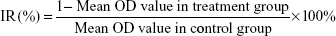Back to Journals » OncoTargets and Therapy » Volume 9
Antitumor effect of manumycin on colorectal cancer cells by increasing the reactive oxygen species production and blocking PI3K-AKT pathway
Authors Zhang J, Jiang H, Xie L, Hu J, Li L, Yang M, Cheng L, Liu B, Qian X
Received 13 December 2015
Accepted for publication 14 March 2016
Published 24 May 2016 Volume 2016:9 Pages 2885—2895
DOI https://doi.org/10.2147/OTT.S102408
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 6
Editor who approved publication: Dr Faris Farassati
Jingyu Zhang,1 Hua Jiang,2 Li Xie,1 Jing Hu,1 Li Li,1 Mi Yang,1 Lei Cheng,1 Baorui Liu,1 Xiaoping Qian1
1Department of the Comprehensive Cancer Center, Affiliated Nanjing Drum Tower Hospital, Nanjing Medical University, 2Department of Oncology, Affiliated Changzhou No 2 People’s Hospital, Nanjing Medical University, Nanjing, People’s Republic of China
Abstract: Manumycin is a natural, well-tolerated microbial metabolite and is regarded as a farnesyltransferase inhibitor. Some data suggest that manumycin inhibits proliferation of diverse cancer cells through various pathways. However, the antitumor effect of manumycin on colorectal cancer (CRC) remains unknown. In the present study, we investigated the antitumor effect of manumycin on CRC in vitro and in vivo. The results of cell viability assay revealed that the proliferation of the CRC cells was significantly inhibited by manumycin. Moreover, cell apoptosis induced by manumycin was also found in a time- and dose-dependent manner. Interestingly, treatment of the CRC cells with manumycin resulted in increased generation of reactive oxygen species. Subsequently, manumycin also decreased the phosphorylation of phosphatidylinositol 3-kinase (PI3K) and AKT, as well as the expression of caspase-9 and poly(ADP-ribose) polymerase (PARP) in a time-dependent manner. In addition, we found that N-acetyl-L-cysteine (NAC) attenuated the effect of manumycin on the PI3K-AKT pathway, and wortmannin reduced the effect of manumycin on caspase-9 and PARP expression. More importantly, the anticancer effect of manumycin was also observed in established tumor xenografts. Taken together, these findings supported the potential application of manumycin against colorectal carcinoma.
Keywords: manumycin, colorectal cancer, PI3K-AKT pathway, ROS
Introduction
Colorectal cancer (CRC) is one of the most common malignant tumors of the gastrointestinal tract. Owing to the changes in adverse dietary habits, the incidence of CRC has increased gradually in the recent years.1 Meanwhile, >25% of patients are diagnosed with metastatic disease.2 The metastatic CRC patients have a poor survival, and fluorouracil (5-FU) is a fundamental drug for chemotherapy of metastatic CRC. After irinotecan and subsequent oxaliplatin were approved, the overall survival of advanced CRC patients was found to be prolonged to ~20 months when treated with 5-FU combined with irinotecan or oxaliplatin.3,4 Targeted therapy, such as cetuximab, a monoclonal antibody that selectively inhibits the downstream activation of the epithelial growth factor receptor, has obtained great achievement in CRC treatment.5 Unfortunately, the use of the above standard therapeutic protocols results only in a moderate decline in mortality because not all CRCs are sensitive to this antiepidermal growth factor receptor agent. Therefore, more effort should be exerted to identify novel drug targets and therapeutics to prolong the survival time of CRC patients.
Phosphatidylinositol 3-kinase (PI3K) was first discovered >20 years ago and is considered as one of the 15 driver mutations during CRC carcinogenesis.6 The effects of PI3K on tumor growth and progression are mediated by AKT, a downstream effector of PI3K. PI3K induces AKT activation via phosphorylation of key amino acid residues, including Thr-308 and Ser-473. Fully phosphorylated AKT activates a multitude of downstream targets, including the mTOR complex1, BAD, caspase-9, and TSC1.7 It has been demonstrated that the activation of caspase-9 can lead to the activation of downstream caspases and apoptosis.8,9 Mutations in PI3K have been observed in 10%–20% of patients with CRC.10,11 Approximately 15% of patients who showed resistance to cetuximab-based chemotherapy harbored a PIK3CA mutation.12 Many clinical trials are currently assessing novel drugs that interfere with signaling from components of the PI3K pathway. However, development of secondary resistance, unanticipated feedback effects, and pathway cross talk have challenged the efforts to design therapeutically effective compounds.13 Thus, drugs inhibiting different critical nodes within the PI3K pathway with less toxicity are urgently needed.
Manumycin, a natural product derived from Streptomyces parvulus that acts as a potent and selective farnesyltransferase inhibitor, was identified as an effective tumoricide against several cancers, including hepatocellular carcinoma, prostate cancer, and anaplastic thyroid carcinoma.14 It was revealed by histological examination that there was no apparent pathological change in the liver, kidney, and heart after manumycin treatment, indicating that it has the low-toxicity property of the agent.15 However, the detailed mechanisms of its actions are incompletely understood. A recent study reported that manumycin-induced apoptosis involved a cascade that began with a large reactive oxygen species (ROS) induction followed by dephosphorylation of MEK and AKT.16 On the other hand, the tumors containing phospho-stable MEK and AKT exhibited either a delayed response to manumycin-induced apoptosis or no response to apoptosis in the extended time. Moreover, manumycin-induced autophagy was reported to be caused by elevated ROS.17 Additionally, in human CRC, manumycin may induce DNA damage via generation of ROS in HCT116 cells in vitro.18 Meanwhile, manumycin suppressed aberrant crypt foci formation in the azoxymethane-induced colorectal carcinogenesis model.19 When it comes to collaborative optimization, interestingly, combination of chemotherapeutic paclitaxel and manumycin could synergistically kill anaplastic thyroid carcinoma cells.20 Therefore, we hypothesized that manumycin could induce apoptosis associated with the ROS production in the human CRC cells.
Materials and methods
Cell culture
The human CRC cell lines SW480 and Caco-2, obtained from the Cell Bank of the Chinese Academy of Sciences (Shanghai, People’s Republic of China), were grown in Roswell Park Memorial Institute-1640 medium supplemented with 10% fetal bovine serum and incubated in humidified 5% CO2 at 37°C. This study was conducted under a protocol approved by the institutional review board at Nanjing Medical University.
Reagents
Manumycin, dimethyl sulfoxide (DMSO; tissue culture grade), and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyletrazolium bromide (MTT) were purchased from Sigma-Aldrich Co. (St Louis, MO, USA). Roswell Park Memorial Institute-1640 medium was purchased from Thermo Fisher Scientific (Waltham, MA, USA). Fetal bovine serum was purchased from HyClone Laboratories, Inc. (Logan, UT, USA). Reactive Oxygen Species Assay Kit, N-acetyl-L-cysteine (NAC), and wortmannin were purchased from Beyotime Institute of Biotechnology (Haimen, Jiangsu, People’s Republic of China). Trifluoroacetate salt hydrate was purchased from Cayman Chemical Company (Ann Arbor, MI, USA). Anti-PI3K antibody, anti-p-PI3K antibody, anti-AKT antibody, anti-p-AKT antibody, anti-caspase-9 antibody, and anti-poly(ADP-ribose) polymerase (PARP) antibody were purchased from Cell Signaling Technology (Danvers, MA, USA). Anti-proliferating cell nuclear antigen (PCNA) antibody was purchased from BD Biosciences (San Jose, CA, USA). β-Actin mouse monoclonal antibody, goat anti-rabbit secondary antibody, and horseradish peroxidase secondary antibody were purchased from Santa Cruz Biotechnology Inc. (Dallas, TX, USA). Annexin-V-FLUOS Staining Kit and In Situ Cell Death Detection Kit, POD, were purchased from Hoffman-La Roche Ltd. (Basel, Switzerland). Manumycin, being lipophilic was dissolved in DSMO. The stock solution was diluted with the final concentration of DSMO in a culture medium not exceeding 0.1% (volume/volume [v/v]).
Cell proliferation assay
The MTT assay was done to investigate the effect of manumycin on the growth of SW480 and Caco-2 cells. Logarithmic phase cells (5×103 cells/well) were seeded in 96-well tissue culture plates in 5% CO2 at 37°C. After culture of 24 hours for cell attachment, the medium was replaced with medium containing different concentrations of manumycin for the indicated time periods. In all, 20 μL MTT dye (5 mg/mL) was added to each cell, and the plates were incubated at 37°C for another 4 hours. Then the medium was removed, and DMSO (150 μL) was added. At last, the plates were shocked at a low volatility for 15 minutes in the darkness, and absorbance of each well at a wavelength of 490 nm was determined using a microplate reader. A decrease in absorbance was considered as a loss of cell viability. Each experiment was repeated for three times. The optical density was recorded as OD, and the inhibition rate (IR%) was calculated using the following equation:
|
|
Apoptosis analysis
The Annexin-V-FLUOS Staining Kit was used to measure the apoptosis cells. According to the manufacturer’s instructions, cells were seeded in six-well plates at 2×105/well and treated with different reagents for the indicated time periods. The cells were harvested, washed with cold phosphate-buffered saline (PBS) for three times, and resuspended in 400 μL of bing buffer, subsequently 8 μL Annexin-V-FITC solution, and propidium iodide was added to these cells. The cells were then incubated in the darkness for 30 minutes at 37°C followed by the analysis of the apoptosis cells by flow cytometry.
Intracellular ROS level
The Reactive Oxygen Species Assay Kit was used to study intracellular ROS. Cells were seeded in six-well plates at 2×105/well and treated with different reagents for the indicated time periods. The medium was removed and replaced with the serum-free medium containing 10 μM dichloro-dihydro-fluorescein diacetate. After 20 minutes of incubation at 37°C in the darkness, the cells were washed with PBS for three times, and then fluorescent intensity was measured by the flow cytometer with excitation and emission wavelengths of 488 nm and 525 nm, respectively.
Western blot
After various treatments, the culture medium and cells floating in the culture medium were removed. The cells that remained attached to the wells were rinsed with PBS for three times and lyzed in radioimmunoprecipitation assay buffer supplemented with a mixture of protease and phosphatase inhibitors, and the total protein was extracted from the cells. The protein concentration was determined by the bicinchoninic acid protein assay. The cell lysate was separated on 10% sodium dodecyl sulfate-polyacrylamide gel (SDS-PAGE). Equal amounts of total protein extracts were separated on a 10% SDS-PAGE and transferred onto a polyvinylidene fluoride membrane. Membranes were blocked with 10% fat-free milk in TBST for 2 hours and then incubated with the following primary antibodies at 4°C overnight: anti-PI3K antibody (diluted at 1:1,000), anti-p-PI3K antibody (diluted at 1:1,000), anti-AKT antibody (diluted at 1:1,000), anti-p-AKT antibody (diluted at 1:1,000), anti-caspase-9 antibody (diluted at 1:1,000), and anti-PARP antibody (diluted at 1:1,000), followed by incubation with HRP secondary antibody (diluted at 1:3,000) for 1 hour. Protein bands were detected using the chemiluminescent HRP substrate. The blot was incubated for 5 minutes and then exposed to a suitable X-ray film for ~30 seconds. Bands were identified and quantified using AlphaEaseFC software.
Inhibition of tumor growth by manumycin in vivo
The activity of manumycin to inhibit tumor growth in vivo was evaluated in the xenograft nude mouse mode. Male athymic BALB/c mice (8 weeks old, 18–20 g) were purchased from Nanjing University Animals Laboratory (Nanjing, Jiangsu, People’s Republic of China) and feed with enough food and water. The animals and their care was conducted in conformity with 3R principles of animal experimentation and approved by Animal Care and Use Ethic Committee of the Affiliated Nanjing Drum Tower Hospital, Nanjing Medical University. Since similar results in SW480 and Caco-2 cells in vitro were obtained, only SW480 cells (1×106) in 0.1 mL of culture medium were injected subcutaneously into the right scapular region of nude mice to evaluate the activity of manumycin in vivo. When the bearing tumors were ~100 mm3 in size, mice were randomly assigned to the three groups. Either control solution (0.1% DMSO in a tissue culture medium) or manumycin (low: 2.5 mg/kg and high: 5 mg/kg) was administered by intraperitoneal injection (n=5 each group) at a 2-day interval for 3 weeks. Tumor size was measured every 3 days using the formula A×B2×0.5236, where A is length and B is width; all were measured in millimeters. All mice were sacrificed on day 22, and tissue specimens were weighed and fixed in formalin for paraffin embedding.
Determination of tumor cell proliferation
Immunohistochemical PCNA staining of tumor tissues were used to evaluate proliferation activity of tumor cells. Briefly, paraffin-embedded tissues were deparaffinized and rehydrated prior to antibody addition. Anti-PCNA antibody was used at a dilution of 1:1,000. Goat anti-rabbit secondary antibody was used at a dilution of 1:200. To quantify PCNA expression, the PCNA labeling index was calculated as number of PCNA positive cells/total number of cells counted under ×200 magnification in three randomly selected areas in each tissue section.
Terminal deoxynucleotidyl transferase dUTP nick-end labeling detection of apoptotic tumor cells
Terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) staining was performed using the In Situ Cell Death Detection Kit, POD, following the manufacturer’s recommended protocol. In brief, the formalin-fixed and paraffin-embedded tissues were cut into 5 μm thick sections. Subsequently, the slides were incubated with the TUNEL reaction mixture containing terminal deoxynucleotidyl transferase enzyme and fluorescein-dUTP. After removing unbound enzyme conjugate, we visualized the POD retained cells in the immune complex using a substrate reaction. We quantified the percentage of apoptotic cells using ImageJ software. Under the light microscope at ×200 magnification, we randomly chose three different visual areas within the same regions of each section. The positive rates of apoptotic cells were calculated as the density of apoptotic cells (apoptosis index).
Statistical analysis
We analyzed the comparisons between the control and treatment groups using one-factor analysis of variance and expressed them as mean ± SD. The differences between two groups were further analyzed using Student–Newman–Keuls test. All data were analyzed with the Statistical Package for the Social Sciences Version 20.0 (IBM Corporation, Armonk, NY, USA). P-value <0.05 was considered significant.
Results
Manumycin inhibits proliferation in human colorectal carcinoma cells
Cytotoxicity of manumycin was assessed by MTT assay using SW480 and Caco-2 cells. As shown in Figure 1, manumycin could inhibit the proliferation of SW480 cells and Caco-2 cells in a dose- and time-dependent manner with IC50 values of 45.05 μM and 43.88 μM after 24 hours of treatment, respectively (Figure 1A and B).
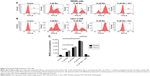
Manumycin elevates ROS production in human colorectal carcinoma cells
To explore whether the anticancer activity of manumycin was associated with the change in intracellular ROS levels, we tested the ROS production after manumycin treatment. As shown in Figure 2, compared with the control group, an increased ROS production was observed with exposure to increasing concentrations of manumycin in SW480 and Caco-2 cells (P<0.05). The pretreatment of NAC (an ROS cleaver) could prevent the ROS accumulation (P<0.05).
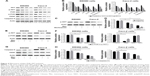
Manumycin induces apoptosis in human CRC cells
We conducted apoptotic analysis to determine whether manumycin induces apoptosis in the CRC cells. As shown in Figure 3, manumycin decreased the percentage of cell apoptosis in a dose- and time-dependent manner in two cell lines (Figure 3A, B, D, and E). Of note, when cells were treated with manumycin and NAC, the decrease in the apoptotic cells was weakened (Figure 3C and F). These results suggested that the apoptotic effect of manumycin was associated with the ROS production.
Manumycin inhibits the PI3K-AKT pathway in human CRC cells
To identify whether the PI3K-AKT pathway was involved in manumycin-induced apoptosis, we tested expression of relevant proteins using immunoblot analysis. In two cell lines, manumycin treatment reduced expression of p-PI3K and p-AKT in a time-dependent manner (Figure 4A and B). There was a similar trend in the expression of PI3K and AKT (Figure 4A and B). Pretreatment of cells with a PI3K inhibitor wortmannin for 1 hour decreased the activation of AKT phosphorylation (Figure 4C and D), while pretreatment of cells with NAC weakened the PI3K and AKT phosphorylation to a smaller extent (Figure 4E and F), indicating ROS production was a upstream of the PI3K-AKT pathway.
Manumycin decreased expression of caspase-9 and PARP in human CRC cells
The caspase family includes important proteins that perform apoptosis. Caspase-9 is the major initiator of the intrinsic apoptotic death pathway, which is activated by mitochondrial perturbation. Caspase-9 then activates caspase-3 and other caspases, subsequently inactivating the DNA repair enzyme PARP. Our results showed that manumycin decreased expression of caspase-9 and PARP in a time-dependent manner (Figure 4A and B). Furthermore, the cells were pretreated with an AKT inhibitor, trifluoroacetate salt hydrate, and the treatment with manumycin led to a further decrease in caspase-9 and PARP (Figure 4G and H). Overall, these data showed that manumycin influenced the level of PARP and capase-9 proteins by modulating the PI3K-AKT pathway.
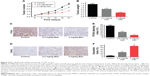
Manumycin significantly inhibits the growth of CRC in vivo
The ability of manumycin as a tumor suppressor was also assessed in the tumor xenograft model. As shown in Figure 5, administration of manumycin by intraperitoneal injection inhibited SW480-derived tumor growth in a dose-dependent manner (Figure 5A). Consistent with tumor volumes, the average tumor weight at 22 days was 0.804±0.059 g and 0.42±0.03 g at the dose of 2.5 mg/kg and 5.0 mg/kg, respectively, being obviously reduced compared with the control group (Figure 5B, P<0.05). Cell proliferation marker PCNA was further examined in situ in tumor samples from the three groups. Manumycin-treated groups had reduced expression of PCNA in comparison with the control group (Figure 5C). Meanwhile, tumor tissue was subjected to TUNEL assay for the determination of apoptotic index. Cell nuclei were stained with nigger brown, which indicated apoptosis and were recorded as TUNEL-positive nuclei. The manumycin-treated group had a significantly higher apoptosis index of tumor cells when compared with the control group (Figure 4D). Moreover, we found no loss in body weights in group treated with manumycin (data not shown). Taken together, manumycin was demonstrated as an efficient and low-toxicity agent against tumors in vivo.
Discussion
Our data provided evidence that manumycin inhibited proliferation and induced apoptosis in the CRC cells. We also demonstrated that manumycin inhibited PI3K-AKT activation and led to ROS accumulation and caspase-9 activation in the CRC cells.
The cellular redox homeostasis is maintained by a tight balance between the rates of ROS production and the ability of the various antioxidant defense systems to modulate redox status. It is well known that the cellular redox status has an important role in varieties of cellular process, including cell proliferation and apoptosis.21,22 Our results showed the anticancer activity of manumycin on colorectal cells was concomitant with the elevated level of intracellular ROS. Moreover, given that antioxidants NAC inhibited accumulation of manumycin-stimulated ROS and attenuated the apoptotic effect on the CRC cells induced by manumycin, the antitumor effect of manumycin might be mediated by ROS generation.
PI3K is an intracellular lipid kinase that plays an important role in cell function and cancer development. Reportedly, PI3K pathway is activated by four principal types of sensors, including the receptor tyrosine kinases, the cytokine- and G-protein-coupled receptors, and the integrins.7,23–25 Upon appropriate binding, these sensors, in conjunction with their cofactors, activate downstream kinases in the PI3K family. PI3K activation leads to the recruitment of AKT and its subsequent phosphorylation by PDK1 and mTORC2 by phosphorylating phosphatidylinositol 4,5-biphosphate to phosphatidylinositol 3,4,5-triphosphate. Phosphorylated AKT exerts signals leading to cell growth and differentiation and angiogenesis and prevents apoptosis by activating a multitude of downstream targets, including caspase-9. We found that manumycin inhibited the phosphorylation of PI3K and AKT. Moreover, the combination of the PI3K inhibitor and manumycin inhibits the phosphorylation of AKT in both CRC cell lines.
Apoptosis is tightly regulated by proapoptotic and antiapoptotic effector molecules and can be mediated by several different pathways in response to various intrinsic and extrinsic signals.26 Caspase-9 is the major initiator of the intrinsic apoptotic death pathway, which results from mitochondrial perturbation.27 Caspase-9 activates caspase-3 and other effector caspases, which proceed to cleave and inactivate the DNA repair enzyme PARP. Hence, PARP cleavage is used as a classic readout for upstream effector caspase activity. Our results indicated that manumycin induced the apoptosis of CRC cell lines and decreased the expression of caspase-9 and PARP, which may be associated with caspase-9 and PARP cleavage. Moreover, the AKT inhibitor significantly decreased the expression of caspase-9 and PARP induced by manumycin. These data confirmed that apoptosis induced by manumycin required capase-9 activation and AKT was an upstream to the caspase-9 activation.
Some studies have demonstrated that wortmannin, a PI3K-AKT inhibitor, considerably inhibits the translocation of nicotinamide adenine dinucleotide phosphate oxidase subunits and reduces ROS production, indicating that PI3K signaling might be involved in this progress.28,29 Actually, the catalytic activity of PTEN is also modulated by ROS, and the cellular PTEN phosphatase activity is inhibited by the oxidative stress.30 Considering the role of PTEN in modulating the PI3K-AKT pathway, there is a feedback loop between ROS and the PI3K-AKT pathway. At the same time, due to massive ROS production in cancer cells, the effective redox adaptation mechanisms would be coped with noxious environment.31 Cancer cells are more vulnerable to oxidative stress caused by exogenous ROS-generating agents. Owing to differential redox states between normal and cancer cells, manumycin might benefit colorectal tumor patients. Our data found that manumycin induced ROS production and subsequently inhibited activation of PI3K and AKT, resulting in cancer cell apoptosis. However, the exact mechanism of ROS production induced by manumycin is not fully understood and is currently under investigation in our laboratory.
Conclusion
In our study, we found that manumycin inhibited proliferation and induced apoptosis on SW480 and Caco-2 cells through modulation of the PI3K-AKT pathway. ROS production played an important role in this progress. Moreover, the anticancer effect of manumycin was further confirmed in an in vivo study of CRC. Our findings provide a preclinical evidence for the potential use of manumycin in the treatment of CRC in the future. However, more work is needed to establish the exact mechanism of manumycin activity in the CRC cells.
Acknowledgments
The authors thank all the people who helped in this study. This work partly was supported by Grant 2011-WS-005 from the Six Talents Peak Foundation of Jiangsu Province, Grant ZKX13022 from the Department of Health key projects of Nanjing City, and Grant 201503013 from Science and Technology Development Program of Nanjing city, Jiangsu Province, People’s Republic of China.
Disclosure
The authors report no conflicts of interest in this work.
References
Kamangar F, Dores GM, Anderson WF. Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol. 2006;24(14):2137–2150. | ||
Ciombor KK, Wu C, Goldberg RM. Recent therapeutic advances in the treatment of colorectal cancer. Annu Rev Med. 2015;66:83–95. | ||
Saltz LB, Cox JV, Blanke C, et al. Irinotecan plus fluorouracil and leucovorin for metastatic colorectal cancer. Irinotecan Study Group. N Engl J Med. 2000;343(13):905–914. | ||
de Gramont A, Figer A, Seymour M, et al. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol. 2000;18(16):2938–2947. | ||
Van Cutsem E, Kohne CH, Hitre E, et al. Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N Engl J Med. 2009;360(14):1408–1417. | ||
Sjoblom T, Jones S, Wood LD, et al. The consensus coding sequences of human breast and colorectal cancers. Science. 2006;314(5797):268–274. | ||
Danielsen SA, Eide PW, Nesbakken A, Guren T, Leithe E, Lothe RA. Portrait of the PI3K/AKT pathway in colorectal cancer. Biochim Biophys Acta. 2015;1855(1):104–121. | ||
Gurumurthy S, Vasudevan KM, Rangnekar VM. Regulation of apoptosis in prostate cancer. Cancer Metastasis Rev. 2001;20(3–4):225–243. | ||
Fesik SW. Promoting apoptosis as a strategy for cancer drug discovery. Nat Rev Cancer. 2005;5(11):876–885. | ||
Frattini M, Signoroni S, Pilotti S, et al. Phosphatase protein homologue to tensin expression and phosphatidylinositol-3 phosphate kinase mutations in colorectal cancer. Cancer Res. 2005;65(23):11227. | ||
Velho S, Oliveira C, Ferreira A, et al. The prevalence of PIK3CA mutations in gastric and colon cancer. Eur J Cancer. 2005;41(11):1649–1654. | ||
De Roock W, Claes B, Bernasconi D, et al. Effects of KRAS, BRAF, NRAS, and PIK3CA mutations on the efficacy of cetuximab plus chemotherapy in chemotherapy-refractory metastatic colorectal cancer: a retrospective consortium analysis. Lancet Oncol. 2010;11(8):753–762. | ||
Yu M, Grady WM. Therapeutic targeting of the phosphatidylinositol 3-kinase signaling pathway: novel targeted therapies and advances in the treatment of colorectal cancer. Therap Adv Gastroenterol. 2012;5(5):319–337. | ||
Li JG, She MR, Lu CY, et al. Manumycin induces apoptosis in prostate cancer cells. Onco Targets Ther. 2014;7:771–777. | ||
Ito T, Kawata S, Tamura S, et al. Suppression of human pancreatic cancer growth in BALB/c nude mice by manumycin, a farnesyl:protein transferase inhibitor. Jpn J Cancer Res. 1996;87(2):113–116. | ||
Sears KT, Daino H, Carey GB. Reactive oxygen species-dependent destruction of MEK and Akt in Manumycin stimulated death of lymphoid tumor and myeloma cell lines. Int J Cancer. 2008;122(7):1496–1505. | ||
Pan J, Song E, Cheng C, Lee MH, Yeung SC. Farnesyltransferase inhibitors-induced autophagy: alternative mechanisms? Autophagy. 2009;5(1):129–131. | ||
Pan J, She M, Xu ZX, Sun L, Yeung SC. Farnesyltransferase inhibitors induce DNA damage via reactive oxygen species in human cancer cells. Cancer Res. 2005;65(9):3671–3681. | ||
Tsuda M, Okamoto K, Muguruma N, et al. Suppressive effect of RAS inhibitor manumycin A on aberrant crypt foci formation in the azoxymethane-induced rat colorectal carcinogenesis model. J Gastroenterol Hepatol. 2013;28(10):1616–1623. | ||
Pan J, Yeung SC. Recent advances in understanding the antineoplastic mechanisms of farnesyltransferase inhibitors. Cancer Res. 2005;65(20):9109–9112. | ||
Scatena R. Mitochondria and cancer: a growing role in apoptosis, cancer cell metabolism and dedifferentiation. Adv Exp Med Biol. 2012;942:287–308. | ||
Nakanishi A, Wada Y, Kitagishi Y, Matsuda S. Link between PI3K/AKT/PTEN pathway and NOX proteinin diseases. Aging Dis. 2014;5(3):203–211. | ||
Chang F, Lee JT, Navolanic PM, et al. Involvement of PI3K/Akt pathway in cell cycle progression, apoptosis, and neoplastic transformation: a target for cancer chemotherapy. Leukemia. 2003;17(3):590–603. | ||
Su CC, Lin YP, Cheng YJ, et al. Phosphatidylinositol 3-kinase/Akt activation by integrin-tumor matrix interaction suppresses Fas-mediated apoptosis in T cells. J Immunol. 2007;179(7):4589–4597. | ||
Papadatos-Pastos D, Rabbie R, Ross P, Sarker D. The role of the PI3K pathway in colorectal cancer. Crit Rev Oncol Hematol. 2015;94(1):18–30. | ||
Chipuk JE, Fisher JC, Dillon CP, Kriwacki RW, Kuwana T, Green DR. Mechanism of apoptosis induction by inhibition of the anti-apoptotic BCL-2 proteins. Proc Natl Acad Sci U S A. 2008;105(51): 20327–20332. | ||
Heck DE, Kagan VE, Shvedova AA, Laskin JD. An epigrammatic (abridged) recounting of the myriad tales of astonishing deeds and dire consequences pertaining to nitric oxide and reactive oxygen species in mitochondria with an ancillary missive concerning the origins of apoptosis. Toxicology. 2005;208(2):259–271. | ||
Ren Z, Raucci FJ Jr, Browe DM, Baumgarten CM. Regulation of swelling-activated Cl(-) current by angiotensin II signalling and NADPH oxidase in rabbit ventricle. Cardiovasc Res. 2008;77(1):73–80. | ||
Ngkelo A, Meja K, Yeadon M, Adcock I, Kirkham PA. LPS induced inflammatory responses in human peripheral blood mononuclear cells is mediated through NOX4 and Gialpha dependent PI-3kinase signalling. J Inflamm (Lond). 2012;9(1):1. | ||
Barbieri SS, Ruggiero L, Tremoli E, Weksler BB. Suppressing PTEN activity by tobacco smoke plus interleukin-1beta modulates dissociation of VE-cadherin/beta-catenin complexes in endothelium. Arterioscler Thromb Vasc Biol. 2008;28(4):732–738. | ||
Li ZY, Yang Y, Ming M, Liu B. Mitochondrial ROS generation for regulation of autophagic pathways in cancer. Biochem Biophys Res Commun. 2011;414(1):5–8. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.