Back to Journals » Journal of Inflammation Research » Volume 15
An Integrated Analysis of Inflammatory Endotypes and Clinical Characteristics in Chronic Rhinosinusitis with Nasal Polyps
Authors Hao D , Wu Y, Li P , Li C, Jiang T , Zhang Q , Liu S , Shi L
Received 3 June 2022
Accepted for publication 15 September 2022
Published 24 September 2022 Volume 2022:15 Pages 5557—5565
DOI https://doi.org/10.2147/JIR.S377301
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Ning Quan
Dingqian Hao, Yisha Wu, Ping Li, Chunhao Li, Tianjiao Jiang, Qian Zhang, Shengyang Liu, Li Shi
Department of Otolaryngology-Head and Neck Surgery, Shandong Provincial ENT Hospital, Shandong University, Jinan, Shandong, People’s Republic of China
Correspondence: Li Shi; Shengyang Liu, Department of Otolaryngology-Head and Neck Surgery, Shandong Provincial ENT Hospital, Shandong University, Jinan, Shandong, 250000, People’s Republic of China, Email [email protected]; [email protected]
Objective: Chronic rhinosinusitis with nasal polyps (CRSwNP) is mainly characterised by type 1 (T1), type 2 (T2) and type 3 (T3) inflammatory endotypes. However, correlations between inflammatory endotypes and clinical features in CRSwNP have not been demonstrated sufficiently. This study aimed to determine the endotype-phenotype associations in CRSwNP.
Methods: Clinical data of 31 control subjects and 106 CRSwNP patients were analysed. Interferon (IFN)-γ (T1), Charcot-Leyden crystal galectin (CLC) (T2) and Interleukin (IL)-17A (T3) were used as biomarkers to identify the inflammatory endotypes.
Results: The mRNA expression level of IFN-γ was positively correlated with IL-17A (r = 0.817; P < 0.0001). Headache/facial pain (P = 0.039) was associated with T1 endotype. Smell loss (P = 0.025) was associated with T2 endotype. Purulent rhinorrhea (P = 0.001) was associated with T3 endotype. Atopy (P = 0.030), asthma (P = 0.005) and recurrence (P = 0.022) were more frequent in T2 endotype. Total Symptom Scores (TSS) of T2 (P < 0.001) and T3 (P = 0.009) endotype were higher than non-T2 and non-T3, respectively. Sino Nasal Outcome Test-22 (SNOT-22) total scores of T3 (P = 0.054) endotype were higher than non-T3.
Conclusion: Identifications of endotype-phenotype associations are useful in clinical diagnoses and targeted therapies for patients with CRSwNP.
Keywords: chronic rhinosinusitis with nasal polyps, endotype, phenotype, diagnosis, therapy
Introduction
Chronic rhinosinusitis (CRS) is a chronic upper airway inflammatory disease with significant heterogeneity. Traditionally, CRS is classified as the two main phenotypes: CRS without nasal polyps (CRSsNP) and CRS with nasal polyps (CRSwNP).1 In all CRS, CRSsNP accounts for approximately 80%, whereas CRSwNP only for 20%. Histopathologically, CRSwNP is further divided into eosinophilic CRSwNP (eos-CRSwNP) and neutrophilic CRSwNP (neu-CRSwNP). In western populations, most patients with CRSwNP have a type 2 (T2) inflammation characterised by eosinophil infiltration and overexpression of interleukin (IL)-4, IL-5 and IL-132. In Asian countries, especially China, the proportions of type 1 (T1) and type 3 (T3) inflammation in CRSwNP are higher.3–5
In a recent paper, interferon (IFN)-γ, Charcot-Leyden crystal galectin (CLC) and IL-17A are identified as biomarkers of T1, T2 and T3 endotypes, respectively.6 As CRSwNP is a complicated and heterogeneous inflammatory response, subgroups of patients with mixed inflammatory endotypes are determined. Like the pathogenesis and pathophysiology of CRSwNP, asthma is a chronic lower airway inflammatory disease characterised by T2 inflammation. Clinically, asthma is severe comorbidity in many CRSwNP patients. In patients with asthma, the correlations between endotypes and phenotypes have been well addressed.7,8 However, the associations between inflammatory endotypes and clinical features are still unclear in CRSwNP.
Currently, there is evidence that the T2 endotype is associated with nasal polyps formation and asthma.9 Pronounced eosinophilia of nasal polyps is observed frequently in T2 inflammation.10 The correlations between the T2 endotype and other clinical phenotypes are still uncertain. Furthermore, the associations between T1, T3 or mixed endotypes and clinical features are not established sufficiently. Therefore, this study aimed to show whether inflammatory endotypes are associated with specific clinical characteristics. Improved understandings of endotype-phenotype associations provide personalised medical treatments for patients with CRSwNP.
Materials and Methods
Subjects and Samples Collection
A total of 106 CRSwNP patients and 31 healthy control subjects were recruited from June 2021 to May 2022 in Shandong Provincial ENT Hospital. Patients undergoing septoplasty for nasal obstruction, repairs of nasal fractures or transnasal endoscopy skull base procedures and without other sinonasal diseases were recruited as control subjects. The otolaryngologists reviewed the medical records for each study subject to obtain clinical data, and reviewers of medical records were blinded to the endotyping results. The diagnosis of CRSwNP was based on the standard criteria according to European Position Paper on Rhinosinusitis and Nasal Polyps 2020 (EPOS 2020).11 The inclusion criteria were as follows: 1. at least one symptom (nasal congestion, purulent rhinorrhea, smell loss, headache/facial pain) lasting at least 12 weeks; 2. none of the patients had been treated with corticosteroids, antibiotics, or biologics within the four weeks before surgery. Patients with immunodeficiency, pregnancy, coagulation disorder, fungal sinusitis, cystic fibrosis and aspirin-exacerbated respiratory disease (AERD) were excluded from the study. Nasal polyps (NP) tissue was obtained during routine functional endoscopic sinus surgery (FESS) performed on patients with CRSwNP. Ethmoid tissue (ET) was taken from healthy control patients. Atopy was measured by the Immuno CAP 250 system (Pharmacia, Uppsala, Sweden) and confirmed based on a positive test for serum antigen-specific IgE (cut-off value: 0.35 kUA/L). All participants signed the informed consent forms. The Ethics Review Committee of Shandong Provincial ENT Hospital approved the protocols and procedures for this study. Detailed clinical information of all subjects is shown in Table 1.
 |
Table 1 Baseline Characteristics of Control Subjects and CRSwNP Patients |
RNA Extraction and Quantitative Real-Time PCR (qPCR)
Total RNA was isolated from nasal biopsy tissues using the mirVana miRNA Isolation Kit (Life Technologies, Grand Island, NY, USA). Total RNA concentration was detected with a spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). 1 μg of total RNA was reverse transcribed into complementary DNA in a 20 µL reaction volume using the iScript cDNA Synthesis Kit (Bio-Rad, Hercules, CA, USA) according to the manufacturer’s protocol. Then, the target genes were amplified using the GoTaq-qPCR Master Mix Kit (Promega, San Luis Obispo, CA, USA). The 2−ΔΔCT method was used to analyse relative mRNA expression levels of specific genes. The housekeeping genes were glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and ribosomal protein L13a (RPL13A). The details of gene primer sequences used are described in Supplementary Table S1.
CRSwNP Endotyping
IFN-γ, CLC and IL-17A were used to indicate T1, T2 and T3 inflammation, respectively. The mRNA expression levels of endotypic biomarkers were detected by quantitative real-time PCR (qPCR) in NP and ET.12 A cutoff of more significant than the 90th percentile of the expression levels in control ET was used to define the inflammatory endotypes. The endotypic patterns were divided into specific endotypes (eg, patients with T1 inflammation), single endotypes (eg, patients with only T2 inflammation), mixed endotypes (eg, patients with both T1 and T3 inflammation) and untypeable (Tun) endotypes (eg, patients without any elevation of T1, T2 or T3 inflammation).
Visual Analogue Scale (VAS) and Sino Nasal Outcome Test-22 (SNOT-22)
VAS and SNOT-22 were used to assess the severity of clinical symptoms in CRSwNP patients. Seven significant symptoms, including nasal congestion, purulent rhinorrhea, smell loss, headache/facial pain, sneezing, nasal itching and eye itching, were evaluated by the patients using a VAS of 0 to 10. Zero signified the absence of any symptom, and ten indicated the presence of the most severe sign. Total Symptom Scores (TSS) was the sum of all symptom scores. SNOT-22 was classified as the five major domains, including rhinologic (need to blow nose, nasal blockage, sneezing, runny nose and decreased sense of smell/taste), extranasal rhinologic (cough, postnasal discharge and thick nasal discharge), ear/facial (ear fullness, dizziness, ear pain and facial pain/pressure), psychological (fatigue, reduced productivity, reduced concentration, frustrated/restless/irritable, sad and embarrassed) and sleep dysfunction (difficulty falling asleep, nighttime awakening, lack of a good night’s sleep and waking up tired). Every item in each domain was scored from 0 to 5.
Lund-Mackay Score (LMS) and Nasal Polyp Score (NPS)
The LMS recorded computed tomography (CT) findings. Each sinus (maxillary, anterior ethmoid, posterior ethmoid, sphenoid and frontal) was scored as 0 (completely clear), 1 (partly opacified) and 2 (completely opacified). The osteomeatal complex (OMC) region score was 0 or 2 (not occluded/occluded). LMS from 0 to 24 included the sum of all sinuses and OMC region scores. NP size was determined by the nasal endoscopy according to the Lund-Kennedy system. NP size was scored as 0 = no NP; 1 = NP in the middle meatus; 2 = NP reaching below the middle turbinate; 3 = NP reaching below the inferior turbinate; 4 = NP blocking the unilateral nasal cavity. NPS from 0 to 8 was the sum of bilateral NP scores.
Statistical Analysis
All statistical calculations were performed using Statistical Product and Service Solutions (SPSS) 26.0 (International Business Machines Corporation, New York, USA). The Chi-square test and Mann–Whitney U-test were used to compare the differences between the two groups. The nonparametric Spearman test was used to analyse the correlations of clinical data. Multivariate logistic regression analysis was used for the associations among different variables. The final data were presented as mean ± standard error (SEM) or median ± interquartile range (IQR). A P value less than or equal to 0.05 was considered statistically significant.
Results
Patterns of Inflammatory Endotypes in CRSwNP
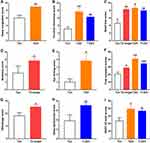
In all CRSwNP, T1, T2, and T3 endotypes accounted for 59%, 75% and 60%, respectively (Figure 1B-D). Additionally, 2%, 24%, and 2% of CRSwNP patients had only T1, T2 and T3 inflammation (single endotypes), respectively (Figure 1A). Among the mixed endotypes, the frequency of T1/2, T1/3, T2/3 and T1/2/3 endotypes was 5%, 12%, 6% and 40%, respectively (Figure 1A). Furthermore, 9% of CRSwNP patients without any T1, T2 and T3 inflammation had an untypeable endotype (Figure 1A). In the comparison between the control and non-T1 CRSwNP, it was found that the IFN-γ mRNA expression level was significantly elevated in T1 CRSwNP (Figure 1E). On the other hand, CLC mRNA expression level was significantly increased in T2 CRSwNP in the comparison between the control and non-T2 CRSwNP (Figure 1F). In the comparison between the control and non-T3 CRSwNP, IL-17A mRNA expression level was significantly upregulated in T3 CRSwNP (Figure 1G). IFN-γ mRNA expression level was positively correlated with IL-17A (Figure 1H). IFN-γ mRNA expression level was not associated with CLC (Figure 1I). CLC mRNA expression level was not correlated with IL-17A (Figure 1J).
Clinical Characteristics in CRSwNP
Compared to in non-T1 endotype, headache/facial pain (P = 0.009; Figure 2D) and ear/facial scores (P = 0.009; Figure 2J) were higher in T1 endotype. Compared to in non-T2 endotype, the scores of nasal congestion (P = 0.033; Figure 2A), purulent rhinorrhea score (P = 0.028; Figure 2B), smell loss (P = 0.004; Figure 2C), sneezing (P = 0.003; Figure 2E), nasal itching (P = 0.024; Figure 2F), eye itching (P = 0.054; Figure 2G), TSS (P < 0.001; Figure 2H), rhinologic (P = 0.038; Figure 2I) and sleep dysfunction (P = 0.057; Figure 2K) were higher in T2 endotype. Compared to in non-T3 endotype, purulent rhinorrhea score (P = 0.001; Figure 2B), eye itching (P = 0.035; Figure 2G), TSS (P = 0.009; Figure 2H), ear/facial score (P = 0.013; Figure 2J) and SNOT-22 total scores (P = 0.054; Figure 2L) were higher in T3 endotype. In T2 endotype, atopy (P = 0.030; Figure 3A), asthma (P = 0.005; Figure 3B) and recurrence (P = 0.022; Figure 3C) were more common. The details of clinical characteristics for all CRSwNP patients are presented in Supplementary Table S2.
 |
Figure 3 Prevalence of comorbid atopy (A), asthma (B) and recurrence (C) in specific inflammatory endotypes. *P < 0.05 and **P < 0.01. |
Associations Between Inflammatory Endotypes and Clinical Characteristics in CRSwNP
Multivariate logistic regression analysis was performed to identify the associations between 7 major clinical symptoms (nasal congestion, purulent rhinorrhea, smell loss, headache/facial pain, sneezing, nasal itching and eye itching) and inflammatory endotypes. Headache/facial pain (OR 1.18, 95% CI [1.01–1.38], P = 0.039; Figure 4A) was positively associated with T1 endotype. Smell loss (OR 1.22, 95% CI [1.03–1.45], P = 0.025; Figure 4B) was positively associated with T2 endotype. Purulent rhinorrhea (OR 1.29, 95% CI [1.10–1.50], P = 0.001; Figure 4C) was positively associated with T3 endotype. Other clinical features were not significantly associated with any endotype when controlling for age, sex, atopy, asthma and recurrence (Supplementary Table S3).
 |
Figure 4 Odds ratio (OR) and 95% confidence interval (CI) of headache/facial pain (A), smell loss (B), purulent rhinorrhea (C) in specific inflammatory endotypes. *P < 0.05 and **P < 0.01. |
Single, Mixed and Untypeable Endotypes in CRSwNP
Single T1 and T3 endotypes were rare (Figure 1A) and were not included in the analysis. Clinical presentations were compared between untypeable endotypes and single endotypes (T2) or mixed endotypes (T1/2, T1/3, T2/3 and T1/2/3) (Supplementary Table S4). Smell loss score (P = 0.001; Figure 5C), sneezing score (P = 0.036; Figure 5D), TSS (P = 0.011; Figure 5F) and rhinologic score (P = 0.026; Figure 5G) were higher in T2 single endotype. The scores for nasal congestion (P = 0.002; Figure 5A), purulent rhinorrhea (P = 0.007; Figure 5B), smell loss (P = 0.021; Figure 5C), eye itching (P = 0.027; Figure 5E), TSS (P = 0.002; Figure 5F) and SNOT-22 total scores (P = 0.042; Figure 5I) were higher in T2/3 endotype. The scores of purulent rhinorrhea (P = 0.001; Figure 5B), smell loss (P = 0.002; Figure 5C), TSS (P < 0.001; Figure 5F), sleep dysfunction (P = 0.049; Figure 5H) and SNOT-22 total scores (P = 0.032; Figure 5I) were higher in T1/2/3 endotype.
Discussion
Over the past few years, T1, T2, and T3 endotypes have been widely used to reveal the inflammatory patterns in CRSwNP.13–15 Compared to in western populations, T1 and T3 inflammation proportions are higher in Asian patients with CRSwNP. Our findings confirmed this point. In the present study, the proportions of T1 and T3 endotypes in CRSwNP were 59% and 60%, respectively (Figure 1). In T1 inflammation, IFN-γ is a critical immune cytokine and exerts antiviral effects. In CRS, Stevens et al found that headache/facial pain was negatively associated with T1 endotype.16 However, our data showed that headache/facial pain was positively associated with the T1 endotype in CRSwNP (Figure 4). This suggests that the presence of NP may affect the associations between inflammatory endotypes and clinical phenotypes. Currently, there are few papers focusing on the associations between T1 endotype and clinical presentations. Our results demonstrated that the T1 endotype was also associated with other clinical features, such as the symptoms of ear/facial domain (ear fullness, dizziness, ear pain and facial pain/pressure) (Figure 2).
In our CRSwNP patients, T2 inflammation, accounting for 75%, is still the most critical endotypic pattern (Figure 1). In patients with asthma, two major inflammatory endotypes, including T2-high asthma and T2-low asthma, have been well described. Furthermore, endotype-phenotype associations in asthma have also been widely studied. Similarly, several clinical characteristics are associated with the T2 endotype in CRSwNP. Wang et al showed that CRSwNP patients with asthma had higher levels of T2 inflammation compared to patients with CRSwNP-alone.17 A study found that smell loss was more common in T2 CRSwNP and that an eosinophil marker CLC was correlated with olfactory defects.18 Likewise, in our results, the prevalence of comorbid asthma was higher in T2 CRSwNP (Figure 3). Moreover, smell loss was positively associated with the T2 endotype in CRSwNP (Figure 4). These results suggest that CRSwNP patients in this study are typical of the general CRSwNP populations. It will be valuable to study how T2 inflammation induces olfactory dysfunction in the future. Additionally, our findings have shown that the allergic symptoms, including sneezing, nasal itching and eye itching, were also associated with the T2 endotype (Figure 2). Hence, we infer that T2 inflammation may be involved in the allergic immune response.
T3-mediated immunity is primarily involved in the host defence against bacteria and fungi.19,20 In T3 inflammation, IL-17A is a crucial effector cytokine mainly secreted by T helper 17 (Th17) cell and innate lymphoid cell 3 (ILC3).21,22 Our data indicated that IL-17A mRNA expression level was positively correlated with IFN-γ in CRSwNP (Figure 1). This suggests that there may be a synergistic effect between T1 and T3 inflammation in the pathogenesis of CRSwNP. It was reported that non-T2 CRSwNP in Asia might display a mixed T1 and T3 endotype with neutrophilia.6 Furthermore, Wang et al reported that the T1/3 endotype is the most common in non-T2 CRSwNP in Beijing.2 Future studies will be required to identify the underlying immunological mechanisms in T1 and T3 CRSwNP.9 In a recent paper, purulent nasal drainage was shown to be positively associated with T3 CRS (16). Similarly, purulent rhinorrhea was positively associated with the T3 endotype in CRSwNP (Figure 4). Pus formation is one of the main symptoms associated with a bacterial infection. These results suggest that T3 inflammation is strongly associated with a bacterial infection. Recently, several studies have demonstrated a central role for IL-17A in CRSwNP generally characterised by neutrophil infiltration.23–25 Therefore, we infer that patients can be responsive to antibiotic treatment in T3 CRSwNP characterised by glucocorticoid insensitivity.
In some cases, mixed endotypes share similar clinical characteristics as single endotypes. For example, nasal congestion, purulent rhinorrhea and smell loss were all associated with the T2/3 endotype (Figure 5). However, there were no significant changes in headache/facial pain and nasal itching scores between untypeable endotypes and single or mixed endotypes (Supplementary Table S3). This suggests that mixed endotypes are not necessarily a simple combination of individual endotypes. In our data, 9% of CRSwNP patients without any T1, T2 and T3 inflammation were untypeable using the current endotyping biomarkers (Figure 1). There may be one or more unrecognised endotypes in untypeable CRSwNP patients.26 Further studies are required to investigate the potential biomarkers that could identify new endotypes in untypeable CRSwNP patients.27 In recent years, monoclonal biologics have been used as an innovative therapy for various chronic inflammatory diseases. Some targeted biologics, including Omalizumab (anti-IgE), Dupilumab (anti-IL-4) and Mepolizumab (anti-IL-5), are currently used in clinical therapies for CRSwNP.28–30 Identifying treatment biomarkers based on endotype-phenotype associations may help optimise treatment strategies and improve the therapeutic efficacy of targeted biologics.
In conclusion, our study shows that inflammatory endotypes are directly associated with several clinical characteristics in CRSwNP. Recognising patterns of inflammatory endotypes is useful in the clinical diagnosis of CRSwNP. Identifying the endotype-phenotype associations may provide more precise targeted therapies for CRSwNP patients.
Data Sharing Statement
All data generated or analysed during this study are included in this published article and its Supplementary Materials.
Ethics and Consent
We briefed all participants in detail on the sample collection process, and all participants provided written informed consent following the declaration of Helsinki. All procedures in this study related to human participants followed the ethical standards of the institutional and/or national research committees. The approval of this study was obtained from the institutional review committee of the participating hospitals (China approval number: XYK-20210703).
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; they took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the report has been submitted; and agree to be accountable for all aspects of the work.
Funding
This work was supported by a grant from the National Natural Science Foundation of China (No. 8187040823).
Disclosure
The authors report no conflicts of interest in relation to this work and declare that the research was conducted without any commercial or financial relationships construed as a potential conflict of interest.
References
1. Bauer AM, Turner JH. Personalized Medicine in Chronic Rhinosinusitis: phenotypes, Endotypes, and Biomarkers. Immunol Allergy Clin North Am. 2020;40(2):281–293. doi:10.1016/j.iac.2019.12.007
2. Wang X, Zhang N, Bo M, et al. Diversity of T(H) cytokine profiles in patients with chronic rhinosinusitis: a multicenter study in Europe, Asia, and Oceania. J Allergy Clin Immunol. 2016;138(5):1344–1353. doi:10.1016/j.jaci.2016.05.041
3. Shi LL, Xiong P, Zhang L, et al. Features of airway remodeling in different types of Chinese chronic rhinosinusitis are associated with inflammation patterns. Allergy. 2013;68(1):101–109. doi:10.1111/all.12064
4. Nakayama T, Sugimoto N, Okada N, et al. JESREC score and mucosal eosinophilia can predict endotypes of chronic rhinosinusitis with nasal polyps. Auris Nasus Larynx. 2019;46(3):374–383. doi:10.1016/j.anl.2018.09.004
5. Cao PP, Li HB, Wang BF, et al. Distinct immunopathologic characteristics of various types of chronic rhinosinusitis in adult Chinese. J Allergy Clin Immunol. 2009;124(3):478–484, 484.e471–472. doi:10.1016/j.jaci.2009.05.017
6. Kato A, Peters AT, Stevens WW, Schleimer RP, Tan BK, Kern RC. Endotypes of chronic rhinosinusitis: relationships to disease phenotypes, pathogenesis, clinical findings, and treatment approaches. Allergy. 2022;77(3):812–826. doi:10.1111/all.15074
7. Education NA, Program P. Expert Panel Report 3 (EPR-3): guidelines for the Diagnosis and Management of Asthma-Summary Report 2007. J Allergy Clin Immunol. 2007;120(5 Suppl):S94–138. doi:10.1016/j.jaci.2007.09.029
8. Salinas YD, Wang Z, DeWan AT. Discovery and Mediation Analysis of Cross-Phenotype Associations Between Asthma and Body Mass Index in 12q13.2. Am J Epidemiol. 2021;190(1):85–94. doi:10.1093/aje/kwaa144
9. Xu X, Reitsma S, Wang Y, Fokkens WJ. Highlights in the advances of chronic rhinosinusitis. Allergy. 2021;76(11):3349–3358. doi:10.1111/all.14892
10. Lee K, Tai J, Lee SH, Kim TH. Advances in the Knowledge of the Underlying Airway Remodeling Mechanisms in Chronic Rhinosinusitis Based on the Endotypes: a Review. Int J Mol Sci. 2021;22(2):214.
11. Fokkens WJ, Lund VJ, Hopkins C, et al. European Position Paper on Rhinosinusitis and Nasal Polyps 2020. Rhinology. 2020;58(Suppl S29):1–464. doi:10.4193/Rhin20.401
12. Huang ZQ, Liu J, Ong HH, et al. Interleukin-13 Alters Tight Junction Proteins Expression Thereby Compromising Barrier Function and Dampens Rhinovirus Induced Immune Responses in Nasal Epithelium. Front Cell Dev Biol. 2020;8:572749. doi:10.3389/fcell.2020.572749
13. Tomassen P, Vandeplas G, Van Zele T, et al. Inflammatory endotypes of chronic rhinosinusitis based on cluster analysis of biomarkers. J Allergy Clin Immunol. 2016;137(5):1449–1456.e1444. doi:10.1016/j.jaci.2015.12.1324
14. Lam M, Hull L, McLachlan R, et al. Clinical severity and epithelial endotypes in chronic rhinosinusitis. Int Forum Allergy Rhinol. 2013;3(2):121–128. doi:10.1002/alr.21082
15. Tan BK, Klingler AI, Poposki JA, et al. Heterogeneous inflammatory patterns in chronic rhinosinusitis without nasal polyps in Chicago, Illinois. J Allergy Clin Immunol. 2017;139(2):699–703.e697. doi:10.1016/j.jaci.2016.06.063
16. Stevens WW, Peters AT, Tan BK, et al. Associations Between Inflammatory Endotypes and Clinical Presentations in Chronic Rhinosinusitis. J Allergy Clin Immunol Pract. 2019;7(8):2812–2820.e2813. doi:10.1016/j.jaip.2019.05.009
17. Wang M, Bu X, Luan G, et al. Distinct type 2-high inflammation associated molecular signatures of chronic rhinosinusitis with nasal polyps with comorbid asthma. Clin Transl Allergy. 2020;10(1):26. doi:10.1186/s13601-020-00332-z
18. Delemarre T, Holtappels G, De Ruyck N, et al. Type 2 inflammation in chronic rhinosinusitis without nasal polyps: another relevant endotype. J Allergy Clin Immunol. 2020;146(2):337–343.e336. doi:10.1016/j.jaci.2020.04.040
19. Hoggard M, Waldvogel-Thurlow S, Zoing M, et al. Inflammatory Endotypes and Microbial Associations in Chronic Rhinosinusitis. Front Immunol. 2018;9:2065. doi:10.3389/fimmu.2018.02065
20. Klingler AI, Stevens WW, Tan BK, et al. Mechanisms and biomarkers of inflammatory endotypes in chronic rhinosinusitis without nasal polyps. J Allergy Clin Immunol. 2021;147(4):1306–1317. doi:10.1016/j.jaci.2020.11.037
21. Scheckenbach K, Wagenmann M. Cytokine Patterns and Endotypes in Acute and Chronic Rhinosinusitis. Curr Allergy Asthma Rep. 2016;16(1):3. doi:10.1007/s11882-015-0583-4
22. Ryu G, Dhong HJ, Park M, et al. Age-associated changes in chronic rhinosinusitis endotypes. Clin Exp Allergy. 2020;50(5):585–596. doi:10.1111/cea.13586
23. Brown HJ, Tajudeen BA, Kuhar HN, Gattuso P, Batra PS, Mahdavinia M. Defining the Allergic Endotype of Chronic Rhinosinusitis by Structured Histopathology and Clinical Variables. J Allergy Clin Immunol Pract. 2021;9(10):3797–3804. doi:10.1016/j.jaip.2021.06.013
24. Lou H, Wang C, Zhang L. Endotype-driven precision medicine in chronic rhinosinusitis. Expert Rev Clin Immunol. 2019;15(11):1171–1183. doi:10.1080/1744666X.2020.1679626
25. Benjamin MR, Stevens WW, Li N, et al. Clinical Characteristics of Patients with Chronic Rhinosinusitis without Nasal Polyps in an Academic Setting. J Allergy Clin Immunol Pract. 2019;7(3):1010–1016. doi:10.1016/j.jaip.2018.10.014
26. Liao B, Liu JX, Li ZY, et al. Multidimensional endotypes of chronic rhinosinusitis and their association with treatment outcomes. Allergy. 2018;73(7):1459–1469. doi:10.1111/all.13411
27. Soler ZM, Schlosser RJ, Bodner TE, et al. Endotyping chronic rhinosinusitis based on olfactory cleft mucus biomarkers. J Allergy Clin Immunol. 2021;147(5):1732–1741.e1731. doi:10.1016/j.jaci.2021.01.021
28. Wang C, Zhang L. Use of biologics in chronic sinusitis with nasal polyps. Curr Opin Allergy Clin Immunol. 2019;19(4):365–372. doi:10.1097/ACI.0000000000000540
29. Mortuaire G, Gengler I, Balden M, Capron M, Lefèvre G. Impact of allergy on phenotypic and endotypic profiles of nasal polyposis. Eur Ann Otorhinolaryngol Head Neck Dis. 2018;135(3):159–162. doi:10.1016/j.anorl.2017.11.005
30. Lal D, Hopkins C, Divekar RD. SNOT-22-based clusters in chronic rhinosinusitis without nasal polyposis exhibit distinct endotypic and prognostic differences. Int Forum Allergy Rhinol. 2018;8(7):797–805. doi:10.1002/alr.22101
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.