Back to Journals » Clinical Ophthalmology » Volume 13
Architecture evaluation of the main clear corneal incisions in femtosecond laser-assisted cataract surgery by optical coherence tomography imaging
Authors Chaves MAPD , de Medeiros AL, Vilar CMC, Magalhães KRP, Gonçalves MR, Tzelikis PFM , Hida WT , Carricondo PC , Alves MR
Received 16 August 2018
Accepted for publication 14 December 2018
Published 14 February 2019 Volume 2019:13 Pages 365—372
DOI https://doi.org/10.2147/OPTH.S184024
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Scott Fraser
Mario Augusto Pereira Dias Chaves,1–3 André Lins de Medeiros,1,2 César Martins Cortez Vilar,1,2,4 Klayne Rafaella Pereira Magalhães,2 Michelle Rodrigues Gonçalves,3 Patrick Frenzel de Moraes Tzelikis,1,2,5 Wilson Takashi Hida,1,2,5,6 Pedro Carlos Carricondo,2,5 Milton Ruiz Alves2,5
1Brasília Ophthalmologic Hospital (HOB), Department of Cataract, João Pessoa, Paraíba, Brazil; 2Renato Ambrosio Ophthalmologic Study Center (CEORA), Brasília, Federal District, Brazil; 3Provisão Paraíba Ophthalmologic Hospital, João Pessoa, Paraíba, Brazil; 4Cullen Eye Institute, Department of Ophthalmology, Baylor College of Medicine, Houston, TX, USA; 5São Paulo University School of Medicine (FM-USP), Department of Ophthalmology, São Paulo, Brazil; 6São Paulo Federal University of School of Medicine (UNIFESP), Department of Ophthalmology, São Paulo, Brazil
Purpose: To assess the stability and reliability of femtosecond laser-assisted cataract surgery (FLACS) incisions design and dimensions using anterior segment optical coherence tomography (AS-OCT) imaging.
Setting: Renato Ambrósio Ophthalmologic Study Center from Hospital Oftalmológico de Brasília, Brasília, Brazil.
Design: Prospective nonrandomized controlled case series.
Methods: Eyes undergoing FLACS with triplanar main temporal clear corneal incision (CCI) were evaluated at the end of the case. Eyes that required any incision hydration, surgical complications or lacked follow-up were excluded. The AS-OCT was performed after femtosecond delivery; at the end of the case; at 1 day and at 30 days after surgery. Data of pachymetry, endothelial and epithelial gaps, Descemet detachment and CCI architecture were compared.
Results: Eleven eyes from 11 patients completed follow-up. Corneal thickness was statistically different between after femtosecond delivery and the end of the case (P-value <0.05), but without difference compared to 30 days evaluation. There was an increase of Descemet detachments (P-value <0.05) and endothelial gaps (P-value =0.0133) at the end of the case compared to post-femtosecond delivery. As for the architecture of the CCI, significant difference was found between the parameters of entry angle and exit angle measured with AS-OCT and the programmed.
Conclusion: The AS-OCT was capable of visualizing changes in the cornea at the CCI. Despite the stress caused by manipulation, results indicated good stability of incision and reproducibility of tunnel length.
Synopsis: Difference of corneal thickness at the CCI between after femtosecond and after phacoemulsification measurements (P-value <0.05), with increase of endothelial gaps (P-value =0.0133) and Descemet detachments (P-value <0.05).
Keywords: femtosecond, incisions, cataract, pachymetry, optical coherence tomography
Introduction
The evolution of the surgical incision was imperative for the success of the phacoemulsification (phaco). Small and sutureless clear corneal incisions (CCI) have become the standard adopted by most surgeons in the world due to its smaller risk of endophthalmitis, less surgically induced astigmatism and consequently better visual results in the immediate postoperative period.1–9
Previous studies have shown that triangular and square-shaped incisions are safe and self-sealing. However, reproducing the same pattern of triangular or square incisions can be a challenge even for experienced surgeons. CCIs are usually performed manually with the aid of angled steel or other cutting material keratomes with predetermined width. The result is susceptible to the amount of stretching in the corneal tissue during incision, generating tunnel extensions inferior to the one intended and variations in the angles of entrance and exit and tunnel length.3,6,7,9–16
In the pursuit for perfection in cataract surgery, the femtosecond (femto) laser technology was incorporated and became adopted in many practices around the world. The theoretical advantages of the technology include high precision and reproducibility in corneal incision, capsulotomy and nucleus fracture. The ultra-short pulse laser causes photo disruption to separate tissue. It can be focused very accurately at different depths of the corneal and lens tissue using anterior segment optical coherence tomography (AS-OCT) for image guiding.2,4,5,9,12,17–21
Still little is known about the healing process and the effect on the adjacent tissue of operative wounds made by the femtosecond lasers. Some studies suggest great precision and safety in performing the incisions while others report the appearance of complications previously uncommon in the traditional manual technique.2–4,20,21
A possible way to evaluate the in vivo corneal changes from the application of the femtosecond laser to perform CCIs is through AS-OCT. Some previous studies showed that it is possible to precisely assess the architecture of the incisions made with both keratomes and femtosecond laser using this technology in a noninvasive manner. However, most of the studies evaluate the results after the end of the case when incision manipulation and stromal hydration have been performed and therefore may influence the results.14,18,22–26
In this study, we intended to evaluate the femtosecond laser incisions and corneal changes immediately after laser delivery and observe the changes in corneal architecture shortly after surgery.
Patients and methods
This study was conducted according with the principles of the Declaration of Helsinki and was approved by the Institutional Review Board of the Hospital Oftalmológico de Brasília, Brazil. The study was a prospective, comparative, controlled, nonrandomized case series. Patients were selected at the cataract outpatient clinic of the Hospital Oftalmológico de Brasília and underwent femtosecond laser-assisted cataract surgery (FLACS) using the LenSx System (Alcon Laboratories, Inc., Fort Worth, TX, USA) between January 2015 and March 2015, performed by a single experienced surgeon (WTH) using topical anesthesia.
All patients provided written informed consent and underwent complete pre and postoperative ophthalmologic assessment, including measurement of uncorrected (UDVA) and corrected (CDVA) visual acuity for far with early treatment diabetic retinopathy study charts using the logarithm of the minimal angle resolution notation; corneal aberrometry (OPD-Scan III, Nidek Co. Japan), Scheimpflug corneal tomography (Oculus Pentacam®, Wetzlar, Germany) and endothelial cell counts (CellCheck Xl Specular Microscope, Konan Medical USA, Inc., Irvine, CA, USA). The exclusion criteria were presence of corneal diseases and opacities; previous ocular surgeries; intraoperative complications or during laser delivery; stromal hydration of the incision at the end of surgery. For incision evaluation, a Spectrum domain OCT with anterior segment module was used (Spectralis®, Heidelberg Engineering Co., Heidelberg, Germany).
Among 20 eyes of 20 patients selected for the trial, 2 eyes of 2 patients were excluded because they had had incision hydration performed at the end of the case and 7 eyes of 7 patients were also excluded for missing one or more postoperative examinations. At the end, 11 eyes from 11 patients met inclusion and exclusion criteria and performed the complete follow-up of postoperative examinations up to 30 days after surgery with the same topical drug regimen.
The main incision was located temporally and programmed to be triplanar with the following characteristics: 2.5 mm wide, 1.8 mm tunnel extension, 90° anterior cutting angle, 80° posterior cutting angle, 20° lamellar cutting angle at 90% depth of the corneal thickness. The energy parameter was 5.8 μJ, 4 μm of spot separation and 3 μm of layer separation. After the success of the patient interface coupling process and confirmation of parameters and image with the AS-OCT, the laser was applied to the patient.
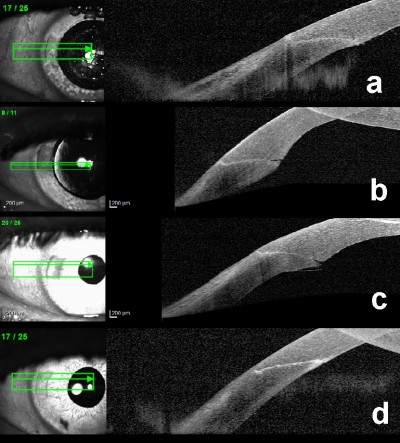
The evaluation of the main corneal incision was made with the AS-OCT immediately after the delivery of the femtosecond laser, prior to any manipulation of the incision by the surgeon (after femto). Three other AS-OCT evaluations were performed: immediately after the end of the surgery (after phaco); first day postoperative (first postop) and after 30 days postoperative (30th postop), Figure 1.
Corneal pachymetry was measured through the AS-OCT instrument’s software and checked through graphic analysis software (Adobe Photoshop CS 2015). The methodology for pachymetric evaluation of the incisions was similar to the study conducted by Lee et al.24 Corneal pachymetry (distance between epithelium and corneal endothelium in a line perpendicular to the corneal surface) was analyzed in the image: at the midpoint of the tunnel of the incision, 1 mm toward the center of the cornea from the midpoint (nasal point) and 1 mm towards the limbus from the midpoint (temporal point). The pachymetric changes at different periods in every point were analyzed and compared (Figure 2).
  | Figure 2 Points of pachymetry measurements at corneal wound. |
We also analyzed the reproducibility of the incisions comparing the data of the cornea entry angle, the incisional tunnel angle in relation to the tangential plane of the corneal surface and the angle of entry in the anterior chamber, hereafter referred as incision exit angle. The same graphic analysis software was used to measure the angles (Figure 3).
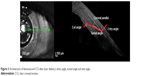  | Figure 3 Architecture of femtosecond CCI after laser delivery: entry angle, tunnel angle and exit angle. |
Complementing the evaluation of the AS-OCT images, we documented the prevalence of epithelial failures at the entrance of the incision (epithelial gaps), endothelial failures at the entrance of the anterior chamber (endothelial gaps) and Descemet’s membrane detachments.
Statistical analysis was performed using software R (R version 3.4.1, Vienna, Austria). In the initial phase of the analysis, the Student’s t-test (for two paired samples) was used to compare pachymetry parameters at each time interval in the measurements comparing averages with normal distribution. To verify the normality of the data, the Shapiro–Wilk test was used. For the nominal variables present in the study (epithelial gaps, endothelial gaps and Descemet detachments), the chi-square test for proportions was used.
Ethics statement
This study was approved by the research ethics committee of the Hospital Oftalmológico de Brasília Institutional Review Board, Brasilia, Brazil.
Results
Both UDVA and CDVA had statistically significant improvement (P <0.05) when comparing preoperative and postoperative measurements Table 1.
The preoperative endothelial cell count (mean =2,513 cells/mm2) for the total recovery period (mean =1,969) had a mean reduction evaluated at 22%.
Pachymetric evolution
The corneal thickness was measured at three points (midpoint, nasal point and temporal point). Immediately after femto, the midpoint was, on average, 704 μm; the nasal point 667 μm and the temporal point 745 μm. After phaco, the measurements, on average, were 948 μm, 902 μm, 858 μm, respectively. During the first postop, the mean point had, on average, 901 μm, the nasal point 901 μm and the temporal point 783 μm; and during 30th day postop, these points measured, 691 μm, 661 μm and 731 μm, on average, respectively Figure 4.
  | Figure 4 Pachymetry progression at each moment of evaluation. |
It was found that there is a statistically significant difference in the corneal thickness between corneal thickness after femto and after phaco for the three points (P <0.05). No significant difference was observed after phaco when compared with the first postop, unless for the temporal point. Between the first postop and the 30th day postop, there were significant differences in the nasal and midpoints Tables 2 and 3.
  | Table 2 Comparison of pachymetry in three different points between the four moments of evaluation |
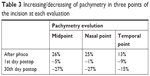  | Table 3 Increasing/decreasing of pachymetry in three points of the incision at each evaluation |
Figure 5 shows the occurrence of endothelial gaps, epithelial gaps and Descemet detachments for every moment. There was a significant increase in the incidence of Descemet detachments (P-value <0.05) and endothelial gaps (chi-squared test, P-value =0.0133), which increases in 72% of cases from after femto to after phaco, followed by gradual reduction in the 1st day postop and 30th postop Figure 5.
  | Figure 5 Endothelial gaps, epithelial gaps and Descemet detachments incidences. |
Four measures related to the incisions were observed after femto: the angle of entry, the angle of exit, the angle of the tunnel and the tunnel length. Such measurements were compared to the standard programmed values, where the input angle measures 90°, the output angle measures 80°, the tunnel angle measures 20°, and length 1,800 μm. There was a statistically significant difference in only two of the measurements (entry angle and exit angle) from the standard values. For the other parameters (tunnel angle and length), there was no significant difference (Table 4).
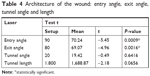  | Table 4 Architecture of the wound: entry angle, exit angle, tunnel angle and length |
Discussion
To our knowledge, this is the first study that evaluated the architecture of the corneal incisions before any manipulation by the surgeon allowing a true comparison of the incisional architecture delivered with what was programmed. Moreover, it eliminates distortions in the evaluation of the incision due to manipulation.
By evaluating UDVA and CDVA data, there was a significant improvement in the vision of patients submitted to FLACS, suggesting that the surgical technique used was effective for visual recovery as suggested in most of the other studies.
There was an average 22% endothelial loss rate observed in the 30th day postop specular microscopy assessment. A further comparative study of cases without the use of FLACS is necessary to assess whether this result is greater than expected or not. Some studies suggest that after a longer postoperative period these differences would not be significant.1,2,19
Based on our inclusion criteria, we selected only those cases that at the end of the surgery did not require stromal hydration for incisional sealing, with no apparent leakage. The goal was to measure how much of the pachymetric changes and other findings were related to laser application or to surgical manipulation. Cases in which stromal hydration was required for incision sealing at the end of the surgery were excluded. It would be necessary to perform the evaluation through AS-OCT before and after hydration to conclude whether those cases that did not require hydration reached sealed incision status due to better incision architecture or due to less manipulation of the incision, but this is not practical in a clinical setting.
When analyzing the pachymetry at the three points measured for each moment, we observed that there was no significant difference between the analysis of the incisions immediately after femto and the 30-day postop evaluation. This suggests that the laser delivery alone does not cause significant damage to the cornea other than the incision path. However, there was a significant difference between corneal pachymetry after femto and after phaco. Considering that the incisions evaluated were not submitted to stromal hydration, this evaluation suggests that structural changes observed in the pachymetry of the three points analyzed, as well as perilesional edema, were related more to manipulation of the incision by the surgeon than by the laser thermal damage.
In our study, we found an occurrence of endothelium gaps of 18% immediately after the application of the laser and of more than 91% after phaco, reducing to 82% on the first day postop and to 55% for the 30-day evaluation. We believe that the high endothelial gap incidence was due to manipulation of the incision at the end of the surgery. Mastropasqua et al in a comparative study between manual and laser incisions found a prevalence of 85.7% for 1 day from surgery and 36.7% for 30 days using femtosecond incisions.18 Wang et al found similar prevalence with one day of surgery (85.7%), but found better results for 1 to 3 months postoperatively (31.8%) using a steel blade and no evidence of endothelial gap after 3 months and high values of stromal hydration, while our study found a similar result for 1 day of surgery without performing any hydration.27 Calladine and Packard found even better values for the incidence of endothelial gaps, 41%, after 1 hour of surgery and suggested that it was more linked to increased intraocular pressure, while Xia et al found incidence of 70% for 1 day of surgery.28,29 Torres et al found smaller values in their study, with 25% endothelial gaps.8
We observed the same prevalence of 27% of epithelial gaps for the first three moments of observation of the AS-OCT image: after femto; after phaco and first day postop. However, only 9% presented this finding after femto which suggests that the manipulation may play an important role for the appearance of epithelial gaps. Other studies have found similar results.18,29,30 In a study by Alio et al with 20 eyes, no epithelial gap was reported.14
In our study, we did not find any case of Descemet detachment immediately after the femtosecond stage, before any manipulation. However, 45% of our cases showed Descemet detachment immediately after phaco, which suggests that this event is more related to the manipulation of the incision than to the femtosecond laser delivery. No Descemet detachments were present at the 30th day postop. None of the Descemet detachment cases were detected in the postoperative slit lamp examination, being observed only through the OCT image. Grewal et al, using spectral domain AS-OCT, observed 0% prevalence for Descemet detachment with 30 days using another laser platform while obtaining 18% prevalence for the same period using a steel blade.15 Xia et al observed a much higher rate of 82% on the first day and associated low intraocular pressure, with manual incisions.29 In our study, all patients at 30 days post-surgery no longer presented visible Descemet detachment in the AS-OCT image.
We found a significant difference between the angles of entry and exit of the incision when compared to the programmed parameters, but the same was not seen for the angle of the tunnel. We hypothesized that the changes in the entrance, tunnel and exit angles were due to the different corneal curvatures or even the docking process, suggesting a rotation of the incision architecture around its own central axis in relation to the plane of the cornea. However, there was no difference in the tunnel length compared to what had been programmed.
Conclusion
In the present study, we conclude that the femtosecond was effective in making clear corneal incisions with desired architecture. The data suggests that the surgical manipulation of the incision was the main cause of the corneal pachymetric changes, endothelial gaps and Descemet detachments.
In agreement with previous studies,8,10,15,22,24,27–30 AS-OCT has proven to be an effective instrument to evaluate the changes in the architecture of the main corneal incisions during the healing process of the surgical wound.
Comparative studies with greater sampling may demonstrate whether the reproducibility of the main incisions delivered by the femtosecond laser could generate better surgical results and lower rates of complications with statistical significance.
Acknowledgments
This paper was presented at the 2017 ASCRS-ASOA Annual Meeting, Los Angeles, CA, USA and awarded best paper at the 2017 Brazilian Ophthalmology Society (SBO) Annual Meeting, Recife, Brazil. This study was supported by the Hospital Oftalmológico de Brasília (HOB) and Centro de Estudos Oftalmológicos Renato Ambrósio (CEORA).
Disclosure
The authors report no conflicts of interest in this work.
References
Abell RG, Kerr NM, Vote BJ. Femtosecond laser-assisted cataract surgery compared with conventional cataract surgery. Clin Exp Ophthalmol. 2013;41(5):455–462. | ||
Dick HB, Schultz T. A review of laser-assisted versus traditional phacoemulsification cataract surgery. Ophthalmol Ther. 2017;6(1):7–18. | ||
Roberts TV, Lawless M, Bali SJ, Hodge C, Sutton G. Surgical outcomes and safety of femtosecond laser cataract surgery: a prospective study of 1500 consecutive cases. Ophthalmology. 2013;120(2):227–233. | ||
Donaldson KE, Braga-Mele R, Cabot F, et al. Femtosecond laser-assisted cataract surgery. J Cataract Refract Surg. 2013;39(11):1753–1763. | ||
Serrao S, Giannini D, Schiano-Lomoriello D, Lombardo G, Lombardo M. New technique for femtosecond laser creation of clear corneal incisions for cataract surgery. J Cataract Refract Surg. 2017;43(1):80–86. | ||
Masket S, Belani S. Proper wound construction to prevent short-term ocular hypotony after clear corneal incision cataract surgery. J Cataract Refract Surg. 2007;33(3):383–386. | ||
Masket S. Is there a relationship between clear corneal cataract incisions and endophthalmitis? J Cataract Refract Surg. 2005;31(4):643–645. | ||
Torres LF, Saez-Espinola F, Colina JM, et al. In vivo architectural analysis of 3.2 mm clear corneal incisions for phacoemulsification using optical coherence tomography. J Cataract Refract Surg. 2006;32(11):1820–1826. | ||
Roberts TV, Lawless M, Chan CC, et al. Femtosecond laser cataract surgery: technology and clinical practice. Clin Exp Ophthalmol. 2013;41(2):180–186. | ||
Fine IH, Hoffman RS, Packer M. Profile of clear corneal cataract incisions demonstrated by ocular coherence tomography. J Cataract Refract Surg. 2007;33(1):94–97. | ||
Teuma EV, Bott S, Edelhauser HF. Sealability of ultrashort-pulse laser and manually generated full-thickness clear corneal incisions. J Cataract Refract Surg. 2014;40(3):460–468. | ||
Bali SJ, Hodge C, Lawless M, Roberts TV, Sutton G. Early experience with the femtosecond laser for cataract surgery. Ophthalmology. 2012;119(5):891–899. | ||
Masket S, Sarayba M, Ignacio T, Fram N. Femtosecond laser-assisted cataract incisions: architectural stability and reproducibility. J Cataract Refract Surg. 2010;36(6):1048–1049. | ||
Alió JL, Abdou AA, Soria F, et al. Femtosecond laser cataract incision morphology and corneal higher-order aberration analysis. J Refract Surg. 2013;29(9):590–595. | ||
Grewal DS, Basti S. Comparison of morphologic features of clear corneal incisions created with a femtosecond laser or a keratome. J Cataract Refract Surg. 2014;40(4):521–530. | ||
Serrao S, Lombardo G, Schiano-Lomoriello D, Ducoli P, Rosati M, Lombardo M. Effect of femtosecond laser-created clear corneal incision on corneal topography. J Cataract Refract Surg. 2014;40(4):531–537. | ||
Friedman NJ, Palanker DV, Schuele G, et al. Femtosecond laser capsulotomy. J Cataract Refract Surg. 2011;37(7):1189–1198. | ||
Mastropasqua L, Toto L, Mastropasqua A, et al. Femtosecond laser versus manual clear corneal incision in cataract surgery. J Refract Surg. 2014;30(1):27–33. | ||
Day AC, Gore DM, Bunce C, Evans JR; Cochrane Eyes and Vision Group. Laser-assisted cataract surgery versus standard ultrasound phacoemulsification cataract surgery. Cochrane Database Syst Rev. 2016;39(9):Cd010735. | ||
Mayer WJ, Klaproth OK, Hengerer FH, et al. In vitro immunohistochemical and morphological observations of penetrating corneal incisions created by a femtosecond laser used for assisted intraocular lens surgery. J Cataract Refract Surg. 2014;40(4):632–638. | ||
Nosé RM, Rivera-Monge MD, Forseto AS, Nosé W. Descemet membrane detachment in femtosecond laser-assisted cataract surgery. Cornea. 2016;35(4):562–564. | ||
Schallhorn JM, Tang M, Li Y, Song JC, Huang D. Optical coherence tomography of clear corneal incisions for cataract surgery. J Cataract Refract Surg. 2008;34(9):1561–1565. | ||
Wang L, Jiang L, Hallahan K, Al-Mohtaseb ZN, Koch DD. Evaluation of femtosecond laser intrastromal incision location using optical coherence tomography. Ophthalmology. 2017;124(8):1120–1125. | ||
Lee H, Kim EK, Kim HS, Kim TI. Fourier-domain optical coherence tomography evaluation of clear corneal incision structure according to blade material. J Cataract Refract Surg. 2014;40(10):1615–1624. | ||
Weikert MP, Wang L, Barrish J, Dimalanta R, Koch DD. Quantitative measurement of wound architecture in microincision cataract surgery. J Cataract Refract Surg. 2012;38(8):1460–1466. | ||
Holladay JT, Moran JR, Kezirian GM. Analysis of aggregate surgically induced refractive change, prediction error, and intraocular astigmatism. J Cataract Refract Surg. 2001;27(1):61–79. | ||
Wang L, Dixit L, Weikert MP, Jenkins RB, Koch DD. Healing changes in clear corneal cataract incisions evaluated using Fourier-domain optical coherence tomography. J Cataract Refract Surg. 2012;38(4):660–665. | ||
Calladine D, Packard R. Clear corneal incision architecture in the immediate postoperative period evaluated using optical coherence tomography. J Cataract Refract Surg. 2007;33(8):1429–1435. | ||
Xia Y, Liu X, Luo L, et al. Early changes in clear cornea incision after phacoemulsification: an anterior segment optical coherence tomography study. Acta Ophthalmol. 2009;87(7):764–768. | ||
Fukuda S, Kawana K, Yasuno Y, Oshika T. Wound architecture of clear corneal incision with or without stromal hydration observed with 3-dimensional optical coherence tomography. Am J Ophthalmol. 2011;151(3):413–419. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.