Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 14
The Diagnostic Value of Serum Ferritin for Telogen Effluvium: A Cross-Sectional Comparative Study
Authors Cheng T , Fang H, Wang Y, Wang Y, Yang Z , Wu R, Yang D
Received 16 November 2020
Accepted for publication 18 January 2021
Published 10 February 2021 Volume 2021:14 Pages 137—141
DOI https://doi.org/10.2147/CCID.S291170
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Jeffrey Weinberg
Tiantian Cheng,1,2 Huijuan Fang,1,2 Yongchao Wang,3 Ying Wang,2 Zhishan Yang,1,2 Ruiying Wu,1,2 Dingquan Yang2
1School of Clinical Medicine, Beijing University of Chinese Medicine, Beijing, People’s Republic of China; 2Department of Dermatology, China-Japan Friendship Hospital, Beijing, People’s Republic of China; 3Department of Clinical Laboratory, China-Japan Friendship Hospital, Beijing, People’s Republic of China
Correspondence: Dingquan Yang
Department of Dermatology, China-Japan Friendship Hospital, Beijing, People’s Republic of China
Tel +86 13901218671
Email [email protected]
Purpose: This study aimed to explore the relationship between serum ferritin levels and telogen effluvium.
Patients and Methods: A total of 193 telogen effluvium patients and 104 female androgenetic alopecia patients were included. We collected the test result of serum ferritin levels, compared with the results of 183 healthy subjects. Receiver Operator Characteristic curves were generated to assess the potential diagnostic value of serum ferritin in telogen effluvium patients.
Results: The serum ferritin in telogen effluvium patients were significantly lower than that in the healthy control group (P = 0.000) or female androgenetic alopecia patients (P =0.000). Patients with lower serum ferritin levels got high odds to have telogen effluvium. The areas under the Receiver Operator Characteristic curve of serum ferritin levels were 0.735 and 0.645 for distinguishing telogen effluvium patients from healthy control subjects or female androgenetic alopecia patients.
Conclusion: Serum ferritin could be a potential biomarker for clinical diagnosis of telogen effluvium.
Keywords: serum ferritin, telogen effluvium, female androgenetic alopecia, ROC curve
Introduction
Telogen effluvium (TE) is common alopecia in women, characterized by diffuse hair loss. TE used to occur in 2–3 months after the surgery, weight loss, high fever, thyroid disease, and other trigger events.1 Female androgenic alopecia (FAGA) also showed diffuse alopecia.2 Currently, TE diagnosis mainly relies on clinical investigation and dermoscopy, and there is no reliable laboratory indicator for clinical diagnosis.
Iron is involved in DNA synthesis. In the bulge region, the expression of CDC2, NDRG1, ALAD, and RRM2 up-regulated, while Decorin and DCT decreased, and all of them are regulated by iron.3,4 Serum ferritin levels are often used to determine iron reserves.6 Iron deficiency has a statistical correlation with emotional stress;5 stress and anxiety also a common trigger for hair loss.
A series of studies has reported serum iron deficiency in the TE patients, though some other studies observed no correlation between iron deficiency and TE incidence. The relationship between serum ferritin and alopecia is still controversial.7–10 The primary limitation for the contradictory conclusions of past studies is data bias or small sample size. Cheung et al’s studies involved a large sample size but they used a hypothetical cut-off value from prior experience, which undermined the validity of their conclusions.8 Furthermore, the cut-off value in their study was determined based on experience from previous studies. In this research, we tested the serum ferritin levels among TE, FAGA patients, and healthy controls. Our study showed serum ferritin could be used as a diagnostic indicator to distinguish TE patients from FAGA patients or healthy people.
Patients and Methods
This study included 264 TE patients and 124 female androgenetic alopecia patients who presented to our dermatology department from January 2020 to September 20, 2020, from the China-Japan friendship hospital. All patients were independently evaluated by four doctors and diagnosed based on clinical history, physical, and dermoscopy. FAGA was diagnosed based on hair diameter density >20%. Clinical history of diffuse shedding and positive pull test were used to diagnosis of TE.11 Female patients aged 15–40 years old with TE or androgenetic alopecia were involved in this study. Patients with history of iron, folic acid, vitamin supplements within the past 3 months, with endocrine abnormalities, kidney diseases, metabolic diseases, hematological system diseases, gastrointestinal diseases, or cancer etc were excluded from this study. A control group consisted of 183 healthy women of the same age range were recruited in the Physical Examination Center of China-Japan Friendship Hospital. Patients were fast for 12 h before serum collection. Serum ferritin levels were measured using chemiluminescence method in the Beckman DXI800 (Beckman Coulter Unicel DXI800, CA, USA) in 2 h after sample collection, with a normal reference range of 11 ng/mL - 306.8 ng/mL.
Sample size was calculated by Power Analysis and Sample Size Calculation Statistical Software V.11.0 based on the results of the pre-experiment of this study. We used Shapiro–Wilk test to check normal distribution of data in different groups. The differences of serum ferritin between groups were compared with independent t-tests, one-way ANOVA, post hoc test, or chi-square test. We used binary logistic regression to determine the risk of iron deficiency leading to TE. Receiver Operator Characteristic curves were generated and the area under the curve was calculated to assess the diagnostic accuracy of serum ferritin. P-value < 0.05 indicated statistical significances. All statistical analyses were performed using SPSS version 26, SPSS Inc. The research was approved by the Ethics Committee for Clinical Research of China-Japan Friendship Hospital (No.2018-160-K116). Our study was conducted in accordance with the Declaration of Helsinki. Patient consents were waived as the study did not collect information regarding patient privacy and identifiable features.
Results
We involved 193 TE patients,104 FAGA patients, and 183 healthy people in our study (Figure 1). The average age of TE, FAGA, and healthy people were 28.37±5.11 years, 28.91±5.08 years, 29.11±5.46 years, respectively (F=1.002, P=0.368). Physical/emotional stress and nutritional deficiencies (losing weight) were the most common causes of TE with a rate of 64.2%. More clinical information on involved patients is shown in Supplementary Tables 1s and 2s. Serum ferritin was 24.27±17.11 (1.6~91.9) ng/mL, 40.40±35.43 (5.4~203) ng/mL, 45.55±37.88 (3.3~272.9) ng/mL in the three groups, respectively. The levels of serum ferritin differed significantly (F=24.166, P=0.000) in the three groups. The mean serum ferritin levels in TE patients were significantly lower than that in healthy controls (t=6.975, P=0.000) or FAGA patients (t=4.378, P=0.000); no significant difference in the serum ferritin was observed between FAGA patients and healthy people (t=1.132, P=0.259) (Table 1) through independent t-tests. Meanwhile, post hoc test also got similar results (Supplementary Table 3s). Interestingly, we found that if serum ferritin levels< 20 ng/mL, the number of FAGA patients was significantly different from that of healthy people (X2=11.27, P=0.001). There is no difference in the number of patients with serum ferritin ≤ 30 ng/mL between FAGA and healthy controls (X2=1.05, P=0.305) (Table 2). Binary logistic regression analysis showed that lower serum ferritin levels correlated with high incidence of TE occurrence (Table 3).
 |
Figure 1 Flowchart of patients inclusion and exclusion. |
 |
Table 1 Mean Serum Ferritin Levels for Each Group |
 |
Table 2 The Number of Patients in Different Serum Ferritin Levels |
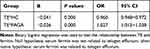 |
Table 3 Binary Logistic Regression Analysis Between Telogen Effluvium and Ferritin |
A ROC curve was generated using the serum ferritin levels from TE patients and healthy people, with an AUC of 0.735. The optimum cut-off value of 24.45 ng/mL was obtained from the ROC curve, from which the sensitivity, specificity, precision, and accuracy were calculated to be 64.2% and 73.8%, 0.721%, 0.689%, respectively (Figure 2). We further generated a ROC curve using the ferritin levels from TE patients and FAGA patients, which showed the AUC, sensitivity, specificity, precision, and accuracy was 0.645, 64.2%, 59.6%, 74.7%, 62.6%, based on the optimum cut-off value 24.6 ng/mL (Figure 2).
Discussion
It is well known that iron plays an important role in hair follicles. Iron levels in the human body could be obtained from a variety of food sources, especially from animal-derived food. The mechanism of iron excretion is complicated and the potential routes of excretion include sweat, menstruation, shedding of hair and skin cells, and through rapid turnover and excretion of enterocytes.12,13 Menstruation was thought to be a primary cause of iron deficiency in women. The cut-off value of serum ferritin for iron deficiency diagnosis was 40 ng/mL, with a sensitivity and specificity of 98%.3 In our study, the average serum ferritin level of healthy people was 45.55 ng/mL, which denied menstrual cause in the TE occurrence. The average serum ferritin level in patients with TE was 24.27ng/mL, and the optimum cut-off value was 24.45ng/mL, which were significantly lower than that in healthy subjects. The low levels of serum ferritin could be attributed to the following causes: (1) High fever with inflammatory, surgery or postpartum hemorrhage, emotional/physical stress, and malignant tumors cause abnormal iron metabolism, inducing iron deficiency, which undermined the TE.12,13 (2) Insufficient iron uptake from diet may lead to serum ferritin level below normal range. The development of vegan diet for weight-loss have reduced the source of iron intake, which is another cause of TE.14,15 However, many other factors also influenced the hair follicle cycle, such as anticoagulants, anticonvulsants, or high-dose contraceptives, etc.1 Our study showed that serum ferritin could be used as a biomarker for TE diagnosis with sensitivity and specificity of 64.2% and 73.8%, respectively. When using serum ferritin to differentiate TE and FAGA, the sensitivity and specificity was 64.2%, and 59.6%, which may need other biomarkers to improve its diagnosis value.
In this research, we generated a ROC curve to evaluate the diagnostic value of serum ferritin in distinguishing TE patients from FAGA patients or healthy people for the first time. Compared to previous study, the data are more representative for its larger sample size and healthy people as the control group. Meanwhile, the cut-off value was selected based on ROC curve. Normal serum ferritin levels ranged from 30 to 300 ng/mL for men and 10 to 200 ng/mL for women.12,13 The optimum cut-off value of our study is in the normal range. However, low serum ferritin levels indicated high risk of TE. Serum ferritin may have a potential value in the prevention of TE. Thus, Iron supplements in people with serum ferritin levels below 24.45ng/mL may benefit TE prevention.
The number of patients with serum ferritin < 20 ng/mL in FAGA group was significantly different from that in healthy controls, but not when serum ferritin < 30 ng/mL. A lower serum ferritin level indicated more severe iron deficiency, which may affect hair follicle matrix cells and lead hair shedding. In our study, the optimum cut-off value of serum ferritin to distinguish TE from FAGA was 24.6 ng/mL, with a low specificity of 59.6%. Thus, it is necessary to incorporate other biomarkers, such as serum testosterone and estradiol, to improve the specificity.
Our research had some limitations; most of the involved patients come from one city and the retrospective collection of clinical data may introduce bias. Our study had involved over 100 TE patients, as well as similar number of FAGA patients and healthy controls to strengthen the conclusion. Though the sample size was relatively large, we still cannot eliminate the bias concerns. A prospective, multiple-center study is needed to prove the potential value of serum ferritin in the diagnosis of TE. Furthermore, low levels of serum ferritin do not reflect low iron in the body (serum iron, total iron-binding capacity, etc.). To clarify the relationship between serum ferritin and TE occurrence, we need to include a series of factors, such as genetic background, total iron-binding capacity, hemoglobin, and other iron-related indicators into consideration.
Conclusion
In conclusion, we performed a retrospective study to explore the value of serum ferritin in TE patient diagnosis. There is a significant difference in serum ferritin levels between TE patients and healthy people or FAGA patients, and the lower the serum ferritin level was, the more likely a patient suffered from TE. The ROC curve also showed that serum ferritin could be used to distinguish TE patients from healthy subjects or FAGA patients. The findings of our study deepened our understanding of the diagnostic value of serum ferritin in telogen effluvium patients.
Acknowledgments
We would like to especially thank Dc. Xiaohui Zou in the Laboratory of Microbiology and Infection, China-Japan Friendship Hospital for his valuable advice and criticism. Our team is mainly engaged in dermatological diseases including alopecia areata, androgenic alopecia, and telogen effluvium et al.
Funding
This study was supported by the National Natural Science Foundation of China (No. 81973691).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Mysore V, Parthasaradhi A, Kharkar RD, et al. Expert consensus on the management of Telogen Effluvium in India. Int J Trichology. 2019;11(3):107–112. doi:10.4103/ijt.ijt_23_19
2. Carmina E, Azziz R, Bergfeld W, et al. Female pattern hair loss and androgen excess: a report from the multidisciplinary androgen excess and PCOS committee. J Clin Endocrinol Metab. 2019;104(7):2875–2891. doi:10.1210/jc.2018-02548
3. Sant’Anna Addor FA, Donato LC, Melo CSA. Comparative evaluation between two nutritional supplements in the improvement of telogen effluvium. Clin Cosmet Investig Dermatol. 2018;11:431–436. doi:10.2147/CCID.S173082
4. Almohanna HM, Ahmed AA, Tsatalis JP, Tosti A. The role of vitamins and minerals in hair loss: a review. Dermatol Ther (Heidelb). 2019;9(1):51–70. doi:10.1007/s13555-018-0278-6
5. Malkud S. A hospital-based study to determine causes of diffuse hair loss in women. J Clin Diagn Res. 2015;9(8):WC01–4. doi:10.7860/JCDR/2015/14089.6170
6. Camaschella C. Iron deficiency. Blood. 2019;133(1):30–39. doi:10.1182/blood-2018-05-815944
7. Rasheed H, Mahgoub D, Hegazy R, et al. Serum ferritin and vitamin d in female hair loss: do they play a role? Skin Pharmacol Physiol. 2013;26(2):101–107. doi:10.1159/000346698
8. Cheung EJ, Sink JR, English Iii JC. Vitamin and mineral deficiencies in patients with Telogen Effluvium: a retrospective cross-sectional study. J Drugs Dermatol. 2016;15(10):1235–1237.
9. Bhat YJ, Saqib NU, Latif I, Hassan I. Female pattern hair loss-an update. Indian Dermatol Online J. 2020;11(4):493–501. PMID: 32832434; PMCID: PMC7413422. doi:10.4103/idoj.IDOJ_334_19.
10. Miniaci MC, Irace C, Capuozzo A, et al. Cysteine prevents the reduction in keratin synthesis induced by iron deficiency in human keratinocytes. J Cell Biochem. 2016;117(2):402–412. doi:10.1002/jcb.25286
11. Olsen EA, Reed KB, Cacchio PB, Caudill L. Iron deficiency in female pattern hair loss, chronic telogen effluvium, and control groups. J Am Acad Dermatol. 2010;63(6):991–999. doi:10.1016/j.jaad.2009.12.006
12. Ems T, St Lucia K, Huecker MR. Biochemistry, Iron Absorption. StatPearls. StatPearls Publishing Copyright © 2020, StatPearls Publishing LLC.; 2020.
13. Wang W, Knovich MA, Coffman LG, Torti FM, Torti SV. Serum ferritin: past, present and future. Biochim Biophys Acta. 2010;1800(8):760–769. doi:10.1016/j.bbagen.2010.03.011
14. Calder RK, Mussap AJ. Factors influencing women’s choice of weight-loss diet. J Health Psychol. 2015;20(5):612–624. doi:10.1177/1359105315573435
15. Goette DK, Odom RB. Alopecia in crash dieters. JAMA. 1976;235(24):2622–2623. doi:10.1001/jama.1976.03260500038026
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

