Back to Journals » International Medical Case Reports Journal » Volume 12
Acute hypervitaminosis A misdiagnosed as malaria in a 7-year-old Nigerian boy
Authors Babatola AO , Olatunya OS , Ogundare EO , Ajite AB , Oluwayemi IO , Thomas AA, Taiwo AB , Fatunla OA, Komolafe AK , Alfred A
Received 30 April 2019
Accepted for publication 15 June 2019
Published 9 July 2019 Volume 2019:12 Pages 213—216
DOI https://doi.org/10.2147/IMCRJ.S212848
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Ronald Prineas
Adefunke Olarinre Babatola,1,2 Oladele Simeon Olatunya,1,2 Ezra Olatunde Ogundare,1,2 Adebukola Bidemi Ajite,1,2 Isaac Oludare Oluwayemi,1,2 Awolowo Anthony Thomas,3 Adekunle Bamidele Taiwo,2 Odunayo Adebukola Fatunla,2 Akinwumi Kolawole Komolafe,2 Airemionkhale Alfred2
1Department of Paediatrics, Ekiti State University, Ado-Ekiti, Ekiti State, Nigeria; 2Department of Paediatrics, Ekiti State University Teaching Hospital, Ado-Ekiti, Ekiti State, Nigeria; 3Department of Radiology, Ekiti State University, Ado-Ekiti, Ekiti State, Nigeria
Abstract: Vitamin A supplementation program where single high-dose vitamin A supplements are provided to the qualified (infants and children) at regular intervals is operational in Nigeria as a public health initiative to control vitamin A deficiency which is prevalent in our country in accordance with the WHO recommendations. Reports of symptomatic acute hypervitaminosis A are scarce. We report a case of acute hypervitaminosis A resulting from accidental ingestion of vitamin A supplement capsules. This is to reiterate the need for caregivers to keep drugs out of the reach of children. Clinicians should also have a high index of suspicion.
Keywords: vitamin A supplementation, malaria, accidental ingestion, raised intracranial pressure
Introduction
Vitamin A belongs to the group of fat-soluble vitamins that can be readily obtained through the intake of green leafy vegetables, palm oil, and other sources. It is an essential micronutrient that is important for various functions and mediates some bioactive functions in human. These include maintenance of immune system and healthy growth and development in children. Its deficiency impairs innate and adaptive immunity and subsequent increased risk of severe infection and visual impairments.1 According to the WHO, vitamin A deficiency occurs in about one-third of children aged 6–59 months, and the highest rate occurs in Sub-Saharan Africa.2 Vitamin A deficiency is a public health problem in Nigeria, and its prevalence ranges between 11% and 28%.3,4 Due to the high burden of vitamin A deficiency and its clinical consequences, Nigeria has also adopted the World Health Organization recommendation of routine vitamin A supplementation for infants and children between 6 and 59 months in areas where vitamin A deficiency is a public health problem.5
Case report
A 7-year-old boy was referred to our facility from a comprehensive health center where he presented with vomiting, generalized body weakness, headache, and blurred vision. He was managed for malaria having tested positive for malaria parasite by Rapid Diagnostic Test kit. Symptoms, however, worsened despite commencement of antimalarial medications. Following further clerking on presentation at our facility, it was found that child had ingested 10 capsules of vitamin A, each of 200,000 IU (red colored) about 4 hrs prior to the onset of symptoms. There was eye pain and neck pain. There was no history of trauma to the head, no fever, and no personality changes. His mother, who works with a health care facility, had stored the vitamin A capsules in a cupboard, and the child had thought the capsules were the usual multivitamins. On examination, he was weak, not febrile, and not pale. He had convergent squint of the right eye and bilateral papilloedema. The pulse rate and blood pressure were 72 beats/min and 90/50 mmHg, respectively. The child was conscious, had neck stiffness, had pupils that were equal bilaterally, and reacted normally to light both direct and consensual. The child was unable to abduct the right eye laterally. There were no lateralizing signs. A provisional diagnosis of hypervitaminosis A with intracranial hypertension was made. The complete blood count (CBC) was normal (packed cell volume [PCV] 38%, white blood cell count [WBC] 5.8×109/L, polymorphs 67%, lymphocytes 23%, others 10%, platelet count 248×109/L). The patient was commenced on oral prednisolone (100 mg) once daily and acetazolamide (250 mg) once daily. In addition, magnetic resonance imaging (MRI) was requested. Twenty-four hours into admission, the child complained of double vision, leg pain, abdominal pain, and reduced appetite in addition to the headache. The patient still had some body weakness though vomiting had subsided. Examination revealed that the pulse rate had reduced to 60 beats/min, blood pressure increased to 100/50 mmHg, while respiratory rate remained normal at 22 cycles/min. He had his medications reviewed with prednisolone reduced to 50 mg daily. The MRI with contrast revealed mild tortuosity and thickening of the optic nerve sheath with prominent subarachnoid space and flattening out of the pituitary gland. There were also mucosa thickenings of the walls of both sphenoid sinuses. The brain architecture and position, the globe, and retrobulbar structures were all reported to be normal (Figure 1).
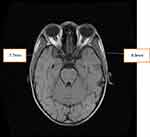 |
Figure 1 Right and left optic nerve sheath diameters. Normal diameters =3.8–7.4 mm. Thickened optic nerve sheath diameter suggests raised intracranial pressure (ICP). |
The drugs earlier prescribed were continued. Five days into admission, the child improved remarkably evidenced by normal vital signs, improved activity and reduced intensity, as well as severity of headache. However, he still had squint and blurred vision which improved on tilting the head to one side. The medications were reviewed with prednisolone reduced to 50 mg daily and acetazolamide maintained at 250 mg daily for another 5 days. Subsequently, the child was reviewed and found to have improved significantly apart from mild blurring of vision and squint. The patient was discharged home on his medications and given an appointment for clinic follow-up in 5 days. Repeat CBC was normal except for mild leukocytosis (PCV 33%, WBC 12.7×109/L, platelets 245×109/L). Electrolytes, urea, and creatinine were essentially normal (chloride 111 mmol/L, sodium-145 mmol/L, potassium 3.02 mmol/L, urea 3.8 mmol/L, creatinine 43.0 mmol/L). He was then followed up weekly and drugs tapered off over a 6-week period by which time the patient had recovered completely.
Discussion
Vitamin A is essential for the functioning of the immune system, healthy growth, and development of children. Its deficiency impairs innate and adaptive immunity and subsequently increased risk of severe infection and visual impairments.1 The WHO has classified vitamin A deficiency as a public health problem affecting about one-third of children aged 6–59 months, with the highest rates of about 48% in sub-Saharan Africa, Nigeria inclusive.2 The prevalence of vitamin A deficiency in Nigeria ranges between 11% and 28%.3,4 Due to this high burden of vitamin A deficiency and its clinical consequences, the WHO recommends vitamin A supplementation for infants and children between 6 and 59 months in areas where vitamin A deficiency is a public health problem.5 This intervention has been shown to reduce the risk of all-cause mortality by 24%1 and reduced the prevalence of vitamin A deficiency over time as reported in some areas in Nigeria.6,7
The global prevalence of vitamin A toxicity is not known, and there is no local data on it in Nigeria. However, the 34th annual report of the American Association of Poison Control Centers in 2016 reported 60,000 cases of vitamin A poisoning.8
Vitamin A toxicity can occur in both acute overdose settings and chronic use settings. The acute toxic dose of vitamin A is 25,000 IU/kg and the chronic toxic dose is 4,000 IU/kg every day for 6–15 months.9 The pathophysiology of vitamin A toxicity is not entirely known, but it is thought to be related to oversaturation of its binding protein, and being a fat-soluble vitamin, the excess unbound vitamin accumulates in the body until toxic level is reached. The duration of exposure, weight, and dose exposed to seem to interplay.10,11
The symptoms of acute poisoning include nausea, vomiting, diarrhea, hemorrhage, skin peeling, erythema, blurred vision, headache, abdominal pain, squint, sores around the mouth and eyes, and raised intracranial pressure among others. The chronic manifestations include changes in the long bones, liver damage, and hypercalcemia, and all the acute symptoms may also be present.10,12–15 In infancy, transient bulging of anterior fontanelle has been reported following administration of doses between 100,000 IU and 200,000 IU.13 The treatment of vitamin A toxicity entails early detection, withdrawal of all forms of vitamin A, and supportive management.
Our 7-year-old patient presented with vomiting, generalized body weakness, headache and blurred vision, eye pain, and neck pain which could be features of acute toxicity of vitamin A; however, many of these features are nonspecific and could be present in some other clinical conditions like malaria and viral infections. Our patient was thought to have had malaria and was initially treated for it at the referring center and referred to our facility on account of worsening of the symptoms. The presence of headache, blurring of vision, and squint in our patient suggest raised intracranial pressure which prompted the request for a contrasted MRIwhich showed no mass but only features of mild intracranial pressure. This observation alongside the history of ingestion of 10 capsules (each of vitamin A of 200,000 IU strength) further strengthens the diagnosis of pseudotumor cerebri secondary to acute vitamin A toxicity in the child for which he was placed on a carbonic anhydrase inhibitor (acetazolamide) and corticosteroids. The patient responded well and the symptoms completely resolved by 6 weeks. The health workers at the referral center were informed of the final diagnosis and commended for referring the patient. The vitamin A formulations in use come in 100,000 IU (colored blue) and 200,000 IU (colored red) soft gelatin capsules. This color coding was believed to have resulted in improved training and operational efficiencies on the field.5 However, given their attractiveness, the color coding and the high dose in each capsule could pose an increased risk of accidental ingestion and possible toxicity in children as found in this case. Our patient might have been attracted to the drug on account of the red color of the capsules. We do not have the facilities to estimate retinyl ester concentration in the serum of our patient. The limitation of the study was that we were unable to do serum calcium and alkaline phosphatase.10 Despite these limitations, this case report illustrates the need and importance of detailed clerking and physical examination by clinicians in resource-poor settings in arriving at clinical diagnosis. In addition, it also illustrates the need for caregivers to keep drugs and other poisonous substances out of the reach of children. Furthermore, drug manufacturers need not make drugs in fascinating or bright colors to attract children as this could pose threat to their lives as found in this case. This case also highlights the importance of collaborations among different levels of health care services toward achieving optimal care for patients.
Ethical considerations
This case does not require institutional ethical approval. However, written informed consent of the caregiver (parent) was obtained to have the case details and accompanying images published.
Disclosure
The authors report no conflicts of interest in this work.
References
1. World Health Organization. Vitamin A supplementation. Available from: https://www.who.int/immunization/programmes-systems/interventions/vitamin A/en/.
2. United Nations Children Fund. Vitamin A deficiency. Available from: https://data.unicef.org/topic/nutrition/vitamin-a-deficiency/.
3. Ajaiyeoba AI. Vitamin A deficiency in Nigerian children. Afri J Biomed Res. 2001;4:107–110.
4. Adelekan DA, Fatusi AO, Fakunle JB, et al. Prevalence of malnutrition and vitamin A deficiency in Nigerian preschool children subsisting on high intakes of carotenes. Nutr Health. 1997;12:17–24. doi:10.1177/026010609701200102
5. World Health Organization. Vitamin A supplementation in infants and children 6-59 months. E-Library of Evidence for Nutrition Actions(eLENA). Available from: https://www.who.int/elena/titles/full-recommendations/vitamina/supp/en/.
6. Abolurin OO, Adegbola AJ, Oyelami OA, Adegoke SA, Bolaji OO. Prevalence of Vitamin A deficiency among under-five children in South-Western Nigeria. Niger Postgrad Med J. 2018;25:13–16. doi:10.4103/npmj.npmj_20_18
7. Adamu A, Jiya NM, Ahmed H, et al. Prevalence of vitamin A deficiency among malnourished children in Usmanu Danfodiyo University Teaching Hospital, Sokoto, Northwestern Nigeria. Pak J Nutr. 2015;15:821–828.
8. Gummin DD, Mowry JB, Spyker DA, Brooks DE, Fraser MO, Banner W. Annual report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 34th annual report. Clin Toxicol (Phila). 2017;55(10):1072–1252. doi:10.1080/15563650.2017.1388087
9. Rosenbloom M. Vitamin toxicity [Updated December 26, 2017]. Available from https://emedicine.medscape.com/article/819426-overview.
10. Blomhoff R. Vitamin A and carotenoid toxicity. Food Nutr Bull. 2001;22:320–331. doi:10.1177/156482650102200309
11. Tan KP, Kosuge K, Yang M, Ito S. NRF2 as a determinant of cellular resistance in retinoic acid cytotoxicity. Free Radic Biol Med. 2008;45(12):1663–1673. doi:10.1016/j.freeradbiomed.2008.09.010
12. Maija Z. Vitamin A deficiencies and excess. In: kliegman, editor. Nelson Text Book of Paedaitrics.
13. WHO. Adverse events following administration vitamin A supplements. Available from:http://www.who.int/immunization/programmes_systems/interventions/Adverse events_vitA.pdf. Accessed October 12th, 2018.
14. Baineni R, Gulati R. Delhi CK vitamin A toxicity presenting as bone pain. Arch Dis Child. 2017;102:556–558. doi:10.1136/archdischild-2016-310631
15. Cheruvattath R, Orrego M, Gautam M, et al. Vitamin A toxicity: when one a day doesn’t keep the doctor away. Liver Transpl. 2006;12:1888–1891. doi:10.1002/lt.21007
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
