Back to Journals » Infection and Drug Resistance » Volume 12
Antimicrobial activities of ceftazidime–avibactam, ceftolozane–tazobactam, and other agents against Escherichia coli, Klebsiella pneumoniae, and Pseudomonas aeruginosa isolated from intensive care units in Taiwan: results from the Surveillance of Multicenter Antimicrobial Resistance in Taiwan in 2016
Authors Liao CH , Lee NY, Tang HJ, Lee SSJ , Lin CF, Lu PL , Wu JJ, Ko WC , Lee WS, Hsueh PR
Received 6 November 2018
Accepted for publication 22 January 2019
Published 4 March 2019 Volume 2019:12 Pages 545—552
DOI https://doi.org/10.2147/IDR.S193638
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Chun-Hsing Liao,1,2 Na-Yao Lee,3 Hung-Jen Tang,4,5 Susan Shin-Jung Lee,6,7 Chin-Fu Lin,8 Po-Liang Lu,9–11 Jiunn-Jong Wu,12 Wen-Chien Ko,13 Wen-Sen Lee,14 Po-Ren Hsueh15,16
1Department of Internal Medicine, Far Eastern Memorial Hospital, Taipei, Taiwan; 2Department of Medicine, Yang-Ming University, Taipei, Taiwan; 3Department of Internal Medicine, National Cheng Kung University Medical College and Hospital, Tainan, Taiwan; 4Department of Internal Medicine, Chi Mei Medical Center, Tainan, Taiwan; 5Department of Health and Nutrition, Chia Nan University of Pharmacy and Science, Tainan, Taiwan; 6Department of Internal Medicine, Kaohsiung Veterans General Hospital, Kaohsiung, Taiwan; 7Faculty of Medicine, School of Medicine, National Yang Ming University, Taipei, Taiwan; 8Department of Pathology and Laboratory Medicine, Taichung Veterans General Hospital, Taichung, Taiwan; 9Department of Internal Medicine, Kaohsiung Medical University Hospital, College of Medicine, Kaohsiung Medical University, Kaohsiung, Taiwan; 10Department of Laboratory Medicine, Kaohsiung Medical University Hospital, Kaohsiung, Taiwan; 11Graduate Institute of Medicine, College of Medicine, Kaohsiung Medical University, Kaohsiung, Taiwan; 12Department of Biotechnology and Laboratory Science in Medicine, National Yang-Ming University, Taipei, Taiwan; 13Department of Medicine, College of Medicine, National Cheng Kung University, Tainan, Taiwan; 14Division of Infectious Diseases, Department of Internal Medicine, Wan Fang Medical Center, Taipei Medical University, Taipei, Taiwan; 15Department of Laboratory Medicine, National Taiwan University Hospital, National Taiwan University College of Medicine, Taipei, Taiwan; 16Department of Internal Medicine, National Taiwan University Hospital, National Taiwan University College of Medicine, Taipei, Taiwan
Objective: The aim of this study was to investigate the in vitro antimicrobial susceptibilities of clinically important Gram-negative bacteria from seven intensive care units in Taiwan in 2016.
Materials and methods: In total, 300 non-duplicate isolates of Escherichia coli (n=100), Klebsiella pneumoniae (n=100), and Pseudomonas aeruginosa (n=100) collected from 300 patients were studied. The minimum inhibitory concentrations (MICs) of these isolates to antimicrobial agents were determined using the broth microdilution method. Carbapenemase-encoding genes (blaKPC, blaNDM, blaIMP, blaVIM, and blaOXA-48-like) were studied for the isolates that were not susceptible to any carbapenems. Sequencing analysis of the mcr genes (mcr-1–5) was conducted for all isolates with colistin MICs ≥4 mg/L.
Results: Ertapenem non-susceptibility was detected in 3% (n=3) E. coli and 12% (n=12) K. pneumoniae isolates. The susceptibility rates of imipenem, ceftazidime–avibactam (CAZ–AVB), and ceftolozane–tazobactam (CLZ–TAZ) were 99%, 99%, and 88%, respectively, for E. coli, 91%, 100%, and 80%, respectively, for K. pneumoniae, and 66%, 91%, and 93%, respectively, for P. aeruginosa. Carbapenemase-encoding genes were not detected in E. coli, were detected in four (33.3%) K. pneumoniae isolates that were not susceptible to ertapenem (three harboring blaKPC and one harboring blaOXA-48-like), and were not detected in P. aeruginosa isolates that were not susceptible to imipenem. One K. pneumoniae isolate was resistant to colistin (MIC 4 mg/L) and negative for mcr genes.
Conclusion: CAZ–AVB exhibited excellent activity against carbapenem-resistant Enterobacteriaceae, and CLZ–TAZ exhibited good activity against imipenem-resistant P. aeruginosa.
Keywords: carbapenem resistance, second-generation, β-lactam, β-lactamase inhibitor combinations, carbapenemase-encoding genes, mcr
Introduction
Carbapenemase-producing bacteria, especially Enterobacteriaceae (Carbapenemase-producing Enterobacteriaceae, CPE), are emerging worldwide and causing significant morbidity and mortality.1–6 In early 2000, carbapenem resistance was mostly reported in Pseudomonas aeruginosa (mediated by the blaIMP, blaVIM, and blaSIM carbapenemases) and Acinetobacter baumannii (mediated by the blaOXA-23, blaOXA-24, and blaOXA-58 carbapenemases).7,8 Currently, Klebsiella pneumoniae carbapenemase (blaKPC) and New Delhi metallo-β-lactamase (blaNDM) have become matters of primary concern as carbapenem resistance has spread from non-fermenters (nosocomial opportunistic pathogens) to Enterobacteriaceae, which can easily disseminate and cause infections in the community.9,10 The major risk factors for patients infected with carbapenem-resistant Gram-negative bacteria (GNB) are serious underlying illness, long-term care facility residence, and exposure to carbapenem.11–13
Owing to limited treatment options, colistin has become the main antimicrobial agent, either alone or in combination with other drugs.3–5 However, high failure rates have been noted with colistin treatment in previous reviews.1,3–5 New β-lactam combination agents, including ceftazidime–avibactam (CAZ–AVB) and ceftolozane–tazobactam (CLZ–TAZ), exhibit potent in vitro activities against CPE and possess the potential to replace colistin.14,15 A recently published case series of patients with CPE infections who were treated with these two new agents demonstrated the superior efficacies of CAZ–AVB and CLZ–TAZ compared to that of colistin.16–18 However, the susceptibility rates of bacteria to these new agents vary among countries due to different resistance mechanisms,1,5,15,19 and the British guidelines recommend performing molecular typing for carbapenemases for selecting the most suitable agent for CPE treatment.5 Although determination of the molecular mechanisms of carbapenem resistance is difficult, several new methods for accomplishing this have recently become available.20
Antimicrobial resistance among clinically important bacteria collected from intensive care units (ICUs) which were assessed for >10 years in Taiwan2 and increase in carbapenem resistance among Enterobacteriaceae and P. aeruginosa have been noted.21 The purpose of this study was to delineate the in vitro antibacterial activities of CAZ–AVB and CLZ–TAZ against Escherichia coli, K. pneumoniae, and P. aeruginosa isolates collected from ICUs in Taiwan.
Materials and methods
Collection of isolates
Three hundred consecutive, non-duplicate E. coli (n=100), K. pneumoniae (n=100), and P. aeruginosa (n=100) isolates were collected from various clinical specimens of 300 patients in ICUs at seven major teaching hospitals in Taiwan (two in the northern part, one in the middle part, and four in the southern part of Taiwan) from January 1, 2016, to December 31, 2016 (Table 1). The majority of these isolates were recovered from sputum/endotracheal aspirates (n=181, 60.3%), urine (n=55, 18.3%), and blood (n=32, 10.7%) samples of the ICU patients (Table 1). The institutional review board of the National Taiwan University Hospital (201512064RSB) approved this study and waived the requirement for written informed consent. The ethical committees waived the need for informed consent because limited private health information was collected and this research involved minimal risk to the subjects.
Antimicrobial susceptibility testing
In this study, the broth microdilution method with Sensititre™ Gram-negative minimum inhibitory concentration (MIC) plates (Thermo Fisher Scientific, Waltham, MA, USA) was used to determine the MICs of the evaluated antibiotics. E. coli determine the MICs of the evaluated antibiotics 25922 and P. aeruginosa ATCC 27853 were used for quality control on each testing day. The MIC break points recommended by the Clinical and Laboratory Standards Institute (CLSI) in 2018 were used to define the susceptibility of the isolates.22 For E. coli and K. pneumoniae isolates, MICs of ≤8/4 and ≥16/4 mg/L for CAZ–AVB are identified as susceptible and resistant, respectively, whereas MICs of ≤2/4, 4/4, and ≥8/4 mg/L for CLZ–TAZ are classified as susceptible, intermediate, and resistant, respectively, in the CLSI guidelines.22 For P. aeruginosa isolates, MICs of ≤8/4 and ≥16/4 mg/L for CAZ–AVB are identified as susceptible and resistant, respectively, and those of ≤4/4, 8/4, and ≥16/4 mg/L for CLZ–TAZ are classified as susceptible, intermediate, and resistant, respectively, in the CLSI guidelines.22 For E. coli and K. pneumoniae isolates, no CLSI MIC break points for colistin and tigecycline for defining susceptibilities are recommended.22 However, the CLSI defines the susceptibility of E. coli and K. pneumoniae isolates to colistin as wild type (WT; MICs of ≤2 mg/L) and non-WT (MICs of ≥4 mg/L). For P. aeruginosa isolates, MICs of ≤2 and ≥4 mg/L for colistin are identified as susceptible and resistant, respectively.22 For defining the susceptibility of E. coli and K. pneumoniae isolates to tigecycline, MICs of ≤1 and >2 mg/L recommended by the European Committee on Antimicrobial Susceptibility Testing (EUCAST) were adopted for defining susceptibility and resistance, respectively.23
Detection of carbapenemases
For E. coli and K. pneumoniae isolates displaying non-susceptibility to any carbapenem agents (ertapenem, imipenem, meropenem, or doripenem) and for P. aeruginosa isolates exhibiting non-susceptibility to imipenem, meropenem, or doripenem, the Xpert® Carba-R assay (Cepheid, Sunnyvale, CA, USA) was used to detect the carbapenemase-encoding alleles, including blaKPC, blaNDM, blaIMP, blaVIM, and blaOXA-48-like.24 CPE and carbapenemase-producing P. aeruginosa isolates were defined as Enterobacteriaceae or P. aeruginosa, respectively, harboring genes encoding any carbapenemase.
Detection of mcr-1 to mcr-5
PCR amplification of the whole-cell DNA of the isolates with colistin MICs of ≥2 mg/L was performed using previously described primers specific for mcr-1, mcr-2, mcr-3, mcr-4, and mcr-5, and the amplification products were sequenced.24
Statistical analyses
To compare the antimicrobial susceptibility between imipenem-susceptible and non-susceptible P. aeruginosa isolates, Pearson’s chi-squared test or Fisher’s exact test were used. Two-tailed P-values of <0.05 were considered to indicate significant differences. The analysis was performed using SPSS Version 17 (SPSS Inc., Chicago, IL, USA).
Results
Antimicrobial susceptibilities of the isolates
The MIC ranges of CLZ–TAZ and CAZ–AVB for E. coli ATCC 25922 were 0.25–0.5 and 0.12–0.25 mg/L, respectively, whereas those for P. aeruginosa ATCC 27853 were 0.25–0.5 and 1–4 mg/L, respectively. The MIC ranges of the other agents tested against E. coli ATCC 25922 and P. aeruginosa ATCC 27853 were within the MIC ranges recommended by the CLSI.18 Table 2 summarizes the susceptibilities of CLZ–TAZ, CAZ–AVB, and other antimicrobial agents against the 300 isolates of E. coli, K. pneumoniae, and P. aeruginosa. Overall, amikacin exhibited excellent activity (≥96%) against all isolates tested. The observed rates of non-susceptibility to ertapenem were 3% among E. coli and 12% among K. pneumoniae isolates. The rates of susceptibility to imipenem were 99% for E. coli, 91% for K. pneumoniae, and 66% for P. aeruginosa. All the E. coli isolates were inhibited by 0.5 mg/L tigecycline. In contrast, 89% of the K. pneumoniae isolates were inhibited by 1 mg/L tigecycline (susceptible based on the EUCAST criteria), whereas eight and three isolates exhibited MICs of 2 mg/L (intermediate by the EUCAST criteria) and 4 mg/L (resistant by the EUCAST criteria), respectively. All P. aeruginosa isolates were susceptible to colistin (MICs of ≤2 mg/L) and all E. coli isolates were inhibited by 0.5 mg/L colistin (all WT isolates). Five of the P. aeruginosa isolates exhibited colistin MICs of 2 mg/L. Among the K. pneumoniae isolates, 99% were inhibited by 2 mg/L colistin, whereas one exhibited colistin MIC of 4 mg/L (ie, non-WT).
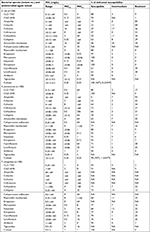  | Table 2 In vitro susceptibilities of E. coli, K. pneumoniae, and P. aeruginosa isolates collected from patients admitted to the ICUs of seven major teaching hospitals across Taiwan in 2016 to 19 antimicrobial agents Note: The MICs were interpreted based on the criteria of the 2018 CLSI.18 Abbreviations: CAZ–AVB, ceftazidime–avibactam; CLSI, Clinical and Laboratory Standards Institute; CLZ–TAZ, ceftolozane–tazobactam; E. coli, Escherichia coli; ICUs, intensive care units; K. pneumoniae, Klebsiella pneumoniae; MICs, minimum inhibitory concentrations; NA, non-applicable; NWT, non-WT; P. aeruginosa, Pseudomonas aeruginosa; WT, wild type. |
The rates of susceptibility to CAZ–AVB were 99% for E. coli, 100% for K. pneumoniae, and 91% for P. aeruginosa. The MIC of the E. coli isolate resistant to CAZ–AVB was 16/4 mg/L. The rates of susceptibility to CLZ–TAZ were 88% for E. coli, 80% for K. pneumoniae, and 93% for P. aeruginosa.
Comparison between imipenem-susceptible and imipenem-non-susceptible P. aeruginosa isolates
For imipenem-non-susceptible P. aeruginosa isolates, the most potent agent was colistin (susceptibility rate 100%), followed by amikacin (97.1%), CLZ–TAZ (85.3%), CAZ–AVB (79.4%), and ciprofloxacin (64.8%; Figure 1). All the differences in susceptibility rates to the seven selected agents between the imipenem-susceptible and imipenem-non-susceptible P. aeruginosa isolates were significant (P<0.05).
Detection of carbapenemase-encoding genes
Among the isolates tested, 1 (1%) isolate of E. coli and 12 (12%) isolates of K. pneumoniae were not susceptible to one of the four carbapenems tested. Carbapenemase-encoding genes were detected in none of the E. coli isolates and in four (33.3%) K. pneumoniae isolates (three harboring blaKPC and one harboring blaOXA-48-like). These four isolates were susceptible to CAZ–AVB (MICs of 0.12–4 mg/L) and colistin (MICs of 0.5–1 mg/L), while their MICs for tigecycline ranged from 0.5 to 2 mg/L. One blaKPC-K. pneumoniae isolate was resistant to amikacin, and all three blaKPC-K. pneumoniae isolates were resistant to CLZ–TAZ. The blaOXA-48-like-K. pneumoniae isolate was susceptible to ceftriaxone, cefepime, CLZ–TAZ, and fluoroquinolones, intermediate to ertapenem (MIC of 1 mg/L), and susceptible to the other carbapenems tested, but resistant to piperacillin–tazobactam. No carbapenemase-encoding genes were detected among the 34 isolates of P. aeruginosa that were not susceptible to imipenem.
Detection of mcr-1 to mcr-5
mcr-1 to mcr-5 were not detected in the K. pneumoniae isolate with colistin MIC of 4 mg/L.
Discussion
New β-lactam combination agents, including CAZ–AVB and CLZ–TAZ, have been approved in 2015 and 2014, respectively, for use in cases of urinary tract infections and intra-abdominal infections based on the results of randomized controlled trials.14 However, the increasing prevalence of carbapenem-resistant pathogens is a major hurdle for effective treatment using these two agents. In a recently published evaluation of CLZ–TAZ for the treatment of serious infections (51% pneumonia) caused by carbapenem-resistant P. aeruginosa,16 the successful treatment rate was 74% on average (70% for monotherapy and 87% for combination therapy with another active agent). Treatment failure was associated with isolate MICs ≥8 mg/L. Similarly, a multicenter evaluation of CAZ–AVB against CPE showed significant reduction in all-cause hospital mortality compared to colistin.18 In that study, 37% patients were treated with CAZ–AVB monotherapy, whereas only 6% patients were treated with colistin monotherapy. A pooled analysis of CAZ–AVB Phase III clinical trials indicated that its efficacy was comparable to that of carbapenems used for critical infections, including nosocomial pneumonia caused by multidrug-resistant pathogens.25 An increase in the use of the new β-lactamase inhibitors can be anticipated, although careful susceptibility testing and surveillance studies are warranted as the rates and mechanisms of resistance vary among regions and resistance may develop during treatment.1,5,14,19
In this surveillance study of isolates collected from patients admitted to seven ICUs in Taiwan in 2016, the non-susceptibility rate of E. coli isolates to ertapenem (3%) was lower than that to ceftriaxone (53%) and cefepime (37%). For K. pneumoniae, 12% isolates were not susceptible to ertapenem, and carbapenemase-encoding genes were detected in four isolates (three isolates harboring blaKPC and one harboring blaOXA-48-like). CAZ–AVB exhibited excellent in vitro activities against E. coli (99%) and K. pneumoniae (100%) isolates, whereas CTL-TAZ exhibited lower activities (88% and 80%, respectively). These observations imply that CAZ–AVB can be confidently recommended as an empirical and definite treatment for infections caused by Enterobacteriaceae in the ICU setting in Taiwan.
For P. aeruginosa, the imipenem susceptibility rate was only 66%. In this study, carbapenem non-susceptibility was associated with decreased susceptibility to other antimicrobial agents tested with the exception of amikacin and colistin. The overall susceptibility rate of CLZ–TAZ was 93% for all P. aeruginosa isolates, but decreased to 85% for imipenem non-susceptible isolates, whereas the corresponding rates for CAZ–AVB were 91% and 79%, respectively. Nevertheless, the rates of bacterial susceptibility to these new two β-lactamase inhibitors were higher than those to other current drugs, including cefepime and piperacillin–tazobactam. It is surprising that the Xpert® Carba-R assay detected no carbapenemase-encoding genes among the carbapenem non-susceptible P. aeruginosa. According to previous studies on carbapenem-resistant P. aeruginosa in Taiwan, the overproduction of active efflux pump and OprD polymorphisms were the major mechanisms of resistance,26,27 which might explain the negative results.
In the present study, colistin and amikacin were active in vitro against the three tested major pathogenic bacteria. Compared to other antimicrobial agents in Taiwan, the use of aminoglycosides is gradually decreasing owing to the possibility of developing nephrotoxicity as a side effect. In a previous 9-year study between 2003 and 2011, the mean consumption of amikacin (defined daily dose per 1,000 patient-days) was 8, compared to 56 for extended-spectrum cephalosporins, 17 for piperacillin–tazobactam, 22.4 for carbapenems (with an increase each year), and 52 for quinolone.28 However, the emergence of resistant microorganisms warrants further clinical study on the role of aminoglycosides, which have been proposed to be useful as part of combination therapies16,18 or high-dose monotherapy.29,30 Meanwhile, although tigecycline has maintained a high susceptibility rate against Enterobacteriaceae for >10 years since its first use,31 it should be applied cautiously and be limited to the treatment of intra-abdominal and soft tissue infections according to the British guidelines.5
Our study had several limitations. First, CAZ–AVB and CLZ–TAZ combinations were tested against only a limited number of drug-resistant, β-lactamase-harboring isolates. Second, we did not perform the in vitro susceptibilities of some oral carbapenems (tebipenem and faropenem), ceftolozane, and other new β-lactam combination agents, including diazabicyclooctane-based inhibitors and boronic acid-based inhibitor such as vaborbactam (meropenem–vaborbactam). The current Sensititre format does not include these agents, and we cannot obtain or purchase the standard powders of these agents for in-house broth microdilution study. Furthermore, these agents have not yet been launched in Taiwan now, and most of them will not be available in the near future in this country.
Conclusion
CAZ–AVB and CLZ–TAZ, which have not yet been launched in Taiwan, have high susceptibility rates against E. coli, K. pneumoniae, and P. aeruginosa isolates collected from ICUs in Taiwan. With the recent publication of a case series showing their clinical superiority to colistin, these two agents may be used instead of the current colistin-based treatment for carbapenem-resistant pathogens in the near future.
Acknowledgments
We thank all investigators of the participating hospitals for their cooperation and support in the Surveillance of Multicenter Antimicrobial Resistance in Taiwan (SMART) program. This surveillance study was sponsored by internal funding.
Disclosure
The authors report no conflicts of interest in this work.
References
Tzouvelekis LS, Markogiannakis A, Psichogiou M, Tassios PT, Daikos GL. Carbapenemases in Klebsiella pneumoniae and other Enterobacteriaceae: an evolving crisis of global dimensions. Clin Microbiol Rev. 2012;25(4):682–707. | ||
Jean SS, Lee WS, Yu KW, et al. Rates of susceptibility of carbapenems, ceftobiprole, and colistin against clinically important bacteria collected from intensive care units in 2007: results from the surveillance of multicenter antimicrobial resistance in Taiwan (SMART). J Microbiol Immunol Infect. 2016;49(6):969–976. | ||
Li J, Nation RL, Turnidge JD, et al. Colistin: the re-emerging antibiotic for multidrug-resistant Gram-negative bacterial infections. Lancet Infect Dis. 2006;6(9):589–601. | ||
Paul M, Daikos GL, Durante-Mangoni E, et al. Colistin alone versus colistin plus meropenem for treatment of severe infections caused by carbapenem-resistant Gram-negative bacteria: an open-label, randomised controlled trial. Lancet Infect Dis. 2018;18(4):391–400. | ||
Hawkey PM, Warren RE, Livermore DM, et al. Treatment of infections caused by multidrug-resistant Gram-negative bacteria: report of the British Society for antimicrobial Chemotherapy/Healthcare infection Society/British infection association joint working Party. J Antimicrob Chemother. 2018;73(Suppl 3):iii2–iii78. | ||
Lee CH, Su TY, Ye JJ, et al. Risk factors and clinical significance of bacteremia caused by Pseudomonas aeruginosa resistant only to carbapenems. J Microbiol Immunol Infect. 2017;50(5):677–683. | ||
Livermore DM, Woodford N. Carbapenemases: a problem in waiting? Curr Opin Microbiol. 2000;3(5):489–495. | ||
Poirel L, Nordmann P. Carbapenem resistance in Acinetobacter baumannii: mechanisms and epidemiology. Clin Microbiol Infect. 2006;12(9):826–836. | ||
Nordmann P, Cuzon G, Naas T. The real threat of Klebsiella pneumoniae carbapenemase-producing bacteria. Lancet Infect Dis. 2009;9(4):228–236. | ||
Kumarasamy KK, Toleman MA, Walsh TR, et al. Emergence of a new antibiotic resistance mechanism in India, Pakistan, and the UK: a molecular, biological, and epidemiological study. Lancet Infect Dis. 2010;10(9):597–602. | ||
Ting SW, Lee CH, Liu JW. Risk factors and outcomes for the acquisition of carbapenem-resistant Gram-negative Bacillus bacteremia: a retrospective propensity-matched case control study. J Microbiol Immunol Infect. 2018;51(5):621–628. | ||
Tsao LH, Hsin CY, Liu HY, Chuang HC, Chen LY, Lee YJ. Risk factors for healthcare-associated infection caused by carbapenem-resistant Pseudomonas aeruginosa. J Microbiol Immunol Infect. 2018;51(3):359–366. | ||
Lee CM, Lai CC, Chiang HT, et al. Presence of multidrug-resistant organisms in the residents and environments of long-term care facilities in Taiwan. J Microbiol Immunol Infect. 2017;50(2):133–144. | ||
van Duin D, Bonomo RA. Ceftazidime/avibactam and ceftolozane/tazobactam: second-generation β-lactam/β-lactamase inhibitor combinations. Clin Infect Dis. 2016;63(2):234–241. | ||
Sader HS, Castanheira M, Shortridge D, Mendes RE, Flamm RK. Antimicrobial activity of Ceftazidime-Avibactam tested against multidrug-resistant Enterobacteriaceae and Pseudomonas aeruginosa isolates from U.S. medical centers, 2013 to 2016. Antimicrob Agents Chemother. 2017;61(11):e01045–17. | ||
Munita JM, Aitken SL, Miller WR, et al. Multicenter evaluation of ceftolozane/tazobactam for serious infections caused by carbapenem-resistant Pseudomonas aeruginosa. Clin Infect Dis. 2017;65(1):158–161. | ||
Shields RK, Nguyen MH, Chen L, et al. Ceftazidime-avibactam is superior to other treatment regimens against carbapenem-resistant Klebsiella pneumoniae bacteremia. Antimicrob Agents Chemother. 2017;61(8):e00883–17. | ||
van Duin D, Lok JJ, Earley M, et al. Colistin versus ceftazidime-avibactam in the treatment of infections due to carbapenem-resistant Enterobacteriaceae. Clin Infect Dis. 2018;66(2):163–171. | ||
Katchanov J, Asar L, Klupp EM, et al. Carbapenem-resistant Gram-negative pathogens in a German University medical Center: prevalence, clinical implications and the role of novel β-lactam/β-lactamase inhibitor combinations. PLoS One. 2018;13(4):e0195757. | ||
Lutgring JD, Limbago BM. The problem of carbapenemase-producing-carbapenem-resistant-Enterobacteriaceae detection. J Clin Microbiol. 2016;54(3):529–534. | ||
Jean SS, Hsueh PR, Lee WS, et al. Carbapenem susceptibilities and non-susceptibility concordance to different carbapenems amongst clinically important Gram-negative bacteria isolated from intensive care units in Taiwan: results from the surveillance of multicentre antimicrobial resistance in Taiwan (SMART) in 2009. Int J Antimicrob Agents. 2013;41(5):457–462. | ||
Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing: Twenty-Eighth Informational Supplement M100-S28. Wayne, PA: CLSI; 2018. | ||
European Committee on Antimicrobial Susceptibility Testing [webpage on the Internet]. Breakpoint Tables for Interpretation of MICs and Zone Diameters (version 8.0). Available from: http://www.eucast.org/clinical_breakpoints/. Accessed September 9, 2018. | ||
Jean SS, Lu MC, Shi ZY, et al. In vitro activity of ceftazidime-avibactam, Ceftolozane-Tazobactam, and other comparable agents against clinically important Gram-negative bacilli: results from The 2017 surveillance of multicenter antimicrobial resistance in Taiwan (SMART). Infect Drug Resist. 2018;11:1983–1992. | ||
Stone GG, Newell P, Gasink LB, et al. Clinical activity of ceftazidime/avibactam against MDR Enterobacteriaceae and Pseudomonas aeruginosa: pooled data from the ceftazidime/avibactam Phase III clinical trial programme. J Antimicrob Chemother. 2018;73(9):2519–2523. | ||
Kao CY, Chen SS, Hung KH, et al. Overproduction of active efflux pump and variations of OprD dominate in imipenem-resistant Pseudomonas aeruginosa isolated from patients with bloodstream infections in Taiwan. BMC Microbiol. 2016;16(1):107. | ||
Shu JC, Kuo AJ, Su LH, et al. Development of carbapenem resistance in Pseudomonas aeruginosa is associated with OprD polymorphisms, particularly the amino acid substitution at codon 170. J Antimicrob Chemother. 2017;72(9):2489–2495. | ||
Lee HS, Loh YX, Lee JJ, Liu CS, Chu C. Antimicrobial consumption and resistance in five Gram-negative bacterial species in a hospital from 2003 to 2011. J Microbiol Immunol Infect. 2015;48(6):647–654. | ||
Gálvez R, Luengo C, Cornejo R, et al. Higher than recommended amikacin loading doses achieve pharmacokinetic targets without associated toxicity. Int J Antimicrob Agents. 2011;38(2):146–151. | ||
Sadeghi K, Hamishehkar H, Najmeddin F, et al. High-dose amikacin for achieving serum target levels in critically ill elderly patients. Infect Drug Resist. 2018;11:223–228. | ||
Chen YH, Lu PL, Huang CH, et al. Trends in the susceptibility of clinically important resistant bacteria to tigecycline: results from the tigecycline in vitro surveillance in Taiwan study, 2006 to 2010. Antimicrob Agents Chemother. 2012;56(3):1452–1457. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


