). Our results indicate that MoCA is more sensitive than MMSE for assessing cognitive impairment in Chinese PD patients.
  | Table 2 Cognitive impairment prevalence and PD duration |
Cognitive deficit profiles in PD-MCI and PD-D
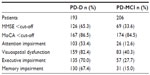
We employed a set of standard cognitive tests to investigate cognitive deficit profiles in PD-MCI and PD-D patients (Table 4). In PD-MCI patients, the most common cognitive deficits were visuospatial dysfunction (40.3%), executive dysfunction (27.7%), memory impairment (15.0%), and attention impairment (12.6%). Although we used different tests to assess attention and executive function, the basic cognitive processes of these two domains cannot be clearly divided, and often overlap. For this reason, we merged the two domains for analysis purposes, and found that 83 (40.3%) PD-MCI patients exhibited executive/attention dysfunction. Overall, most cognitive impairment in PD-MCI patients corresponded to visuospatial dysfunction and attention/executive impairment; only 15% of cognitive impairment in PD-MCI patients was amnestic (Table 4).
In PD-D patients, approximately 82.4% (159 patients) had visuospatial dysfunction; the frequencies of executive dysfunction, memory impairment, and attention impairment were 70.0%, 67.4%, and 53.4%, respectively (Table 4). When we merged the executive impairment and attention domains for analysis, 168 (87.0%) PD-D patients had executive and/or attention dysfunction. Overall, the attention/executive and visuospatial domains were the most commonly impaired; about one-third of PD-D patients were nonamnestic.
Predictive risk factors for PD-related cognitive impairment
Among demographic factors (Table 5), having longer education and a professional career, were potentially protective factors against cognitive impairment. Males were less likely to have PD-MCI (OR =0.63, P=0.02), but gender was not a significant predictive factor for PD-D (OR =0.97, P=0.90). Incidence of PD-MCI was much lower in patients with self-support finance independency (OR =0.44, P=0.002), but this was not the case with PD-D (OR =0.77, P=0.30). With regard to age, PD patients who were at least 75 years old were twenty-one times more likely to develop PD-D, and four times more likely to suffer from PD-MCI (P<0.001), than patients younger than 55 years. However, age at PD onset was not an influential factor.
Among clinical predictive risk factors (Table 5), cognitive impairment was associated with higher axial UPDRS impairment score, and advanced HY stages. Based on the Movement Disorder Society criteria, PD-D occurred in 12.1% of HY Stage I patients, 17.9% of Stage II patients, and 42.9% of patients at Stage III or higher. For PD-MCI, these values were 15.6%, 22.7%, and 30.9%, respectively (Stage ≥III versus Stage I PD-MCI: OR =3.97; Stage ≥III versus Stage I PD-D: OR =4.77; P=0.001). Patients with axial impairment were two times more likely to have cognitive impairment (PD-MCI: OR =2.07; PD-D: OR =2.57; P<0.05). Levodopa dosage was not a significantly influential factor. In addition, 24.4% (n=220) of all PD patients suffered from depression, and 7.0% experienced hallucinations. In this series of patients, severity of depression symptoms was positively associated with dementia risk (OR=1.98, P=0.03, the data was calculated in patients with Hdes-17 scores ≥12 verses patients with Hdes-17 scores <7), and patients with hallucinations were more likely to have dementia (OR =3.15; P<0.001). Clearly, cognitive impairment is associated with the severity of PD motor and neuropsychiatric symptoms in this population.
Discussion
We thoroughly and systematically investigated the prevalence of cognitive impairment in Chinese PD patients, and found that 22.8% had PD-MCI, while 21.4% met the PD-D criteria of the Movement Disorder Society. Two large studies, from Germany and Norway, have reported similar frequencies of PD-MCI (30.6%11 and 25.8%,12 respectively). The prevalence of PD-D described in other large studies varies greatly; a population-based study of PD patients in the Netherlands reported 22% dementia,13 and a study of 1,449 PD outpatients in Germany found that 29% exhibited dementia.14 A smaller German study (605 subjects), reported that 13.1% had dementia.11 The differences in PD-MCI and PD-D frequencies between studies are probably due to different study methods, varying criteria, and differences in disease severity. In addition, the studies mentioned above were cross-sectional, and only described point prevalence; cumulative prevalence is often much greater, as has been shown in a Norwegian cohort,15 and by the Sydney Multicenter Study.16,17 For example, in the former study, the point prevalence of dementia is 26%, while the 8-year cumulative prevalence of dementia is 78.2%.15 In the latter study, the point prevalence of dementia in patients at 20 years is 83%.16 Although it has been reported that Asian populations may have a lower incidence of dementia overall,18 our results indicate that the prevalence of cognitive impairment in Chinese PD patients is not significantly different from that observed in other races and areas.
With regard to cognitive impairment in PD-MCI and PD-D, the frequencies of memory impairment in our study are very close to those reported by others: 13.3%–15.1%12,19 in PD-MCI patients, and 67.4% in PD-D patients.20 In similarity with an earlier study,19 we found that attention/executive impairment and visuospatial dysfunction are the predominating forms of cognitive impairment in Chinese PD-MCI and PD-D patients. Memory and executive defects had been reported to be the most common impairment profiles in PD.21 Impaired attention was found to be an important aspect of cognitive impairment in PD; it is the most useful parameter to differentiate PD-D from Alzheimer’s disease.22 We employed the WAIS-R scale to assess attention, because it is routinely employed by the clinics involved in this study. Although WAIS-R is usually employed to measure working memory, the capacity to maintain attention is prerequisite for completing the test. Notably, the frequency of nonamnestic domain impairment was higher in our study sample than has been reported by other studies.12,19 The different results may be due to the use of different study methods, varying degrees of disease severity, and different demographic factors. However, it may also reflect diverse patterns of neuronal degeneration associated with PD.23 A recently published hypothesis for PD proposes two different patterns of disease evolution: ie, frontal-striatal basal ganglia circuits, and posterior cortical syndromes. Executive and attention defects are more obvious when the frontal-striatal pathway is involved, whereas visuospatial and memory dysfunction are more representative of posterior cortical deficits.24 In summary, the profile of cognitive domain impairment described here is similar to that reported in other races. Difference in the frequencies of domain impairment may be due to differences in methodology, racial composition, and disease evolution.
Understanding which factors can be used to predict cognitive impairment in PD is very important. We confirmed a previously reported series of potential risk factors for PD cognitive impairment in the current study. Patients with higher axial UPDRS impairment scores, HY stages,25,26 and ages26–28 are more likely to manifest cognitive impairment. Conversely, higher educational attainment,4,26 male gender,28 professional occupation, and financial independence are, potentially, protective factors against cognitive impairment. The latter two factors may arise because receiving education is more predominant among men during economically difficult times, while better chronic disease management increased during better economic conditions. In our study, age was an influential factor for cognitive impairment in PD, in contrast to the findings reported by Arnaldi et al.29 Although age at onset of motor symptom was not a significant influential factor, there are some studies which suggest that late-onset PD is predictive of cognitive impairment.27,30 Medications used to treat PD may affect cognition. But our study, and others,4,28 shows that the levodopa equivalent dose is not associated with the risk of cognitive impairment in PD. Hallucinations15,30,31 and depression26 have also been associated with cognitive impairment in PD patients, but these have different genetic explanations. With regard to hallucinations, one study reported that visual perception is more globally impaired in patients with PD-D than in PD-NoCI or healthy controls.32 A recent publication posited that visual hallucinations are a common feature of disorders that affect temporal lobe structures and impair ascending monoaminergic pathways.33 This provides a reasonable explanation for how visual hallucination is linked to cognitive decline. Depression and symptom severity are associated with cognitive impairment in PD. However, it is thought that the rate and comorbidity of depression and dementia may both be driven by the severity of PD.26,34,35 In summary, potential risk factors for cognitive impairment in the Chinese population are similar to those reported in other races.
However, there are several limitations to our study. All of our subjects were recruited consecutively from the outpatient departments of selected centers; the population may have not included some bedridden or severely disabled PD patients. With regard to the related risk factors for cognitive impairment in PD, we did not include disease comorbidity, medications other than for treatment of PD, or family history. Further studies should follow this cohort to explore cumulative prevalence and related risk factors, and focus on the progression and prognosis of cognitive impairment in PD.
In conclusion, the study aimed to explore the point prevalence and cognitive impairment profile in Chinese PD patients. Notably, it was found that MoCA is a more sensitive tool than the MMSE for identifying cognitive impairment in this population.
Acknowledgement
This study was supported by Peking Union Medical College Hospital (grant no 2006332).
Disclosure
The authors declare that they have no competing interests.
References
Aarsland D, Beyer MK, Kurz MW. Dementia in Parkinson’s disease. Curr Opin Neurol. 2008;21(6):676–682. | |
Emre M, Aarsland D, Brown R, et al. Clinical diagnostic criteria for dementia associated with Parkinson’s disease. Mov Disord. 2007;22(12):1689–1707; quiz 1837. | |
Barone P, Aarsland D, Burn D, Emre M, Kulisevsky J, Weintraub D. Cognitive impairment in nondemented Parkinson’s disease. Mov Disord. 2011;26(14):2483–2495. | |
Wu Q, Chen L, Zheng Y, et al. Cognitive impairment is common in Parkinson’s disease without dementia in the early and middle stages in a Han Chinese cohort. Parkinsonism Relat Disord. 2012;18(2):161–165. | |
Nie K, Zhang Y, Wang L, et al. A pilot study of psychometric properties of the Beijing version of Montreal Cognitive Assessment in patients with idiopathic Parkinson’s disease in China. J Clin Neurosci. 2012;19(11):1497–1500. | |
Chen W, Xu ZM, Wang G, Chen SD. Non-motor symptoms of Parkinson’s disease in China: a review of the literature. Parkinsonism Relat Disord. 2012;18(5):446–452. | |
Goetz CG, Poewe W, Rascol O, et al. Movement Disorder Society Task Force report on the Hoehn and Yahr staging scale: Status and recommendations The Movement Disorder Society Task Force on rating scales for Parkinson’s disease. Mov Disord. 2004;19(9):1020–1028. | |
Fahn S, Elton RL, UPDRS program members. Uni?ed Parkinsons Disease Rating Scale. In: Fahn S, Marsden CD, Goldstein M, Calne DB, editors. Recent Developments in Parkinsons Disease. Vol 2. Florham Park, NJ: Macmillan Healthcare Information; 1987:153–163. | |
Nasreddine ZS, Phillips NA, Bedirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695–699. | |
Dubois B, Burn D, Goetz C, et al. Diagnostic procedures for Parkinson’s disease dementia: recommendations from the movement disorder society task force. Mov Disord. 2007;22(16):2314–2324. | |
Balzer-Geldsetzer M, Costa AS, Kronenburger M, et al. Parkinson’s disease and dementia: a longitudinal study (DEMPARK). Neuroepidemiology. 2011;37(3–4):168–176. | |
Aarsland D, Bronnick K, Williams-Gray C, et al. Mild cognitive impairment in Parkinson disease: a multicenter pooled analysis. Neurology. 2010;75(12):1062–1069. | |
de Lau LM, Schipper CM, Hofman A, Koudstaal PJ, Breteler MM. Prognosis of Parkinson disease: risk of dementia and mortality: the Rotterdam Study. Arch Neurol. 2005;62(8):1265–1269. | |
Riedel O, Klotsche J, Spottke A, et al. Frequency of dementia, depression, and other neuropsychiatric symptoms in 1,449 outpatients with Parkinson’s disease. J Neurol. 2010;257(7):1073–1082. | |
Aarsland D, Andersen K, Larsen JP, Lolk A, Kragh-Sorensen P. Prevalence and characteristics of dementia in Parkinson disease: an 8-year prospective study. Arch Neurol. 2003;60(3):387–392. | |
Hely MA, Reid WG, Adena MA, Halliday GM, Morris JG. The Sydney multicenter study of Parkinson’s disease: the inevitability of dementia at 20 years. Mov Disord. 2008;23(6):837–844. | |
Caviness JN, Driver-Dunckley E, Connor DJ, et al. Defining mild cognitive impairment in Parkinson’s disease. Mov Disord. 2007;22(9):1272–1277. | |
Sahadevan S, Saw SM, Gao W, Tan L, Chin JJ, Venketasubramanian N. Ethnic differences in Singapore’s dementia prevalence: the stroke, Parkinson’s disease, epilepsy, and dementia in Singapore study. J Am Geriatr Soc. 2008;56(11):2061–2068. | |
Mamikonyan E, Moberg PJ, Siderowf A, et al. Mild cognitive impairment is common in Parkinson’s disease patients with normal Mini-Mental State Examination (MMSE) scores. Parkinsonism Relat Disord. 2009;15(3):226. | |
Noe E, Marder K, Bell KL, Jacobs DM, Manly JJ, Stern Y. Comparison of dementia with Lewy bodies to Alzheimer’s disease and Parkinson’s disease with dementia. Mov Disord. 2004;19(1):60–67. | |
Muslimovicć D, Post B, Speelman JD, Schmand B. Cognitive profile of patients with newly diagnosed Parkinson disease. Neurology. 2005;65(8):1239–1245. | |
Bronnick K, Emre M, Lane R, Tekin S, Aarsland D. Profile of cognitive impairment in dementia associated with Parkinson’s disease compared with Alzheimer’s disease. J Neurol Neurosurg Psychiatry. 2007;78(10):1064–1068. | |
Jellinger K. Post mortem studies in Parkinson’s disease – is it possible to detect brain areas for specific symptoms? J Neural Transm Suppl. 1999;56:1. | |
Marder K. Cognitive impairment and dementia in Parkinson’s disease. Mov Disord. 2010;25(S1):S110–S116. | |
Poletti M, Frosini D, Pagni C, et al. Mild cognitive impairment and cognitive-motor relationships in newly diagnosed drug-naive patients with Parkinson’s disease. J Neurol Neurosurg Psychiatry. 2012;83(6):601–606. | |
Riedel O, Klotsche J, Spottke A, et al. Cognitive impairment in 873 patients with idiopathic Parkinson’s disease. J Neurol. 2008;255(2):255–264. | |
Hobson P, Meara J. Risk and incidence of dementia in a cohort of older subjects with Parkinson’s disease in the United Kingdom. Mov Disord. 2004;19(9):1043–1049. | |
Uc E, McDermott M, Marder K, et al. Incidence of and risk factors for cognitive impairment in an early Parkinson disease clinical trial cohort. Neurology. 2009;73(18):1469–1477. | |
Arnaldi D, Campus C, Ferrara M, et al. What predicts cognitive decline in de novo Parkinson’s disease? Neurobiol Aging. 2012;33(6):1127. e11–20. | |
Liu J, Li X, Ye J, et al. Cognitive impairments in Parkinson’s disease. Aging Ment Health. 2012;16(4):529–536. | |
Galvin JE, Pollack J, Morris JC. Clinical phenotype of Parkinson disease dementia. Neurology. 2006;67(9):1605–1611. | |
Mosimann UP, Mather G, Wesnes K, O’brien J, Burn D, McKeith I. Visual perception in Parkinson disease dementia and dementia with Lewy bodies. Neurology. 2004;63(11):2091–2096. | |
Botha H, Carr J. Attention and visual dysfunction in Parkinson’s disease. Parkinsonism Relat Disord. 2012;18(6):742–747. | |
Ziropadja L, Stefanova E, Petrovic M, Stojkovic T, Kostic V. Apathy and depression in Parkinson’s disease: The Belgrade PD study report. Parkinsonism Relat Disord. 2012;18(4):339–342. | |
Liu Z, Chen H. Apathy and related factors in patients with Parkinson’s disease. Chinese J Neurol. 2012;45(6):373–376. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.