Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
Absence of Adverse Effects of Tiotropium/Olodaterol Compared with the Monocomponents on Long-Term Heart Rate and Blood Pressure in Patients with Moderate-to-Very-Severe COPD
Authors Andreas S , McGarvey L, Bothner U, Trampisch M, de la Hoz A, Fležar M, Buhl R, Alter P
Received 17 January 2020
Accepted for publication 12 June 2020
Published 10 August 2020 Volume 2020:15 Pages 1935—1944
DOI https://doi.org/10.2147/COPD.S246348
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Dr Richard Russell
Stefan Andreas,1,2 Lorcan McGarvey,3 Ulrich Bothner,4 Matthias Trampisch,4 Alberto de la Hoz,4 Matjaz Fležar,5 Roland Buhl,6 Peter Alter7
1Department of Cardiology and Pneumology, University Medical Center Göttingen, Göttingen, Germany; 2LungClinic Immenhausen, Immenhausen, Germany, Member of the German Center for Lung Research (DZL); 3Queen’s University Belfast, Belfast, UK; 4Boehringer Ingelheim International GmbH, Ingelheim am Rhein, Germany; 5Klinika Golnik, Golnik, Slovenia; 6Pulmonary Department, Johannes Gutenberg University Mainz, Mainz, Germany; 7Department of Medicine, Pulmonary and Critical Care Medicine, Philipps University of Marburg (UMR); Member of the German Center for Lung Research (DZL), Marburg, Germany
Correspondence: Stefan Andreas
Department of Cardiology and Pneumology, University Medical Center Göttingen, Robert-Koch-Str. 40, Göttingen, Germany
Tel +49 05673 501 1112
Fax +49 05673-501-1101
Email [email protected]
Introduction: Long-acting β2-agonists (LABAs) and long-acting muscarinic antagonists (LAMAs) are established maintenance bronchodilator treatments for chronic obstructive pulmonary disease (COPD) with the potential to increase heart rate (HR) and impact blood pressure (BP). While previous studies indicate that HR and BP are not negatively influenced by tiotropium or olodaterol monotherapy, the effect of tiotropium/olodaterol has not been evaluated. We report a post hoc analysis of the effect of dual bronchodilation with tiotropium/olodaterol versus monocomponents on HR and BP in patients with moderate-to-very-severe COPD included in the large TONADO® study.
Methods: The TONADO® trials (1237.5 [NCT01431274] and 1237.6 [NCT01431287]) were two replicate, randomized, double-blind, parallel-group, 52-week, Phase III trials that compared tiotropium/olodaterol (5/5 μg and 2.5/5 μg) with tiotropium (5 μg and 2.5 μg) and olodaterol (5 μg) in patients with moderate-to-very-severe COPD. Patients with cardiovascular comorbidities were included. Changes in HR and systolic/diastolic BP were measured before and after dosing with the study medication at each visit (baseline, Week 12, Week 24 and Week 52).
Results: Overall, 3,100 patients were included in this analysis. Over 52 weeks, small changes from baseline in mean HR (< 2 beats per minute [bpm]) and small changes from pre- to post-dose (< 1 bpm) were evident at different time points. There was a non-significant increase from baseline in mean diastolic and systolic BP (< 2 mmHg) observed over 52 weeks of treatment. The short-term (1 hour pre- to 1 hour post-dose) mean changes in systolic and diastolic BP over 52 weeks in the tiotropium/olodaterol 5/5 μg group were comparable with those observed for the monocomponents at all time points.
Conclusion: There were no differences in HR or BP among patients on tiotropium/olodaterol when compared with monocomponents. This supports the already demonstrated cardiovascular safety profile of tiotropium/olodaterol as long-acting maintenance bronchodilator treatment for COPD, including patients with cardiovascular comorbidities.
Keywords: chronic obstructive pulmonary disease, tiotropium, olodaterol, heart rate, blood pressure
Plain Language Summary
In people with chronic obstructive pulmonary disease (COPD), increased heart rate and changes in blood pressure can occur. It is important to know whether drugs used to treat COPD affect the risk of these conditions. Therefore, we looked at heart rate and blood pressure data from a large clinical trial called TONADO® that included 3,100 patients with COPD.
Patients were given one of two medications routinely used to manage COPD—tiotropium or olodaterol—or both together (tiotropium/olodaterol). We measured blood pressure and heart rate at the start of the trial and after 12, 24 and 52 weeks of treatment, both shortly before and after patients received their treatment.
Reassuringly, we saw only small changes in heart rate and blood pressure during the trial. Overall, we observed no relevant differences in heart rate or blood pressure in patients who received tiotropium/olodaterol compared with tiotropium or olodaterol alone. Our findings support other trials showing that tiotropium/olodaterol is a well-tolerated and effective treatment for patients with COPD, including those with existing heart or circulatory conditions.
Introduction
In patients with chronic obstructive pulmonary disease (COPD), the Global Initiative for Chronic Obstructive Lung Disease (GOLD) strategy document recommends long-acting bronchodilator maintenance therapy.1
Tiotropium is an established once-daily long-acting muscarinic antagonist (LAMA) that improves lung function, patient-oriented outcomes, such as dyspnea and quality of life, and reduces exacerbations in patients with COPD.2 Olodaterol is an approved once-daily long-acting β2-agonist (LABA) that is highly selective, provides 24-hour bronchodilation and has been shown to improve lung function and symptomatic benefit in patients with COPD.3 The additional benefits of combining tiotropium with olodaterol as a fixed-dose combination have been demonstrated in clinical studies, including the two 52-week, Phase III TONADO® trials.4
Patients with COPD often have comorbidities that can impact treatment and disease management. The most serious and prevalent comorbidities are cardiovascular diseases,5–9 which are known to be leading causes of hospitalization and death.6,10 Cardiovascular morbidity and mortality are related to elevations in resting heart rate (HR) and blood pressure (BP).11 Increased HR may also be a marker of amplified neurohumoral activation.12 Neurohumoral activation can contribute to the progression of heart failure,13 and this may be a potential mechanism which links HR to increases in mortality in COPD.12–16 A post hoc analysis of 16,485 patients in the SUMMIT trial characterized the relationship between BP and HR and mortality and morbidity in COPD, and concluded that increases in HR are associated with elevated mortality and cardiovascular events.11 In addition, they also demonstrated a “U-shaped” relationship between BP and cardiovascular events in patients with COPD (ie, increased mortality and cardiovascular events were linked to both high and low BP in individuals with COPD,11 as has also been seen with patients without COPD17,18).
LABAs and LAMAs have been linked with cardiovascular adverse events in both observational studies and clinical trials.19 In addition, LABAs have been suspected of causing increased HR and impacting BP via β2-adrenoceptors in the atria, ventricles and peripheral vasculature.19,20 However, LAMA/LABA and LABA/inhaled corticosteroid (ICS) combination treatments both significantly improved cardiac function and lung hyperinflation compared with placebo in patients with COPD,21–23 demonstrating that sufficient bronchodilation has beneficial effects beyond just improving pulmonary function.21,22
Previous studies have not found any adverse effects on HR or BP with olodaterol monotherapy.24 In addition, analyses looking at Holter electrocardiogram (ECG) data from clinical trials of tiotropium and olodaterol in patients with COPD found that these LAMA and LABA monotherapies were not associated with arrhythmia or persistent changes in HR compared with placebo.25,26 Moreover, results from a large meta-analysis indicate that LAMA/LABA therapies have a good cardiovascular safety profile.27 However, there are no published studies investigating the effects of tiotropium/olodaterol versus monocomponents on HR and BP.
This post hoc analysis, the first to comprehensively investigate HR and BP in a large Phase III study of dual bronchodilation, will investigate the effect of tiotropium/olodaterol versus monocomponents on HR and BP in patients with moderate-to-very-severe COPD included in the large TONADO® study population.
Methods
Study Designs
The TONADO® trials (1237.5 [NCT01431274] and 1237.6 [NCT01431287]) were two replicate, randomized, double-blind, parallel-group, 52-week, Phase III trials that compared tiotropium/olodaterol (5/5 μg and 2.5/5 μg) with the monocomponents tiotropium (5 μg and 2.5 μg) and olodaterol (5 μg) (all via the Respimat® inhaler, two puffs once daily) in patients with moderate-to-very-severe COPD. A detailed study design and methodology has been published previously.4 In this analysis, we will focus only on the licensed doses; therefore, only data for tiotropium/olodaterol 5/5 µg, tiotropium 5 µg and olodaterol 5 µg are presented.
The main inclusion criteria, as stated in the TONADO® trials, were: outpatients aged ≥40 years with a history of moderate-to-very-severe COPD (GOLD stage 2–4); post-bronchodilator forced expiratory volume in 1 second (FEV1) <80% predicted normal and post-bronchodilator FEV1/forced vital capacity <70% (European Community for Steel and Coal28); and current or ex-smokers with a smoking history of >10 pack-years.
Patients were excluded from the TONADO® trials if they had a significant disease other than COPD, had clinically relevant abnormal baseline parameters or a history of asthma. Patients were also excluded if they had a diagnosis of paroxysmal tachycardia; myocardial infarction within 1 year of screening; had unstable/life-threatening cardiac arrhythmia; or had been hospitalized due to cardiac failure within the previous year.
Assessments
In the TONADO® studies, vital signs (including BP) and ECGs (including HR) were repeated at Visits 2, 5 and 7 (baseline [Day 0], Week 12 [Day 85] and Week 24 [Day 169], respectively), and on completion of the randomized treatment period of the trial at Visit 10 (Week 52 [Day 365]). For detection of acute changes at the time of inhalational dosing, HR was measured 30 minutes pre-dose and 40 minutes post-dose and systolic/diastolic BP were measured at 60 minutes pre-dose and 60 minutes post-dose at all visit time points, each prior to pulmonary function and other scheduled tests. Measurements were obtained with the patient lying in supine position (ECGs/HR) or seated (BP) having rested for a minimum of 5 minutes. Centralized ECGs were completed using equipment provided by eResearch Technology (Mortara ELI250 12-Lead Resting ECG Machine, Milwaukee, WI, USA). Data were transmitted to, and read by, a cardiologist at eResearch Technology. HRs were taken from the ECG summary report of each examination, calculated as a mean value from a 10-s rhythm strip (lead II).
Statistical Analysis
HR and BP were analyzed at the planned time points by means of a mixed effect model for repeated measures, similar to that used for the primary analysis of the TONADO® trials.4 The model was adjusted by fixed effects including treatment, visit, treatment-by-visit interaction, baseline and baseline-by-visit interaction. Patients were considered random, and an unstructured covariance structure was used. Changes from pre-dose baseline, and from pre-dose to post-dose measurement, within the treatment arms were considered statistically significant if the 95% confidence interval did not include zero, corresponding to P<0.05.
Results
Baseline Demographics
This post hoc analysis included 3,100 patients. Baseline demographics are presented in Table 1. Most patients were male (72.9%) and the overall mean age was 64.0 years. All patients were either ex-smokers (63.0%) or current smokers (37.0%), and the mean post-bronchodilator FEV1 at screening was 1.37 L (49.8% of predicted normal). In the olodaterol 5 µg, tiotropium 5 µg and tiotropium/olodaterol 5/5 µg groups, the proportions of cardiac arrhythmias were 1.3%, 0.4% and 0.7%, respectively. For coronary artery disease, proportions were 6.3%, 5.1% and 4.7%, respectively. Cardiovascular medication, such as β-blockers, was represented in all treatment groups (olodaterol 5 µg 9.8%; tiotropium 5 µg 10.6%; tiotropium/olodaterol 5/5 µg 10.7%). There were no significant differences across the baseline characteristic groups.
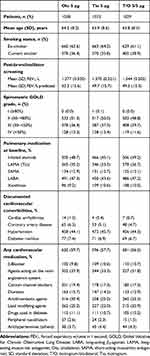 |
Table 1 Demographic and Baseline Patient Characteristics by Treatment Group for the 52-Week, Parallel-Group Trials (Treated Patient Population) |
Heart Rate
Long-Term Mean Change in HR from Baseline Over 52 Weeks
For the long-term mean change in HR, there was no significant increase from baseline with tiotropium/olodaterol or olodaterol; however, there was a very small increase with tiotropium at 52 weeks (<2 beats per minute [bpm]) (Figure 1). The long-term mean changes in HR from baseline, across all time points and treatment groups, ranged from –1.3 to +1.2 bpm. There was a slight increase in mean HR from Week 12 to 52 in all treatment groups: the mean change from baseline in HR was 0.3 bpm (P=0.32) with tiotropium/olodaterol 5/5 µg, 0.2 bpm (P=0.56) with olodaterol 5 µg, and 1.2 bpm (P=0.0001) with tiotropium 5 µg at Week 52. Tiotropium/olodaterol 5/5 µg did not increase HR when compared with monocomponents.
Short-Term (30 Min Pre- to 40 Min Post-Dose) Mean Change in HR from Pre- to Post-Dose Over 52 Weeks
Mean change in HR from 30 minutes pre-dose to 40 minutes post-dose was <1 bpm with all treatments over 52 weeks (Figure 2). At Week 52, there was a mean change in HR from pre-dose to post-dose of 0.8 bpm (P=0.0006) with tiotropium/olodaterol 5/5 µg, a mean change of –0.1 bpm (P=0.67) with olodaterol 5 µg, and a mean change of –0.3 bpm (P=0.25) with tiotropium 5 µg. Tiotropium/olodaterol 5/5 µg was associated with a small HR increase (<1 bpm) at all visits; this increase was not present for tiotropium 5 µg or olodaterol 5 µg.
Blood Pressure
Long-Term Mean Change in Systolic and Diastolic BP from Baseline Over 52 Weeks
There was no increase in mean diastolic or systolic BP from baseline with any treatment observed over 52 weeks (Figure 3). Systolic BP changes in the tiotropium/olodaterol 5/5 µg group were similar to the monocomponents. At Week 52, tiotropium/olodaterol 5/5 µg showed an adjusted mean change from baseline of –1.34 mmHg (P=0.0023), olodaterol 5 µg showed a change of –1.22 mmHg (P=0.0071) and tiotropium 5 µg showed a change of –0.76 mmHg (P=0.0916). With diastolic BP, there was no increase in BP with the combination of tiotropium/olodaterol 5/5 µg versus the monocomponents. The long-term mean changes in diastolic BP from baseline, across all time points and treatment groups, ranged from –1.25 to –0.01 mmHg. At Week 52, tiotropium/olodaterol 5/5 µg showed an adjusted mean change from baseline of –1.00 mmHg (P=0.0003), whereas olodaterol 5 µg changed by –1.02 mmHg (P=0.0003) and tiotropium 5 µg by –0.40 mmHg (P=0.16).
Short-Term (1 Hour Pre- to 1 Hour Post-Dose) Mean Change in Systolic and Diastolic BP Over 52 Weeks
The short-term (1 hour pre- to 1 hour post-dose) mean changes in systolic and diastolic BP over 52 weeks in the tiotropium/olodaterol 5/5 µg group were generally similar to those observed for the monocomponents at all time points (Figure 4). The short-term mean change from pre- to post-dose in systolic BP from baseline, across all time points and treatment groups, ranged from –1.88 mmHg to –0.59 mmHg. At Week 52, tiotropium/olodaterol 5/5 µg showed a change from pre- to post-dose of –0.97 mmHg (P=0.0028), olodaterol 5 µg showed a change of –0.59 mmHg (P=0.0785), and tiotropium 5 µg showed a change of –1.88 mmHg (P<0.0001). The short-term mean change in diastolic BP from pre- to post-dose, across all time points and treatment groups, ranged from –1.21 mmHg to –0.75 mmHg. At Week 52, tiotropium/olodaterol 5/5 µg showed an adjusted mean change from pre- to post-dose of –0.81 mmHg (P=0.0003), olodaterol 5 µg showed a change of –1.21 mmHg (P<0.0001), and tiotropium 5 µg showed a change of –1.00 mmHg (P<0.0001).
Notable and Marked Changes
An analysis of notable/marked changes in HR and BP was performed to show the more extreme values and more extreme variation. Less than 1.5% of all patients recorded notable/marked HR increases pre-dose (n=26/2,537) or post-dose (n=26/2,537), defined as ≥25% increase from baseline and on-treatment HR >100 bpm, whereas <0.5% (n=7/2,537 pre-dose; n=9/2,537 post-dose) recorded notable/marked HR decreases, defined as ≥25% decrease from baseline and on-treatment HR <50 bpm. Less than 3% of all patients recorded notable/marked BP decreases, either in terms of systolic BP (<100 mmHg and a decrease >10 mmHg below baseline; n=47/3,077) or diastolic BP (<60 mmHg and a decrease of >10 mmHg below baseline; n=45/3,077). Notable/marked BP increases were reported in <5% of patients, both for systolic BP (>150 mmHg and an increase ≥25 mmHg above baseline; n=83/3,077) and diastolic BP (>90 mmHg and an increase >10 mmHg above baseline; n=117/3,077). The results showed that there was no indication of an additive effect of tiotropium/olodaterol 5/5 µg versus monotherapies.
Discussion
To our knowledge, this is the first study to comprehensively investigate HR and BP in a large Phase III study of dual bronchodilation. The changes in HR and BP are all quantitatively small; there was only a small increase from baseline with tiotropium at 52 weeks (<2 bpm) for long-term HR, and a very small increase in HR pre- to post-dose for tiotropium/olodaterol 5/5 µg (<1 bpm). These results, whilst statistically significant, represent very small changes in absolute values, which may indicate disease progression and can be affected by many factors including the age and fitness of the patients. There were only small changes in mean BP <2 mmHg observed over 52 weeks of treatment. There was no sign of increased effects on HR and BP with tiotropium/olodaterol versus the monocomponents.
It has been suggested that treatment with LAMA/LABA can have negative cardiovascular effects19 and recent heart failure guidelines suggest that “Long-term safety of cardioactive inhaled pulmonary drugs is uncertain and the need for their use should be reconsidered in patients with heart failure with reduced ejection fraction.”29
Data from a healthcare database study have also suggested there may be adverse cardiovascular effects associated with the initiation of either LABA or LAMA therapy.30 However, a pooled analysis of four studies showed that tiotropium was not associated with changes in HR versus placebo; similarly, olodaterol has been shown to have no adverse effects on HR or BP.24,25 In the present study, combination tiotropium/olodaterol therapy had little or no clinically meaningful effect on HR or BP when compared with monocomponents over 52 weeks in patients with GOLD 2–4 COPD. This is in line with previous studies investigating other LAMA/LABA combinations, which also concluded that the rate of cardiovascular adverse events was comparable with both placebo and monotherapies.31,32 Furthermore, studies assessing combined LABA/ICS treatment have shown it to be well tolerated, with no adverse effects on mortality or cardiovascular outcomes.33,34 Some studies, such as the CLAIM study21 and a further study by Stone et al,22 have also suggested that bronchodilation can have a positive effect on heart function by reducing hyperinflation. In these studies, cardiac function was significantly improved in patients treated with indacaterol/glycopyrronium (P<0.0001) or fluticasone furoate/vilanterol (P<0.001) compared with patients treated with placebo.21,22
Previous analyses comparing tiotropium/olodaterol 5/5 µg with tiotropium and olodaterol monotherapies in patients participating in the TONADO® studies showed that there were no dose- or time-related trends in QT interval change across treatment groups; therefore, there was no concern with QT prolongation.35 Additionally, Holter ECG data have shown that patients treated with tiotropium or olodaterol have no increased risk of cardiac arrhythmia versus placebo;25,26 Holter ECG analysis of dual bronchodilation with tiotropium/olodaterol has also found no increased incidence of cardiac arrhythmia compared with the monocomponents.36 No statistically significant or clinically relevant shift from baseline (ie, proportion of patients whose premature supraventricular or ventricular beats increased or decreased from baseline) was observed with tiotropium/olodaterol combination or its monocomponents.25,26,36 In our analysis, all treatment arms had a slight increase in HR over the 52-week study period, which could be due to the progression of COPD severity over time. It is also known that increases in HR are associated with an aging population. This slight increase in HR is comparable with previous published studies.24
The strengths of the present analysis include the large size of the TONADO® trials, and the inclusion of patients with cardiovascular comorbidities and those receiving cardiovascular medication; 58% of all patients were receiving cardiovascular medication, including 10% on β-blockers. We also used highly standardized methodologies for collection of HR and BP data across all centers, and the trials were blinded and active-comparator-controlled. A limitation of the analysis was that patients with very severe or unstable conditions were excluded, therefore data may not be representative of patients with extreme individual conditions encountered in clinical practice. Furthermore, resting HR and BP are not necessarily reflective of variability during the daily activity of the patients, and there was no placebo group analyzed, thus limiting interpretation.
Conclusions
There were no differences in HR or BP among patients on tiotropium/olodaterol when compared with monocomponents. This supports the already demonstrated cardiovascular safety profile of tiotropium/olodaterol as maintenance treatment for COPD, including patients with cardiovascular comorbidities.
Abbreviations
BP, blood pressure; bpm, beats per minute; COPD, chronic obstructive pulmonary disease; ECG, electrocardiogram; FEV1, forced expiratory volume in 1 second; GOLD, Global Initiative for Chronic Obstructive Lung Disease; HR, heart rate; ICS, inhaled corticosteroid; LAMA, long-acting muscarinic antagonist; LABA, long-acting β2-agonist.
Data Sharing Statement
Data are available from the corresponding author upon reasonable request.
Ethics Approval and Informed Consent
All studies included in this analysis were performed in accordance with the provisions of the Declaration of Helsinki (1996 version), the International Conference on Harmonisation Tripartite Guideline for Good Clinical Practice, and applicable regulatory requirements and Boehringer Ingelheim Standard Operating Procedures. All patients provided written informed consent. This article does not report individual patient data; all data presented here are anonymized. The clinical trial protocols and the informed consent and patient information forms were reviewed and received approval/favorable opinion from a constituted local Institutional Review Board or an Independent Ethics Committee at each center prior to the start of the study. Approval was granted from institutional review boards (Coordinating Investigator’s [Prof. Roland Buhl] Independent Ethics Committee: Ethik-Kommission bei der Landesärztekammer Rheinland-Pfalz, Deutschhausplatz 3, 55,116 Mainz, Germany).
Acknowledgments
Medical writing assistance, in the form of the preparation and revision of the manuscript, was supported financially by Boehringer Ingelheim and provided by Francesca Lomas of MediTech Media (Manchester, UK), based on a draft provided by the authors, their feedback and under their conceptual direction.
Author Contributions
All the authors have made substantial contributions to study design, data acquisition, analysis or interpretation, drafting the article, or critically revising the content, provided final approval of the submitted version and agree to be accountable for all aspects of the work.
Disclosure
SA reports personal fees from Boehringer Ingelheim and GlaxoSmithKline, and payments for presenting from Boehringer Ingelheim, AstraZeneca, Berlin Chemie, Chiesi and Novartis, outside the submitted work. LM reports personal fees and non-financial support from Boehringer Ingelheim and personal fees from Applied Clinical Intelligence, during the conduct of the study; personal fees from Merck, Afferent, AstraZeneca, Bellus Health and European Union Interreg VA Health & Life Science Programme, grants, personal fees and non-financial support from Bionorica, grants and non-financial support from Chiesi, and personal fees and non-financial support from GlaxoSmithKline, outside the submitted work. UB, MT and AdlH are employees of Boehringer Ingelheim. RB reports grants to Mainz University and personal fees from Boehringer Ingelheim, GlaxoSmithKline, Novartis, and Roche, as well as personal fees from AstraZeneca, Chiesi, Cipla, Sanofi and Teva, outside the submitted work. PA reports grants from the German Federal Ministry of Education and Research (BMBF) Competence Network Asthma and COPD (ASCONET), AstraZeneca, GlaxoSmithKline, Grifols Deutschland, MSD Sharp & Dohme, Pfizer, Takeda, Boehringer Ingelheim and Novartis Deutschland, grants and non-financial support from Bayer Schering Pharma AG and Chiesi, grants, personal fees and non-financial support from Novartis Deutschland, and grants and personal fees from Novartis Deutschland, outside the submitted work. The authors report no other conflicts of interest in this work.
References
1. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease (2019 report). Published 2019. Available from: https://goldcopd.org/wp-content/uploads/2018/11/GOLD-2019-v1.7-FINAL-14Nov2018-WMS.pdf.
2. Tashkin DP, Celli B, Senn S, et al. A 4-year trial of tiotropium in chronic obstructive pulmonary disease. N Engl J Med. 2008;359(15):1543–1554. doi:10.1056/NEJMoa0805800
3. Koch A, Pizzichini E, Hamilton A, et al. Lung function efficacy and symptomatic benefit of olodaterol once daily delivered via Respimat® versus placebo and formoterol twice daily in patients with GOLD 2–4 COPD: results from two replicate 48-week studies. Int J Chron Obstruct Pulmon Dis. 2014;9:697–714. doi:10.2147/COPD.S62502
4. Buhl R, Maltais F, Abrahams R, et al. Tiotropium and olodaterol fixed-dose combination versus mono-components in COPD (GOLD 2-4). Eur Respir J. 2015;45(4):969–979. doi:10.1183/09031936.00136014
5. Feary JR, Rodrigues LC, Smith CJ, Hubbard RB, Gibson JE. Prevalence of major comorbidities in subjects with COPD and incidence of myocardial infarction and stroke: a comprehensive analysis using data from primary care. Thorax. 2010;65(11):956–962. doi:10.1136/thx.2009.128082
6. Fuhrman C, Jougla E, Nicolau J, Eilstein D, Delmas MC. Deaths from chronic obstructive pulmonary disease in France, 1979–2002: a multiple cause analysis. Thorax. 2006;61(11):930–934. doi:10.1136/thx.2006.061267
7. Huiart L, Ernst P, Suissa S. Cardiovascular morbidity and mortality in COPD. Chest. 2005;128(4):2640–2646. doi:10.1378/chest.128.4.2640
8. Karch A, Vogelmeier C, Welte T, et al. The German COPD cohort COSYCONET: aims, methods and descriptive analysis of the study population at baseline. Respir Med. 2016;114:27–37. doi:10.1016/j.rmed.2016.03.008
9. Alter P, Mayerhofer B, Kahnert K, et al. Prevalence of cardiac comorbidities, and their underdetection and contribution to exertional symptoms in COPD: results from the COSYCONET cohort. Int J Chron Obstruct Pulmon Dis. 2019;14:2163–2172. doi:10.2147/COPD.S209343
10. Sidney S, Sorel M, Quesenberry CP, DeLuise C, Lanes S, Eisner MD. COPD and incident cardiovascular disease hospitalizations and mortality: Kaiser Permanente Medical Care Program. Chest. 2005;128(4):2068–2075. doi:10.1378/chest.128.4.2068
11. Byrd JB, Newby DE, Anderson JA, et al. Blood pressure, heart rate, and mortality in chronic obstructive pulmonary disease: the SUMMIT trial. Eur Heart J. 2018;39(33):3128–3134. doi:10.1093/eurheartj/ehy451
12. Andreas S, Anker SD, Scanlon PD, Somers VK. Neurohumoral activation as a link to systemic manifestations of chronic lung disease. Chest. 2005;128(5):3618–3624. doi:10.1378/chest.128.5.3618
13. Cohn JN, Levine TB, Olivari MT, et al. Plasma norepinephrine as a guide to prognosis in patients with chronic congestive heart failure. N Engl J Med. 1984;311(13):819–823. doi:10.1056/NEJM198409273111303
14. Jensen MT, Suadicani P, Hein HO, Gyntelberg F. Elevated resting heart rate, physical fitness and all-cause mortality: a 16-year follow-up in the Copenhagen Male Study. Heart. 2013;99(12):882–887. doi:10.1136/heartjnl-2012-303375
15. Andreas S, Haarmann H, Klarner S, Hasenfuss G, Raupach T. Increased sympathetic nerve activity in COPD is associated with morbidity and mortality. Lung. 2014;192(2):235–241. doi:10.1007/s00408-013-9544-7
16. Schrier RW, Abraham WT. Hormones and hemodynamics in heart failure. N Engl J Med. 1999;341(8):577–585. doi:10.1056/NEJM199908193410806
17. Bangalore S, Messerli FH, Wun CC, et al. J-curve revisited: an analysis of blood pressure and cardiovascular events in the Treating to New Targets (TNT) trial. Eur Heart J. 2010;31(23):2897–2908. doi:10.1093/eurheartj/ehq328
18. Messerli FH, Panjrath GS. The J-curve between blood pressure and coronary artery disease or essential hypertension: exactly how essential? J Am Coll Cardiol. 2009;54(20):1827–1834. doi:10.1016/j.jacc.2009.05.073
19. Lahousse L, Verhamme KM, Stricker BH, Brusselle GG. Cardiac effects of current treatments of chronic obstructive pulmonary disease. Lancet Respir Med. 2016;4(2):149–164. doi:10.1016/S2213-2600(15)00518-4
20. Cazzola M, Imperatore F, Salzillo A, et al. Cardiac effects of formoterol and salmeterol in patients suffering from COPD with preexisting cardiac arrhythmias and hypoxemia. Chest. 1998;114(2):411–415. doi:10.1378/chest.114.2.411
21. Hohlfeld JM, Vogel-Claussen J, Biller H, et al. Effect of lung deflation with indacaterol plus glycopyrronium on ventricular filling in patients with hyperinflation and COPD (CLAIM): a double-blind, randomised, crossover, placebo-controlled, single-centre trial. Lancet Respir Med. 2018;6(5):368–378. doi:10.1016/S2213-2600(18)30054-7
22. Stone IS, Barnes NC, James WY, et al. Lung deflation and cardiovascular structure and function in chronic obstructive pulmonary disease: a randomized controlled trial. Am J Respir Crit Care Med. 2016;193(7):717–726. doi:10.1164/rccm.201508-1647OC
23. Vogel-Claussen J, Schonfeld CO, Kaireit TF, et al. Effect of indacaterol/glycopyrronium on pulmonary perfusion and ventilation in hyperinflated COPD patients (CLAIM): a double-blind, randomised, crossover trial. Am J Respir Crit Care Med. 2019;199(9):1086–1096. doi:10.1164/rccm.201805-0995OC
24. Andreas S, Bothner U, Trampisch M, Haensel M, Buhl R, Alter P. Effect of long-acting β2-agonists olodaterol and formoterol on heart rate and blood pressure in chronic obstructive pulmonary disease patients. Pulm Pharmacol Ther. 2018;52:1–6. doi:10.1016/j.pupt.2018.08.002
25. Hohlfeld JM, Furtwaengler A, Konen-Bergmann M, Wallenstein G, Walter B, Bateman ED. Cardiac safety of tiotropium in patients with COPD: a combined analysis of Holter-ECG data from four randomised clinical trials. Int J Clin Pract. 2015;69(1):72–80. doi:10.1111/ijcp.12596
26. Andreas S, Bothner U, de la Hoz A, Kloer I, Trampisch M, Alter P. A post hoc Holter ECG analysis of olodaterol and formoterol in moderate-to-very-severe COPD. Int J Chron Obstruct Pulmon Dis. In press 2020.
27. Rogliani P, Matera MG, Ora J, Cazzola M, Calzetta L. The impact of dual bronchodilation on cardiovascular serious adverse events and mortality in COPD: a quantitative synthesis. Int J Chron Obstruct Pulmon Dis. 2017;12:3469–3485. doi:10.2147/COPD.S146338
28. Quanjer PH, Tammeling GJ, Cotes JE, Pedersen OF, Peslin R, Yernault JC. Lung volumes and forced ventilatory flows. Eur Respir J. 1993;6(suppl 16):5–40. doi:10.1183/09041950.005s1693
29. Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016;37(27):2129–2200. doi:10.1093/eurheartj/ehw128
30. Gershon A, Croxford R, Calzavara A, et al. Cardiovascular safety of inhaled long-acting bronchodilators in individuals with chronic obstructive pulmonary disease. JAMA Intern Med. 2013;173(13):1175–1185. doi:10.1001/jamainternmed.2013.1016
31. Blair HA, Deeks ED. Umeclidinium/vilanterol: a review of its use as maintenance therapy in adults with chronic obstructive pulmonary disease. Drugs. 2015;75(1):61–74. doi:10.1007/s40265-014-0326-1
32. Kozielski J. Cardiovascular safety of two bronchodilators’ fixed-dose combination: indacaterol and glycopyrronium. Pneumonol Alergol Pol. 2015;83(2):164–170. doi:10.5603/PiAP.2015.0026
33. Vestbo J, Leather D, Diar Bakerly N, et al. Effectiveness of fluticasone furoate-vilanterol for COPD in clinical practice. N Engl J Med. 2016;375(13):1253–1260. doi:10.1056/NEJMoa1608033
34. Calverley PM, Anderson JA, Celli B, et al. Cardiovascular events in patients with COPD: TORCH study results. Thorax. 2010;65(8):719–725. doi:10.1136/thx.2010.136077
35. Buhl R, Magder S, Bothner U, et al. Long-term general and cardiovascular safety of tiotropium/olodaterol in patients with moderate to very severe chronic obstructive pulmonary disease. Respir Med. 2017;122:58–66. doi:10.1016/j.rmed.2016.11.011
36. Andreas S, Bothner U, de la Hoz A, Kloer I, Trampisch M, Alter P. No influence on cardiac arrhythmia or heart rate from long-term treatment with tiotropium/olodaterol versus monocomponents by holter ECG analysis in patients with moderate-to-very-severe COPD. Int J Chron Obstruct Pulmon Dis. In press 2020.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




