Back to Journals » Clinical Epidemiology » Volume 11
A systematic review of the international prevalence of BRCA mutation in breast cancer
Authors Armstrong N , Ryder S , Forbes C , Ross J , Quek RGW
Received 27 February 2019
Accepted for publication 23 May 2019
Published 11 July 2019 Volume 2019:11 Pages 543—561
DOI https://doi.org/10.2147/CLEP.S206949
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Henrik Sørensen
Nigel Armstrong,1 Steve Ryder,1 Carol Forbes,2 Janine Ross,3 Ruben GW Quek4
1Health Economics, Kleijnen Systematic Reviews Ltd., York YO19 6FD, UK; 2Systematic Reviews, Kleijnen Systematic Reviews Ltd., York YO19 6FD, UK; 3Information, Kleijnen Systematic Reviews Ltd., York YO19 6FD, UK; 4Health Economics & Outcomes Research, Pfizer Inc., San Francisco, CA 94105, USA
Abstract: A systematic review was conducted, summarizing international BRCA 1 or 2 (BRCA1/2) mutation prevalence in breast cancer. Databases (eg, Medline and Embase; N=7) and conferences were searched (January 2012 to December 2017). From 17,872 records, 70 studies were included. In 58 large (N>100) studies, BRCA1/2 mutation prevalence varied widely from 1.8% (Spain) in sporadic breast cancer to 36.9% (United States) in estrogen receptor/progesterone receptor low+ (1–9% on immunohistochemistry/human epidermal growth factor receptor 2–negative [HER2-]) breast cancer. In 2 large studies unselected for family history, ethnicity, sex, or age and no/unclear selection by breast cancer stage or hormone receptor (HR) status, germline BRCA (gBRCA) mutation prevalence was 2.9% (Italy) to 3.0% (South Korea). In the 4 large unselected triple-negative breast cancer studies, gBRCA mutation prevalence varied from 9.3% (Australia) to 15.4% (United States). gBRCA mutation prevalence in 1 large unselected HR positive/HER2- early breast cancer study was 5% (United States). In 2 large unselected metastatic breast cancer studies, gBRCA mutation prevalence was 2.7% (France) and 4.3% (Germany). Locally advanced breast cancer studies were small and not in unselected populations. Poor reporting of gBRCA status and basis of selection implies a need for further large well-reported BRCA mutation prevalence studies in breast cancer.
Keywords: BRCA1, BRCA2, prevalence, systematic review, chemotherapy
Introduction
Breast cancer is a major health burden globally; it is the second most common cancer worldwide and the most common cause of cancer death in women.1 The disease is multifactorial and thought to result from interactions between a number of different environmental, lifestyle, hormonal, and genetic factors, including a family history of breast cancer (hereditary breast cancer). A wealth of evidence indicates that mutations in the key tumor suppressor genes—the breast cancer susceptibility genes 1 or 2 (BRCA1/2)—predisposes an individual to developing breast cancer.2 Such mutations may be inherited (germline) or arise as a result of a combination of genetic and environmental factors (somatic).2 Specific subgroups have been identified as having a higher proportion of individuals who carry a BRCA mutation, including those who have been diagnosed with triple-negative breast cancer (TNBC) and those from different ethnic groups, including Black populations and those of Ashkenazi Jewish heritage.3,4
BRCA proteins play a key role in the DNA damage response, an essential pathway that ensures the survival of both normal and malignant breast cells.5 Patients who carry a high-risk mutation in 1 or both of the BRCA genes (BRCA1 or BRCA2) have a significantly increased risk of developing breast cancer and other cancers (eg, ovarian or prostate cancer).2,4 For those who go on to develop advanced breast cancer (aBC), the newly developed poly adenosine diphosphate–ribose polymerase inhibitors (PARPi) offer a new targeted approach to specifically treat those with germline BRCA1/2 mutations.6 Recently, olaparib became the first of the PARPi drugs to receive approval by the United States Food and Drug Administration (FDA) for use in the treatment of patients with germline BRCA (gBRCA) mutation and human epidermal growth factor receptor 2–negative (HER2-) metastatic breast cancer; talazoparib was approved by the FDA in 2018 and as of this writing is undergoing review by the European Medicines Agency.
Given recent developments in the management of those with BRCA-mutated breast cancer,4,7–10 it is important that international healthcare providers and decision makers are kept informed of the burden of BRCA-mutated disease and the prevalence of the population that would potentially benefit from current and future BRCA mutation-targeted therapeutic options.
The objective of this systematic review was to identify and summarize the latest prevalence of BRCA mutations (including gBRCA mutations wherever specified) in the breast cancer population, focusing on those individuals who are potential targets for BRCA mutation-targeted therapies across a number of countries, specifically Australia, Canada, France, Germany, Israel/Palestine, Italy, Japan, Russia, South Korea, Spain, United Kingdom, and United States.
Methods
This systematic review followed the recommendations of the Centre for Reviews and Dissemination (CRD) guidance for undertaking reviews in healthcare and the Cochrane Handbook for Systematic Reviews of Interventions.11,12 This systematic review was also conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Table S1).
A range of electronic databases was searched (N=7), including Medline, Embase, EconLit, and the Cochrane Database of Systematic Reviews. Searches used a combination of text and database thesaurus terms. Searches of gray literature sources, including conference abstracts, were also conducted. Full details of resources and strategies used are available Appendix 1 in the Supplementary materials.
This manuscript includes observational studies reporting on the prevalence of BRCA mutations (BRCA1, BRCA2, or BRCA1/2), including germline mutations (wherever specified) in male or female breast cancer patients. The prevalence of any mutation was included regardless of whether the mutation was a founder mutation or not. Also, where study authors did not clearly state that mutations were germline or somatic and/or deleterious, pathogenic, or clinically relevant, the mutation was classified as not reported/unclear in order to avoid any misinterpretation. Study inclusion was not limited by language. Only data that were publicly available and reported from January 2012 to December 2017 were eligible for inclusion. This ensured that data were as relevant to current clinical practice as possible, given the rapidly evolving nature of the management of patients with breast cancer associated with BRCA mutation.
Data from the included studies were extracted, stored, and analyzed using Microsoft Excel 2010 (Microsoft Corporation, Redmond, WA) spreadsheets. The principal summary measure that was extracted was percentage prevalence. Given the large amount of data in the topic area, we focused our findings on data from 12 countries: United States, Canada, United Kingdom, Germany, France, Spain, Italy, Australia, Japan, South Korea, Russia, and Israel. Details of the study methods, population characteristics, risk of bias, and prevalence data were extracted and summarized from each study by 1 reviewer and checked for accuracy against the original publication by a second reviewer. Any discrepancies were resolved through consensus or consultation with a third reviewer. Criteria used to assess the risk of bias were taken from the critical appraisal checklist for studies reporting prevalence data of the Joanna Briggs Institute, which assesses the risk at the study level.13
Studies were grouped by country. In addition, within the broader population of interest (patients with gBRCA mutation breast cancer), data have been highlighted and discussed separately for subgroups of patients that were of particular interest: locally advanced/metastatic TNBC and locally advanced/metastatic HR–positive (HR+) and HER2- breast cancer.
In order to focus on the most robust data, we have centered our report mainly on the results of 58 large studies (N>100). Nevertheless a full set of results from all 70 studies that met the inclusion criteria is reported in Appendix 2 of the Supplementary materials.
Results
Study selection
A total of 17,872 titles and abstracts were retrieved from the literature searches and an additional 6 from hand searching reference lists, background papers, and systematic reviews. From these, full papers were obtained for 269 citations. After further review, 88 papers were excluded; the reason(s) for exclusion are listed in Appendix 3 of the Supplementary materials.
From the remaining 181 papers, 73 papers reported BRCA mutation prevalence data for 70 studies across the 12 countries. A summary of the study selection process is reported in Figure 1.
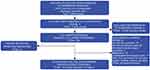 |
Figure 1 PRISMA flow chart detailing literature searches and inclusion screening. |
Prevalence studies
Risk of bias of prevalence studies
Only 3 of the 70 prevalence studies were assessed as at low risk of bias (green shading in Figure 2) on all 10 criteria according to the Joanna Briggs Institute checklist.13–16 Twenty-seven studies of the 70 (37.5%) had no criteria at high risk of bias (blue shading in Figure 2), but there was at least 1 criterion for which the risk of bias was unclear (red shading in Figure 2).14–40 Particular areas of concern across the studies reporting on BRCA status that may affect the cumulative evidence on BRCA prevalence included no adequate description of the source population and setting (n=28; 40.0% of studies), inadequate sample size (n=12; 17.1 of studies), and sample population not necessarily representative of the total population of patients (n=11; 15.7% of studies).
 |
Figure 2 Summary of JBI risk of bias assessment for prevalence studies. Abbreviations: JBI, Joanna Briggs Institute. |
A summary of the risk of bias and further details are reported in Appendix 4 in the Supplementary materials.
Overview of prevalence studies
Seventy studies reported the prevalence of BRCA1 and/or BRCA2 mutations in patients with breast cancer in the following countries: United States (33 studies), Canada (2 studies), United Kingdom (4 studies), Germany (3 studies), France (2 studies), Spain (4 studies), Italy (3 studies [including 1 study from Sardinia]), Australia (2 studies), Japan (1 study), South Korea (11 studies), Russia (2 studies), and Israel (3 studies [including 1 study from Palestine]).
gBRCA mutation prevalence was reported explicitly in only 32 of the 70 studies; the majority of studies did not make it clear whether mutations were germline or somatic. Also, only 45 of the 70 studies specified whether the BRCA mutations were deleterious (or clinically significant) or not. Forty-four of the 70 studies reported separate prevalence data for BRCA1 and BRCA2, including whether a proband carried both BRCA1 and BRCA2 mutations. Also, 4 of these 44 studies did not report this information fully for all subgroups.14,18,41,42 Most studies (62) reported the prevalence of any BRCA mutation, ie, BRCA1 or BRCA2 as opposed to only BRCA1 or only BRCA2.
Nine of the studies failed to report in sufficient detail how individuals were selected for inclusion in the study population. A total of 17 studies reported data for a population explicitly stated to be unselected for family history of breast cancer, 7 studies categorized patients as mixed (some with and others without a family history of breast cancer), 7 studies purely focused on patients with a family history of breast cancer, and 38 studies reported that probands were selected on criteria other than only family history, such as sex, ethnicity, and age at breast cancer diagnosis. When studies were reported as mixed, they included both those individuals with and those without a family history of breast cancer, but they did not report that probands were unselected for family history. Such mixed studies reported on samples that might not have the same proportion of those individuals with a family history as those designed specifically to be unselected for family history. No study reported BRCA mutation prevalence solely in men, 31 reported data for TNBC patients, and 4 reported data for patients with HR+/HER2- disease (2 of which were in a subgroup of those with HR low+ [1–9% on immunohistochemistry]). Seven studies reported on the prevalence of metastatic breast cancer, and only 2 reported on locally aBC. Three studies reported prevalence for multiple subgroups.18,43,44 Eighteen studies did not report data on either HR status or breast cancer stage.
Of the 31 studies reporting on TNBC, there was variation in terms of breast cancer stage, including populations of mixed cancer stages and those for whom the stage of disease was not reported. Twelve studies included populations that were unselected on the basis of family history or mixed (with and without) family history. Only 1 study45 reported BRCA mutation for aBC, and these data were only for a small subgroup of United States patients with metastatic breast cancer (metastatic TNBC), including those with and without a family history of breast cancer.
For the 4 studies that reported BRCA mutation in patients with HR+/HER2- breast cancer, none was in the same population in terms of criteria used to select patients. One of the studies required patients to fulfill a complex set of selection criteria in accordance with the National Medical Insurance Reimbursement in South Korea,15 and another only included females with early breast cancer.46
The 7 studies that reported BRCA mutation in metastatic breast cancer included only 1 study in a population with a specific HR status, which was in TNBC45 (as mentioned above) in the United States.
Neither of the 2 studies in patients with locally aBC reported on the HR status of participants. One United States study43 included patients who varied with respect to their family history of breast cancer, and the other study (from Israel44) included data reported according to the different BRCA mutation proband (BRCA1 and BRCA2) for patients who were female, of Ashkenazi Jewish descent, and diagnosed with early onset breast cancer.
Twenty-five studies reported details on the method used to screen for BRCA mutations. Where reported, most studies used either direct sequencing (8 studies) or next-generation sequencing (NGS) methods (8 studies). Seven studies reported testing for only a subset of BRCA-associated mutations .3,31,44,47–51
A summary of the key study characteristics is provided in Table 1.3,14–22,24–82 Further characteristics of the included studies are provided in Appendix 2 of the Supplementary materials.
 |
Table 1 Summary of key study characteristics |
 |
Table 1 (Continued). |
 |
Table 1 (Continued). |
 |
Table 1 (Continued). |
 |
Table 1 (Continued). |
 |
Table 1 (Continued). |
 |
Table 1 (Continued). |
Summary of BRCA mutation prevalence data
Prevalence between individual studies varied widely. In the 58 large (N>100) studies, the lowest prevalence was reported as 1.8% for deleterious gBRCA mutation in a Spanish study (N=495) of sporadic breast cancer cases within a population with no family history or other criteria warranting hereditary breast cancer screening and a mixture of patients with different HR profiles.18 In contrast, the highest reported prevalence was 36.9% for germline deleterious BRCA mutation in a United States study (N=314).76 The patients in this study were described as having an estrogen receptor/progesterone receptor low+ status (ie, 1–9% on immunohistochemistry) and to have HER2- breast cancer; other risk criteria used to select the study population were not clearly reported.
Figure 3 summarizes the prevalence of any type of BRCA mutation (BRCA1 or BRCA2) in the 19 largest (N>500) included studies. Generally, BRCA mutation prevalence was lowest in those populations that were not selected on the basis of family history and highest in those that were selected on the basis of at least 1 or more criterion, including family history of breast cancer, early onset breast cancer, or male breast cancer. In the 7 largest studies that also reported that mutations were germline, gBRCA mutation prevalence varied from 2.9% in a Sardinian study77 (unselected population) to 26.5% in a German study78 (selected for family history of breast cancer).
 |
Figure 3 Prevalence (%) in largest (N>500) studies. Notes: Horizontal axis has 3 levels: bottom – selection (family history/sex/ethnicity); middle – hormone receptor status (red = TNBC, dark green = mixed, light green = NR/unclear); top – country. BC stage not shown because NR/unclear for all but 2 studies (1 mixed and 1 invasive); *Bar for Son, 201237 is striped in order to distinguish it from bar for Kim, 2012: it is of mixed hormone receptor status.26 Pale blue = germline reported; red text = deleterious/pathogenic/clinically significant reported. High risk: based on fulfilment of at least 1 of a set of criteria, including family history, early onset, or male BC, which vary by study (See Table 1). Mixed: implies that the study included both those individuals with and without a family history of breast cancer, but the study did not report that probands were unselected. Abbreviations: BC, breast cancer; NR, not reported; TNBC, triple-negative breast cancer. |
Among those studies reporting on patients with TNBC, there was a clear trend for studies to report higher BRCA mutation prevalence levels. For example, in the unselected population, BRCA mutation prevalence was 11.2% in a study by Couch et al79 versus 2.9% in a mixed HR-status study by Palomba et al77.
In those studies that fully reported BRCA mutation prevalence according to the BRCA1 and BRCA2 probands, 26 out of 43 reported that mutations in BRCA1 were more common than in BRCA2.
gBRCA mutation prevalence in those unselected on any basis other than hormone receptor status or breast cancer stage
Table 2 summarizes the gBRCA mutation prevalence in 10 large (N>100) studies that included individuals who were unselected on the basis of family history of breast cancer, age, sex, or ethnicity.
 |
Table 2 Germline BRCA mutation prevalence in those unselected for family history, age, sex, or ethnicity |
gBRCA mutation prevalence was similar in 2 studies of populations26,77 with mixed breast cancer HR status or in which the HR status was not reported and the breast cancer stage was not reported or unclear. gBRCA mutation prevalence was 2.9% in a Sardinian study77 and 3.0% in a South Korean study.26
In populations with metastatic breast cancer, 2 studies reported similar gBRCA mutation prevalence data (2.7%80 and 4.3%81). Higher gBRCA mutation prevalence values were reported in the United States study by Tung et al,46 but these data were from a single academic center that only included patients with early breast cancer; this study reported that BRCA mutation prevalence for a subgroup of patients with HR+/HER2- breast cancer was 5.0%.
In 4 studies of TNBC patients that reported on gBRCA mutation prevalence, values ranged from 9.3% in an Australian study (N=439)82 to 15.4% in a United States study (N=207).42 Both of these studies included populations with both early and advanced stages of breast cancer.
BRCA mutation prevalence in advanced breast cancer
Table 3 reports data from 4 large (N>100) studies reporting on BRCA mutation prevalence in patients with metastatic breast cancer. Mutations in BRCA1 were less common than mutations in BRCA2 in 2 studies.80,81 However, in contrast, the prevalence of BRCA1 mutations was more common than BRCA2 in 1 study.45
 |
Table 3 BRCA mutation prevalence in advanced breast cancer |
BRCA mutation prevalence of any kind varied widely across the 4 studies in metastatic breast cancer. gBRCA mutation prevalence was reported to be as low as 2.7% in a French study (N=407)80 in which the population was unselected for family history of breast cancer and patients with breast cancer varied in their HR status. In a large German study (N=1462)81 in a population unselected by family history of breast cancer, the prevalence of gBRCA mutation was also comparatively low (4.3%), but it was not reported whether this included deleterious BRCA mutations. In contrast, gBRCA mutation prevalence was as high as 21.0% in a study (N=195) of patients with a family history of breast cancer and in which patients varied with respect to their HR status.45
Table 3 also shows the studies in locally aBC. Only 1 small study (N=13)44 in Israel reported the prevalence of mutation of any BRCA gene, in a population of Ashkenazi Jewish women with early onset breast cancer.
Discussion
To our knowledge, this is the first systematic review that utilized rigorous review methods11,12 to comprehensively report on the international prevalence of BRCA mutation (including gBRCA [BRCA1 and/or BRCA2 wherever specified] mutation) in breast cancer patients across a broad range of populations. Prevalence was also analyzed according to HER2 status, HR status, and stage of breast cancer (including locally advanced or metastatic).
In the 58 large studies (N>100), the prevalence of BRCA mutation of any kind between individual studies varied very widely from 0.6% to 36.9%. However, in contrast, the prevalence of gBRCA mutation appeared to vary little (≈3%) between studies in a general (unselected) population.26,77 gBRCA mutation prevalence appeared to be unaffected by metastatic breast cancer stage, ranging from 2.7%80 to 4.3%.81 Our results are consistent with a publication by Nelson et al,83 regarding a meta-analysis of 70 studies. None of these 70 studies was published after 2011 and as a result the studies were not included in our review; germline status was also not explicitly reported. The meta-analysis84 reported a BRCA mutation prevalence of 3% in women with breast cancer and 20% in high-risk families. This was consistent with our reported BRCA mutation prevalence results among large studies, where family history was associated with a BRCA mutation prevalence of more than 20% (range of 21.7%84 to 26.5%78). Consistency of our results can be further ascertained with a recent April 2019 publication by Kurian et al who reported germline BRCA1/2 mutation prevalence among United States breast cancer patients in the Georgia and California SEER registries (HR+/HER2-: 5.2% and TNBC: 15.4%); these numbers are largely aligned to what we have summarized in this systematic literature review.85
There did appear to be evidence of a selection effect in our review according to some risk characteristics of breast cancer. This included an increase in BRCA prevalence in those populations selected on the basis of high-risk criteria (based on the fulfillment of at least 1 of a set of criteria, including family history, early onset, or male breast cancer), when compared with those who were not selected on the basis of family history. In the 7 largest studies that also reported on prevalence of gBRCA mutation, gBRCA mutation prevalence varied from 2.9% to 26.5%, but it was difficult to determine any trends in the data as the populations varied widely with respect to their selection criteria. However, a trend was evident in the prevalence of BRCA mutation between those populations with versus those without TNBC; those with TNBC tended to have a higher prevalence of BRCA mutation in line with previous research. There was also a suggestion that in a small majority (26 of 44 studies), mutations in BRCA1 were more common than in BRCA2. However, this trend was reversed among patients with TNBC, in whom the majority of mutations were BRCA1. A published meta-analysis, by Tun et al,86 of prevalence of BRCA1 mutation in female patients with breast cancer, regardless of germline status, found that those with high-risk (including family history and early onset breast cancer) TNBC are much more likely to have BRCA1 mutation compared with those with a non-TNBC phenotype (relative risk 5.65 [95% confidence interval 4.15–7.69]), and that approximately 2 in 9 (≈22%) women with TNBC harbor BRCA1 mutation. Our review also found BRCA1 mutation to be more common than BRCA2 mutation in TNBC, although our estimates of BRCA1 mutation prevalence were mostly (11 of 14 studies) lower than the estimate by Tun et al, ie, 22%.86 There was no obvious explanation for this difference except that very few of the studies included in our review were included in the review by Tun et al86. Indeed, the only study in common between our review and that by Tun et al86 was the study by Bayraktar et al45. This was because all other studies included by Tun et al86 were outside of our scope: 8 of the 12 studies were published before 2012 and the other 3 were China-based population studies.86
The content of any systematic review is dependent on the quality of the included research. We focused on those studies whose specific aim was to investigate the prevalence of BRCA mutation, rather than studies that happen to report ad hoc data on prevalence or data that could be used to calculate prevalence. Nonetheless, some studies had a small sample size and/or poor reporting of study data and methods, which hampered our assessment. Wherever possible, we have provided information regarding the strength of the evidence and have also highlighted any general weaknesses or omissions in the data. In particular, studies often failed to report whether BRCA mutations were germline or somatic and which specific BRCA genes were under investigation (including fully reporting data for BRCA1 and BRCA2 mutations separately and whether a proband carried both BRCA1 and BRCA2 mutations). In addition, it was not clear whether the mutations were deleterious/pathogenic/clinically significant or not and which sequencing method was used (ie, direct or NGS). The HR status of patients (beyond that of TNBC) was similarly poorly described. Where this was not clearly reported by the authors of the primary studies, we labeled the study as unclear to avoid any misinterpretation. This was identified as a weakness in many of the included studies, and researchers should ensure that these details are clearly reported in future studies. In estimating BRCA mutation prevalence, it is also important for the purposes of collating and comparing data across studies that future studies clearly identify how they select their study populations and report on key baseline characteristics such as family history, ethnicity, and personal history of cancer (type, stage, and HR status). Consistency in reporting these variables will help to avoid the issues of heterogeneity raised in this review, including problems in summarizing the overall findings and identifying trends in the data. Studies should also follow the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) statement75 to improve reporting standards in epidemiological studies.
None of the studies included in our review reported prevalences in the main population of interest (gBRCA mutation in HR+/HER2- or TNBC locally advanced/metastatic disease unselected for family history, sex, age, and ethnicity), ie, those individuals who are potentially eligible for PARPi. This implies a challenge for healthcare providers and policy makers who need to estimate the size of the population eligible for PARPi. Future epidemiological studies need to target this specific population of interest to assist healthcare decision makers, policy makers, and payers quantify the population and make informed decisions.
Conclusions
To our knowledge this is the first systematic review to comprehensively report on the international prevalence of BRCA mutations in breast cancer patients across a broad range of populations. BRCA mutation prevalence varied widely within key clinical and demographic subgroups and across countries. Among TNBC populations, the percentage prevalence of gBRCA mutations ranged from 9.3% to 15.4%, and amongst patients with metastatic breast cancer, from 2.7% to 4.3%. Within larger studies the prevalence of BRCA mutations appeared higher for those studies that selected patients based on their family history and the presence of TNBC. However, the interpretation of the prevalence data was hampered by poor reporting on the nature of BRCA mutations (eg, germline versus somatic) and key baseline characteristics of the patients. Further large, well-reported, epidemiological studies of BRCA prevalence are warranted.
Abbreviation list
aBC, advanced breast cancer; BRCA1/2, breast cancer susceptibility genes 1 or 2; CRD, Centre for Reviews and Dissemination; FDA, Food and Drug Administration; gBRCA, germline BRCA; HER2-, human epidermal growth factor receptor 2–negative; HR+, hormone receptor–positive; NGS, next-generation sequencing; PARPi, poly ADP-ribose polymerase inhibitors; TNBC, triple-negative breast cancer.
Acknowledgments
Editorial support was provided by Edwin Thrower, PhD, Chantal Cadwell, PhD, Paula Stuckart, and Dena McWain at Ashfield Healthcare Communications, Middletown, CT, USA, and was funded by Pfizer, Inc. Pfizer Inc. funded the project, and contributed to the analysis and interpretation of the data and writing the report. This work was presented previously as an abstract and poster at the European Society for Medical Oncology (ESMO) 2018 Congress, October 19-23, 2018, Munich, Germany.
Author contributions
All authors made substantial contributions to the conception and design, data acquisition and data analysis and interpretation; drafted the article; provided final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
NA, SR, CF, and JR are employees of Kleijnen Systematic Reviews Ltd., who were paid consultants to Pfizer in connection with the development of this manuscript. RGWQ is an employee of and owns stocks from Pfizer Inc. The authors report no other conflicts of interest in this work.
References
1. Latest global cancer data: Cancer burden rises to 18.1 million new cases and 9.6 million cancer deaths in 2018 [press release]. Geneva, Switzerland: International Agency for Research on Cancer; September 12, 2018. https://www.who.int/cancer/PRGlobocanFinal.pdf. Accessed June 10, 2019.
2. Engel C, Fischer C. Breast cancer risks and risk prediction models. Breast Care (Basel). 2015;10(1):7–12. doi:10.1159/000376600
3. Greenup R, Buchanan A, Lorizio W, et al. Prevalence of BRCA mutations among women with triple-negative breast cancer (TNBC) in a genetic counseling cohort. Ann Surg Oncol. 2013;20(10):3254–3258. doi:10.1245/s10434-013-3205-1
4. National Comprehensive Cancer Network. Genetic/Familial High-Risk Assessment: Breast and Ovarian. Version 1.2018 - October 3, 2017 [Internet]. Fort Washington, PA: NCCN; 2017.
5. Gudmundsdottir K, Ashworth A. The roles of BRCA1 and BRCA2 and associated proteins in the maintenance of genomic stability. Oncogene. 2006;25(43):5864–5874. doi:10.1038/sj.onc.1209874
6. Livraghi L, Garber JE. PARP inhibitors in the management of breast cancer: current data and future prospects. BMC Med. 2015;13:188. doi:10.1186/s12916-015-0425-1
7. Paluch-Shimon S, Friedman E, Berger R, et al. Neo-adjuvant doxorubicin and cyclophosphamide followed by paclitaxel in triple-negative breast cancer among BRCA1 mutation carriers and non-carriers. Breast Cancer Res Treat. 2016;157(1):157–165. doi:10.1007/s10549-016-3800-5
8. National Institute for Health and Care Excellence. Familial breast cancer: classification, care and managing breast cancer and related risks in people with a family history of breast cancer. NICE Clinical Guideline CG164 [Internet]. Manchester: NICE; 2013.
9. Cancer Australia. Recommendations for the Management of Early Breast Cancer in Women with an Identified BRCA1 or BRCA2 Gene Mutation or at High Risk of a Gene Mutation [Internet]. Surry Hills: Australian Government, Cancer Australia; 2014.
10. De Felice F, Marchetti C, Musella A, et al. Bilateral risk-reduction mastectomy in BRCA1 and BRCA2 mutation carriers: a meta-analysis. Ann Surg Oncol. 2015;22(9):2876–2880. doi:10.1245/s10434-015-4532-1
11. Higgins JPT, Green S, eds. Cochrane Handbook for Systematic Reviews of Interventions [Internet]. Version 5.1.0. London, UK: The Cochrane Collaboration; 2011. Updated March 2011.
12. Centre for Reviews and Dissemination. Systematic Reviews: CRD’s Guidance for Undertaking Reviews in Health Care [Internet]. York: University of York; 2009.
13. Munn Z, Moola S, Lisy K, Riitano D, Tufanaru C. Methodological guidance for systematic reviews of observational epidemiological studies reporting prevalence and incidence data. Int J Evid Based Healthc. 2015;13(3):147–153. doi:10.1097/XEB.0000000000000054
14. Pal T, Bonner D, Cragun D, et al. A high frequency of BRCA mutations in young black women with breast cancer residing in Florida. Cancer. 2015;121(23):4173–4180. doi:10.1002/cncr.29645
15. Park B, Sohn JY, Yoon KA, et al. Characteristics of BRCA1/2 mutations carriers including large genomic rearrangements in high risk breast cancer patients. Breast Cancer Res Treat. 2017;163(1):139–150. doi:10.1007/s10549-017-4142-7
16. Tung N, Battelli C, Allen B, et al. Frequency of mutations in individuals with breast cancer referred for BRCA1 and BRCA2 testing using next-generation sequencing with a 25-gene panel. Cancer. 2015;121(1):25–33. doi:10.1002/cncr.29010
17. Asleh-Aburaya K, Fried G. Clinical and molecular characteristics of triple-negative breast cancer patients in Northern Israel: single center experience. Springerplus. 2015;4:132. doi:10.1186/s40064-015-0900-3
18. de Juan Jimenez I, Esteban Cardenosa E, Palanca Suela S, et al. Low prevalence of BRCA1 and BRCA2 mutations in the sporadic breast cancer of Spanish population. Fam Cancer. 2012;11(1):49–56. doi:10.1007/s10689-011-9481-7
19. Duffy J, Greening S, Warwick L, Tucker K, Creighton B. An Australian perspective of genetic testing in breast cancers diagnosed under age 31. Curr Oncol. 2012;19(2):e106.
20. Eccles DM, Li N, Handwerker R, et al. Genetic testing in a cohort of young patients with HER2-amplified breast cancer. Ann Oncol. 2016;27(3):467–473. doi:10.1093/annonc/mdv592
21. Ellsworth RE, Lovejoy LA, Shriver C. Assessment of the hereditary component in 94 cancer predisposition genes to triple negative breast cancer.
22. Emborgo T, Muse KI, Bednar E, et al. Universal BRCA testing and family outreach for women with triple negative breast cancer. Cancer Res. 2016;76(4):
23. Garber JE, Tung NM, Elkin EP, et al. Predisposing germline mutations in a clinic based breast cancer (BC) population. Cancer Res. 2016;76(4):
24. Gonzalez-Rivera M, Lobo M, Lopez-Tarruella S, et al. Frequency of germline DNA genetic findings in an unselected prospective cohort of triple-negative breast cancer patients participating in a platinum-based neoadjuvant chemotherapy trial. [Erratum appears in Breast Cancer Res Treat. 2017;165(2):471. PMID: 28721639. doi:10.1007/s10549-017-4396-0]. Breast Cancer Res Treat. 2016;156(3):507–515. doi:10.1007/s10549-016-3792-1
25. Guerra YC, Sand S, Correa MC, et al. Genetic, clinical and pathological characteristics of BRCA-associated breast cancer (BC) in Hispanic patients in the United States (US) and Latin America (LatAm). J Clin Oncol. 2017;35(15 suppl):1539. doi:10.1200/JCO.2017.35.15_suppl.1539
26. Kim H, Cho DY, Choi DH, et al. Characteristics and spectrum of BRCA1 and BRCA2 mutations in 3,922 Korean patients with breast and ovarian cancer. Breast Cancer Res Treat. 2012;134(3):1315–1326. doi:10.1007/s10549-012-2159-5
27. Kitagawa H, Yagata H, Yoshida A, Nakano E, Yoshino M, Yamauchi H. The prevalence of BRCA mutations among patients with triple-negative breast cancer. Curr Oncol. 2014;21(2):e388.
28. Lee AS, Kang EY, Baek H, Chae S, Kim EK, Kim SW. Evaluation of BRCA1/2 mutation prevalence in Korean women with triple-negative breast cancer. Ann Oncol. 2015;26(Suppl 9):ix16. doi:10.1093/annonc/mdv383
29. Musolino A, Bella MA, Michiara M, et al. BRCA status, molecular profile and clinical variables in primary bilateral breast cancers: a population-based cancer registry study. Ann Oncol. 2012;23:ix175–ix176.
30. Pal T, Bonner D, Cragun D, et al. Proportion of BRCA mutation frequency in young black women with breast cancer. J Clin Oncol. 2014;15(Suppl):1506. doi:10.1200/jco.2014.32.15_suppl.1506
31. Polonikov A, Aristova I, Leitanthem W, et al. Prevalence of BRCA1 and BRCA2 mutations in breast cancer patients in Russia. Ann Acad Med Singapore. 2015;44(10 Suppl 1):S324.
32. Robertson L, Hanson H, Seal S, et al. BRCA1 testing should be offered to individuals with triple-negative breast cancer diagnosed below 50 years. Br J Cancer. 2012;106(6):1234–1238. doi:10.1038/bjc.2012.31
33. Rummel S, Varner E, Shriver CD, Ellsworth RE. Evaluation of BRCA1 mutations in an unselected patient population with triple-negative breast cancer. Breast Cancer Res Treat. 2013;137(1):119–125. doi:10.1007/s10549-012-2348-2
34. Rummel SK, Lovejoy L, Shriver CD, Ellsworth RE. Contribution of germline mutations in cancer predisposition genes to tumor etiology in young women diagnosed with invasive breast cancer. Breast Cancer Res Treat. 2017;164(3):593–601. doi:10.1007/s10549-017-4291-8
35. Sanford RA, Song J, Gutierrez-Barrera AM, et al. Incidence of germline BRCA mutation in patients with ER-low positive/PR negative/HER-2 neu negative tumors. J Clin Oncol. 2014;26(Suppl):2. doi:10.1200/jco.2014.32.26_suppl.2
36. Skandan SP. 5 year overall survival of triple negative breast cancer: a single institution experience. J Clin Oncol. 2016;34(15 Suppl):e12580. doi:10.1200/JCO.2016.34.15_suppl.e12580
37. Son BH, Ahn SH, Kim SW, et al. Prevalence of BRCA1 and BRCA2 mutations in non-familial breast cancer patients with high risks in Korea: the Korean Hereditary Breast Cancer (KOHBRA) study. Breast Cancer Res Treat. 2012;133(3):1143–1152. doi:10.1007/s10549-012-2001-0
38. Vanstone M, Chow W, Lester L, Ainsworth P, Nisker J, Brackstone M. Recognizing BRCA gene mutation risk subsequent to breast cancer diagnosis in southwestern Ontario. Can Fam Physician. 2012;58(5):e258–e266.
39. Vidula N, Isakoff SJ, Niemierko A, et al. Somatic BRCA mutation detection by circulating tumor DNA analysis in patients with metastatic breast cancer: incidence and association with tumor genotyping results, germline BRCA mutation status, and clinical outcomes.
40. Yoon KA, Park B, Lee BI, Yang MJ, Kong SY, Lee ES. Clinically significant unclassified variants in BRCA1 and BRCA2 genes among Korean breast cancer patients. Cancer Res Treat. 2017;49(3):627–634. doi:10.4143/crt.2016.292
41. Kang E, Seong MW, Park SK, et al. The prevalence and spectrum of BRCA1 and BRCA2 mutations in Korean population: recent update of the Korean Hereditary Breast Cancer (KOHBRA) study. Breast Cancer Res Treat. 2015;151(1):157–168. doi:10.1007/s10549-015-3377-4
42. Sharma P, Klemp JR, Kimler BF, et al. Germline BRCA mutation evaluation in a prospective triple-negative breast cancer registry: implications for hereditary breast and/or ovarian cancer syndrome testing. Breast Cancer Res Treat. 2014;145(3):707–714. doi:10.1007/s10549-014-2980-0
43. Keung YK, Hu A, Yeung A, Chan A, Hu E. Higher prevalence of BRCA2 mutations among Chinese breast cancer patients in a community oncology clinic. J Clin Oncol. 2012;15(Suppl):12017.
44. Dagan E, Gershoni-Baruch R, Kurolap A, Fried G. Early onset breast cancer in Ashkenazi women carriers of founder BRCA1/2 mutations: beyond 10 years of follow-up. Eur J Cancer Care. 2017;26:6. doi:10.1111/ecc.12594
45. Bayraktar S, Gutierrez-Barrera AM, Lin H, et al. Outcome of metastatic breast cancer in selected women with or without deleterious BRCA mutations. Clin Exp Metastasis. 2013;30(5):631–642. doi:10.1007/s10585-013-9567-8
46. Tung N, Lin NU, Kidd J, et al. Frequency of germline mutations in 25 cancer susceptibility genes in a sequential series of patients with breast cancer. J Clin Oncol. 2016;34(13):1460–1468. doi:10.1200/JCO.2015.65.0747
47. Cherdyntseva NV, Pisareva LF, Ivanova AA, et al. [Ethnic aspects of hereditary breast cancer in the region of Siberia]. Vestn Ross Akad Med Nauk. 2014;(11–12):72–79. doi:10.15690/vramn.v69i11-12.1186
48. Ellsworth RE, Rummel S, Varner E, Shriver CD. Evaluation of BRCA1 mutations in patients with triple negative breast cancer. Cancer Res. 2012;72(8 Suppl):68.
49. Ghadirian P, Robidoux A, Nassif E, et al. Screening for BRCA1 and BRCA2 mutations among French-Canadian breast cancer cases attending an outpatient clinic in Montreal. Clin Genet. 2014;85(1):31–35. doi:10.1111/cge.12174
50. Stadler ZK, Salo-Mullen E, Patil SM, et al. Prevalence of BRCA1 and BRCA2 mutations in Ashkenazi Jewish families with breast and pancreatic cancer. Cancer. 2012;118(2):493–499. doi:10.1002/cncr.26191
51. Weitzel JN, Clague J, Martir-Negron A, et al. Prevalence and type of BRCA mutations in Hispanics undergoing genetic cancer risk assessment in the southwestern United States: a report from the clinical cancer genetics community research network. [Erratum appears in J Clin Oncol. 2013;31(13):1702]. J Clin Oncol. 2013;31(2):210–216. doi:10.1200/JCO.2011.41.0027
52. Noh JM, Han BK, Choi DH, et al. Association between BRCA mutation status, pathological findings, and magnetic resonance imaging features in patients with breast cancer at risk for the mutation. J Breast Cancer. 2013;16(3):308–314. doi:10.4048/jbc.2013.16.3.308
53. Pal T, Bonner D, Kim J, et al. Early onset breast cancer in a registry-based sample of African-American women: BRCA mutation prevalence, and other personal and system-level clinical characteristics. Breast J. 2013;19(2):189–192. doi:10.1111/tbj.12083
54. Pal T, Bonner D, Cragun D, et al. BRCA sequencing and large rearrangement testing in young Black women with breast cancer. J Community Genet. 2014;5(2):157–165. doi:10.1007/s12687-013-0166-9
55. Sohn JY, Park B, Yoon KA, et al. Prevalence of BRCA1 and BRCA2 small mutation and large genomic rearrangements in breast cancer patients visiting a genetic counseling clinic. Cancer Res. 2016;76(14):3474. doi:10.1158/0008-5472.CAN-16-0584
56. Rahman N, Turnbull A, George A, Strydom A, Kemp Z. Using cancer status is better, simpler and more cost-effective than family history in determining breast cancer genetic testing eligibility.
57. Petersen L, Aubry S, Burgess K, Rao R, Kopkash K, Madrigrano A. Are triple negative breast cancer patients screened for BRCA mutations according to NCCN guidelines? Ann Surg Oncol. 2016;23(1SUPPL. 1):S67–S68.
58. Pal T, Bonner D, Kim J, et al. BRCA mutations and factors associated with surgery type in a registry-based sample of young black women with invasive breast cancer. Curr Oncol. 2012;19(2):e99.
59. Biskupiak JE, Telford C, Yoo M, et al. Evaluation of women with BRCA mutations and breast cancer tested at an NCI designated comprehensive cancer center: a cost of illness estimation. Cancer Res. 2017;77(4 Suppl):P3-12-03. doi:10.1158/1538-7445.SABCS16-P3-12-03.
60. Eccles DM, Copson ER, Maishman T, et al. Does BRCA status affect outcome in young breast cancer patients? Results from the prospective study of outcomes in sporadic and hereditary breast cancer (POSH). Cancer Res. 2017;77(4 Suppl):S2-03. doi:10.1158-7445.SABCS16-S2-03.
61. Susswein LR, Marshall ML, Nusbaum R, et al. Pathogenic and likely pathogenic variant prevalence among the first 10,000 patients referred for next-generation cancer panel testing. Genet Med. 2016;18(8):823–832. doi:10.1038/gim.2015.166
62. Buys SS, Sandbach JF, Gammon A, et al. A study of over 35,000 women with breast cancer tested with a 25-gene panel of hereditary cancer genes. Cancer. 2017;123(10):1721–1730. doi:10.1002/cncr.30498
63. Rhiem K, Engel C, Engel J, et al. BRCA1/2 mutation prevalence in triple-negative breast cancer patients without family history of breast and ovarian cancer. J Clin Oncol. 2016;34(15 Suppl):1090. doi:10.1200/JCO.2016.34.15_suppl.1090
64. Welinsky S, Becker AE, Aronson A, Hernandez YG, Soper E, Lucas AL. No increase in prevalence of pancreatic cysts in BRCA1 or BRCA2 mutation carriers with breast cancer. Gastroenterology. 2016;150(4 Suppl 1):S233. doi:10.1016/S0016-5085(16)30853-8
65. Hartman AR, Kaldate RR, Sailer LM, et al. Prevalence of BRCA mutations in an unselected population of triple-negative breast cancer. Cancer. 2012;118(11):2787–2795. doi:10.1002/cncr.26576
66. Beck A, Yuan H, Imperiale-Hagerman P, et al. Adhering to the guidelines: rates of BRCA mutation using NCCN genetic testing criteria. Ann Surg Oncol. 2017;24(2):257048.
67. Seong MW, Kim KH, Chung IY, et al. A multi-institutional study on the association between BRCA1/BRCA2 mutational status and triple-negative breast cancer in familial breast cancer patients. Breast Cancer Res Treat. 2014;146(1):63–69. doi:10.1007/s10549-014-3006-7
68. Lolas Hamameh S, Renbaum P, Kamal L, et al. Genomic analysis of inherited breast cancer among Palestinian women: genetic heterogeneity and a founder mutation in TP53. Int J Cancer. 2017;141(4):750–756. doi:10.1002/ijc.30771
69. Andres R, Pajares I, Balmana J, et al. Association of BRCA1 germline mutations in young onset triple-negative breast cancer (TNBC). Clin Transl Oncol. 2014;16(3):280–284. doi:10.1007/s12094-013-1070-9
70. Ross JL, Woodson AH, Gutierrez Barrera AM, Litton JK, Arun B. Multi-gene panel testing results in patients with multiple breast cancer primaries.
71. Jung J, Kang E, Gwak JM, et al. Association between basal-like phenotype and BRCA1/2 germline mutations in Korean breast cancer patients. Curr Oncol. 2016;23(5):298–303. doi:10.3747/co.23.3054
72. Tunon De Lara C, Bonnet F, Debled M, et al. Rapid germline BRCA screening for locally advanced breast cancer changes surgical procedure after neoadjuvant chemotherapy. Cancer Res. 2017;77(4Suppl):
73. Zugazagoitia J, Perez-Segura P, Manzano A, et al. Limited family structure and triple-negative breast cancer (TNBC) subtype as predictors of BRCA mutations in a genetic counseling cohort of early-onset sporadic breast cancers. Breast Cancer Res Treat. 2014;148(2):415–421. doi:10.1007/s10549-014-3167-4
74. De La Cruz J, Andre F, Harrell RK, et al. Tissue-based predictors of germ-line BRCA1 mutations: implications for triaging of genetic testing. Hum Pathol. 2012;43(11):1932–1939. doi:10.1016/j.humpath.2012.02.002
75. von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370(9596):1453–1457. doi:10.1016/S0140-6736(07)61602-X
76. Sanford RA, Song J, Gutierrez-Barrera AM, et al. High incidence of germline BRCA mutation in patients with ER low-positive/PR low-positive/HER-2 neu negative tumors. Cancer. 2015;121(19):3422–3427. doi:10.1002/cncr.29572
77. Palomba G, Budroni M, Olmeo N, et al. Triple-negative breast cancer frequency and type of BRCA mutation: clues from Sardinia. Oncol Lett. 2014;7(4):948–952. doi:10.3892/ol.2014.1834
78. Kast K, Rhiem K, Wappenschmidt B, et al. Prevalence of BRCA1/2 germline mutations in 21 401 families with breast and ovarian cancer. J Med Genet. 2016;53(7):465–471. doi:10.1136/jmedgenet-2015-103672
79. Couch FJ, Hart SN, Sharma P, et al. Inherited mutations in 17 breast cancer susceptibility genes among a large triple-negative breast cancer cohort unselected for family history of breast cancer. J Clin Oncol. 2015;33(4):304–311. doi:10.1200/JCO.2014.57.1414
80. Meynard G, Villanueva C, Thiery-Vuillemin A, et al. 284P Real-life study of BRCA genetic screening in metastatic breast cancer. Ann Oncol. 2017;28(suppl_5):
81. Fasching PA, Hu C, Hart SN, et al. Cancer predisposition genes in metastatic breast cancer – association with metastatic pattern, prognosis, patient and tumor characteristics.
82. Wong-Brown MW, Meldrum CJ, Carpenter JE, et al. Prevalence of BRCA1 and BRCA2 germline mutations in patients with triple-negative breast cancer. Breast Cancer Res Treat. 2015;150(1):71–80. doi:10.1007/s10549-015-3293-7
83. Nelson HD, Fu R, Goddard K, et al. Risk Assessment, Genetic Counseling, and Genetic Testing for BRCA-related Cancer: Systematic Review to Update the U.S. Preventive Services Task Force Recommendation: Evidence Synthesis No. 101. AHRQ Publication No. 12-05164-EF-1. Rockville, MD: Agency for Healthcare Research and Quality; 2013. Available from https://www.ncbi.nlm.nih.gov/books/NBK179201/pdf/Bookshelf_NBK179201.pdf
84. Han SA, Kim SW, Kang E, et al. The prevalence of BRCA mutations among familial breast cancer patients in Korea: results of the Korean Hereditary Breast Cancer study. Fam Cancer. 2013;12(1):75–81. doi:10.1007/s10689-012-9578-7
85. Kurian AW, Ward KC, Howlader N, et al. Genetic testing and results in a population-based cohort of breast cancer patients and ovarian cancer patients. J Clin Oncol. 2019;37(15):1305–1315. doi:10.1200/JCO.18.01854
86. Tun NM, Villani G, Ong K, Yoe L, Bo ZM. Risk of having BRCA1 mutation in high-risk women with triple-negative breast cancer: a meta-analysis. Clin Genet. 2014;85(1):43–48. doi:10.1111/cge.12270
87. Loi M, Olmetto E, Desideri I, et al. 187 - BRCA1/2 mutation in breast cancer: biological aspects, patterns of care and impact on outcome in a monoinstitutional cohort. Eur J Cancer. 2017;72(Supplement 1):S19–S19. doi:10.1016/S0959-8049(17)30141-7
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
