Back to Journals » Diabetes, Metabolic Syndrome and Obesity » Volume 14
A Stronger Association of Epicardial Fat Volume with Non-Valvular Atrial Fibrillation Than Measures of General Obesity in Chinese Patients Undergoing Computed Tomography Coronary Angiography
Authors Zhu J, Yang Z, Chen X, Liu J, Zheng L, Zhang L, Pi J, Li G, Zhuang T, Liu C, Wang Y, Liu L, Fan L, Chan P , Tomlinson B , Li Y, Liu Z, Zhang Y
Received 26 July 2020
Accepted for publication 27 January 2021
Published 18 March 2021 Volume 2021:14 Pages 1223—1232
DOI https://doi.org/10.2147/DMSO.S274047
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Juei-Tang Cheng
Jingqi Zhu,1,2,* Zhangwei Yang,2,* Xiaoli Chen,3,* Jie Liu,3 Liang Zheng,3 Lin Zhang,3 Jingjiang Pi,4 Gang Li,2 Tao Zhuang,3 Chang Liu,3 Yinglu Wang,3 Liping Liu,3 Lieying Fan,5 Paul Chan,6 Brian Tomlinson,7 Ying Li,4 Zhongmin Liu,3 Yuzhen Zhang3
1Department of Radiology, Shanghai Tenth People’s Hospital, Tongji University School of Medicine, Shanghai, People’s Republic of China; 2Department of Radiology, Shanghai East Hospital, Tongji University School of Medicine, Shanghai, People’s Republic of China; 3Key Laboratory of Arrhythmias of the Ministry of Education of China, Research Center for Translational Medicine, Shanghai East Hospital, Tongji University School of Medicine, Shanghai, People’s Republic of China; 4Department of Cardiology, Shanghai East Hospital, Tongji University School of Medicine, Shanghai, People’s Republic of China; 5Department of Clinical Laboratory, Shanghai East Hospital, Tongji University School of Medicine, Shanghai, People’s Republic of China; 6Division of Cardiology, Department of Internal Medicine, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan; 7Faculty of Medicine, Macau University of Science and Technology, Macau, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Yuzhen Zhang; Zhongmin Liu
Key Laboratory of Arrhythmias of the Ministry of Education of China, Research Center for Translational Medicine, Shanghai East Hospital, Tongji University School of Medicine, 150 Jimo Road, Pudong New District, Shanghai, 200120, People’s Republic of China
Tel +86-21-61569673
Email [email protected]; [email protected]
Objective: An association of atrial fibrillation (AF) with epicardial fat volume (EFV) varied in different ethnic groups. We evaluated the AF-related risk factors and its association with pericardial fat in Chinese patients.
Methods: Patients referred for coronary computed tomography angiography (CCTA) in Shanghai East Hospital during 2012 to 2014 (n=2042, 43.8% women, mean age 65.0 years) had AF and cardiovascular risk assessment. Pericardial fat depots were measured from CT and the association of EFV with non-valvular AF risk factors was evaluated by multivariate logistic regression models.
Results: AF was present in 8.5% of patients with 11.6% of AF patients having rheumatic heart disease (RHD) and 8.7% having other valvular diseases. With increasing age, the proportion of RHD-related AF decreased and the risk factors for non-valvular AF increased. There was a significantly higher proportion of risk factors for non-valvular AF in men than in women (p=0.008), but RHD-related AF was more prevalent in women than men (p=0.013). The patients with non-valvular AF had significantly higher BMI and EFV with more pronounced elevation of EFV (p< 0.001). Multivariate logistic regression showed a significant association of EFV with AF after adjustment for BMI and clinical risk factors, and the highest EFV quartile was associated with AF independent of left atrial size and obstructive coronary artery disease.
Conclusion: The association of EFV with non-valvular AF in Chinese patients was independent of generalized adiposity and clinical risk factors especially in highest EFV quartile. These findings support the growing appreciation of the association of EFV with AF.
Keywords: atrial fibrillation, epicardial fat, pericardial fat, obesity, body mass index, computed tomography, echocardiography
Introduction
Atrial fibrillation (AF) is the most common cardiac arrhythmia and is strongly associated with stroke and heart failure, which significantly decreases the quality of life and causes a heavy burden on public health care with increasing morbidity and mortality.1,2 There is growing evidence that numerous cardiovascular risk factors are associated with the incidence of AF. Obesity is a major global public health issue and a large proportion of patients with AF in cohort studies tend to be overweight or obese. Obesity is now established as an independent risk factor for the incidence and progression of AF.1
Various studies, including the Framingham Heart Study and a meta-analysis, have indicated that an increase in body mass index (BMI) parallels a marked increase in AF risk.4,5 In a cohort of the Danish Diet, Cancer and Health Study with 47,589 participants prospectively followed up for a mean of 5.7 years, BMI independently correlated with increased AF risk regardless of gender.6 In a recently published cohort of 67,238 patients derived from a database of healthcare claims in the US, obesity was associated with new-onset AF independent of age, diabetes, hypertension and gender.7
Given that BMI is remarkably heterogeneous in its determinants and that adipose tissue distribution appears to be a key factor in determining cardiovascular risk, it is no surprise that the role of distinct adipose tissue depots is of intense interest.8 During recent years, more sophisticated imaging modalities such as CT and MRI are being used increasingly for cardiac imaging and the epicardial adipose tissue (EAT) directly overlying the myocardium can be directly imaged and quantified. Several clinical studies demonstrated associations of the epicardial fat volume (EFV) with the onset, severity and recurrence of AF and the outcomes after AF ablation.9,10
EAT is located between the myocardium and visceral pericardium covering 80% of the heart’s surface and amounting up to 20% of the heart’s weight, predominantly overlying the coronary arteries, atrioventricular and interventricular grooves, but also spanning the atria and ventricles. Paracardial fat refers to the layer of fat external to the parietal pericardium and pericardial fat is the combination of both EAT and paracardial fat.11,12 Far from being a mere storage tissue, pericardial fat is characterized by a complex secretome. Imaging studies have indicated that EFV correlates with increased AF risk in Japanese and the patients of USA,12–14 and a meta-analysis confirming this association between EFV and AF has shown an overall odds ratio of 2.61 for AF with each standard deviation increase in EFV with an even higher odds ratio of 5.43 for persistent AF.15
Ethnic differences in the association were reported in the multi-ethnic study of Atherosclerosis and Jackson Heart Study involving 40% African American, 32% white, 18% Hispanic, and 10% Chinese American.16 The greater pericardial fat volume was associated with higher AF risk in Hispanics, but not overall. However, very few studies have examined the pericardial fat deposition and AF incidence in Japanese or Asian populations12,17,18 and no study was performed in Chinese population. In the current study, we retrospectively collected the data from patients admitted to the Shanghai East Hospital having coronary CT angiography (CCTA) and echocardiography during 2012–2013 and prospectively recruited the outpatients who visited the hospital during 2013–2014 for CCTA to assess the association of pericardial fat with the presence of AF.
Methods
Data Collection
The study was conducted in accordance with the Declaration of Helsinki and the Institutional Review Board of Shanghai East Hospital affiliated to Tongji Medical School approved the study protocol. Written informed consent was obtained from each participant before any sample or data collection in the prospective study.
The participants evaluated in the study included patients admitted to Shanghai East Hospital during January 2012 to December 2013 undergoing CCTA for clinical reasons, whose data was analyzed retrospectively, and outpatients who visited Shanghai East Hospital during March 2013 to December 2014 for CCTA and echocardiography examination who were recruited prospectively. The patients were referred for CCTA because of chest pain, or for routine check-up, or cardiac assessment prior to surgery. The medical history was recorded and anthropometric measurements were made by trained doctors and nurses. Blood samples were taken after at least 10 hours of overnight fasting for laboratory parameters which were measured in the Blood Laboratory of Tongji Medical School affiliated Shanghai East Hospital.19 NT-proBNP was only measured in patients with medical history or clinical features of heart failure. The non-contrast CT (NCT) scan and the CCTA scan were used for the measurement of pericardial fat depots, coronary artery calcium (CAC) scores and coronary artery atherosclerotic plaques by trained specialists of Shanghai East Hospital.
Imaging Acquisition
Non-Contrast-Enhanced CT for Assessment of Pericardial Fat Depots and Coronary Artery Calcium Scores
All participants underwent chest NCT scan for assessment of body ectopic fat depots in the supine position using a dual-source CT scanner (Somatom Definition Flash, Siemens Healthcare, Forchheim, Germany). The average total CT scan dose was 5.415 mSv, with 0.292 mSv for the chest NCT scan, and 5.123 mSv for CCTA. Before the scan, all participants with average heart rates >70 beats/minute and no contraindication to the use of β-blockers were received 25 mg metoprolol orally, and the participants with heart rates >70 beats/minute and having contraindication or insufficient response to metoprolol were excluded. Only the participants with good image quality were included for further image evaluation.
For the thoracic image evaluation, all scans were performed during a single breath hold with an electrocardiogram (ECG)-triggering system with the acquisition window at 30–70% of the R-R interval. The scan range extended from the level of the carina to 10 mm below the diaphragm for the general patients. However, for patients after coronary artery bypass graft (CABG) surgery, the scan range extended from 10 mm above the aortic arch to 10 mm below the diaphragm. The main parameters for the thoracic scan were as follows: 64×0.625-mm collimation, 120-kV tube voltage, 320-mAs tube current, 330-ms gantry rotation time, 3-mm slice thickness, 1.5-mm slice increment, 250-mm field of view, 512×512 matrix, and a pitch of 0.2–0.5.
Coronary CT Angiography (CCTA) for Assessment of Coronary Artery Atherosclerotic Plaques
After the NCT scan, the CCTA scan was performed with an ECG-gating system to enable retrospective registration of image reconstruction in the appropriate cardiac phase (at 30–70% of the R-R interval) during a single breath hold. The main parameters for the CCTA scan were as follows: 64×0.625-mm collimation, 120-kV tube voltage, 320-mAs tube current, 330-ms gantry rotation time, 0.75-mm slice thickness, 0.5-mm slice increment, 250-mm field of view, 512×512 matrix, and a pitch of 0.2–0.5. For the contrast-enhanced scan, 70–90 mL of a nonionic contrast agent (Omnipaque, 350 mgI/mL; GE Healthcare, Princeton, NJ, USA) was injected into an antecubital vein at a flow rate of 4–5 mL/s using a power injector, followed by 30 mL of saline. A bolus tracking technique was used to determine the delay time. The actual scan was started when the region of interest (ROI) within the ascending aorta reached a threshold of 120 Hounsfield units (HU). A medium-smooth convolution kernel (B26f) was chosen for the image reconstruction.
Imaging Analysis
Pericardial Fat Measurements
The body ectopic fat depots were analyzed separately by 2 experienced readers (J.Z., Z.Y.) and the average values were used for the analysis. EFV was defined as the visceral intrapericardial fat contiguous with the myocardial surface, paracardial fat volume (PaCFV) as the fat deposits in the mediastinum outside the parietal pericardium. PaCFV and EFV were measured by calculating the sum of the corresponding volumes measured from the right pulmonary artery to the diaphragm, and pericardial fat volume (PeCFV), the sum of PaCFV and EFV. Periaortic fat volume (PAFV) was defined as the adipose tissue around the thoracic aorta, and the anatomic borders were: 1) superiorly, the right pulmonary artery; 2) inferiorly, the diaphragm; 3) anteriorly, the esophagus; 4) posteriorly, bilateral costovertebral joints.
The body fat, as measured by CT, was defined as any pixel with a density between −195 and −45 HU within the ROI.20 A semiautomatic technique for fat quantification was performed on the NCT images with the use of software (Volume, Siemens Healthcare).
Coronary Artery Calcium Score Measurement and CCTA Analysis
Coronary artery calcium (CAC) scores and CCTA datasets were analyzed separately by 2 experienced readers (J.Z., Z.Y.) in consensus using the same 15-segment American Heart Association model21 and average values for further analysis. Transverse sections, multiplanar reformations, curved multiplanar reformations, maximum intensity projection and volume rendering were used for imaging observation with the aid of cardiac softwares (CaScore, Siemens Healthcare for evaluation of CAC scores, and Circulation, Siemens Healthcare for evaluation coronary artery plaques). CAC scores were quantified by the calcifications in the coronary arteries of NCT images with a detection threshold of 130 HU. Each plaque by CCTA analysis was classified as follows: 1) non-calcified plaque (<130 HU); 2) calcified plaque (≥130 HU); or 3) mixed plaque (inclusive of both non-calcified and calcified plaque components).22
Study-Outcome Definitions
BMI was defined as weight in kilograms divided by height in meters squared. Participants were classified as being generally overweight (BMI ≥ 24 and <28 kg/m2) or obese (BMI ≥ 28 kg/m2) according to the Asian-specific BMI cut-points of the Health Standard of the People’s Republic of China National Health and Family Planning Commission.23 Definite hypertension was defined as an average of two measurements of systolic blood pressure (SBP) ≥140 mmHg or diastolic blood pressure (DBP) ≥90 mmHg, or normal blood pressure with concomitant use of antihypertensive medications. Definite diabetes was defined as fasting plasma glucose (FG) ≥7.0 mmol/l or normal FG with concomitant use of insulin or oral hypoglycaemic agents. Obstructive coronary artery diseases (CAD) was defined as coronary artery and branches having calcified, non-calcified and mixed plaques with plaques ≥70% stenosis of one coronary artery, or ≥50% stenosis of the left main coronary artery or 3-vessels by CCTA.24 AF was identified from discharge diagnosis, or ECGs including 24hr Holter ECG monitoring, and rheumatic heart disease (RHD) and other valvular diseases were identified by medical history and echocardiography.
Statistical Analysis
Descriptive statistics were calculated for all variables and significant differences in continuous variables were determined by ANOVA, and categorical percentile values were compared by Chi-squared test (χ2-test). The patients with RHD, other valvular diseases and systolic heart failure with ejection fraction (EF) well below the normal value (EF < 35%) or with NT-proBNP >900 pg/mL, according to the Cleveland Clinic’s Reference Range, were excluded from logistic regression analysis. The multivariate logistic regression models and EFV quartile multivariate logistic regression models were used to evaluate the association of EFV with AF, with the multivariate model 1 adjusted with age and gender, the model 2, as for model 1, additionally adjusted for BMI, hypertension, diabetes, smoking, total and high-density lipoprotein cholesterol, and use of lipid-lowering medication, and the model 3, as for model 2, additionally adjusted for obstructive CAD and echocardiographic left atrial diameter and EF.
All statistical analyses were performed using SPSS17.0 software (SPSS Inc., Chicago, IL, USA) and a two-tailed P value <0.05 was considered to be statistically significant.
Results
Clinical Characteristics of the Patients Undergoing Coronary CT Angiography Examination
A total of 2042 patients (1128 males and 914 females), average age 65.0 years (males 64.1 years and females 66.1 years) who had CCTA scan and echocardiography examination in Shanghai East Hospital during 2012–2014 were given a detailed analysis of pericardial adipose tissue quantification and association with AF. The clinical characteristics, AF and measurements of pericardial adipose tissue and echo parameters are shown in Table 1. The proportion of participants having AF was 8.5% overall, with a significantly higher proportion in men (9.8%) than in women (7.0%, p=0.013). The male participants had higher occurrence of overweight and obesity than female participants, 41.2% vs. 36.1% (p=0.020) and 13.1% vs. 10.6% (p=0.047) respectively.
 |
Table 1 The Clinical Characteristics and Pericardial Adipose Tissue Quantification in Men and Women |
As for pericardial fat deposition in this population, significantly higher PeCFV was observed in men than in women (122.9 cm3 vs. 109.3 cm3 in men and women, p<0.001), and this was mainly because of higher PaCFV in men than in women (47.9 cm3 vs. 35.8 cm3 in men and women, p<0.001), while there was no significant difference in EFV between men and women (75.0 cm3 vs. 73.4 cm3 in men and women, p=0.261). Also, men had higher PAFV (18.0 cm3 vs. 11.4 cm3 in men and women, p<0.001), and LAD (38.1 cm vs. 36.4 cm in men and women, p<0.001), but lower EF (62.4 vs. 64.5, p<0.001) than women.
The Incidence and Risk Factors for Atrial Fibrillation
The presence of RHD was 11.6% overall in patients with AF with a significantly higher proportion in women than in men (22.2% vs. 5.5%, p=0.013), while no significant difference in the presence of other valvular diseases was observed between men and women (7.3% in men vs. 11.1% in women, p=0.320) with AF. However, there was a significantly higher proportion of non-valvular risk factors related to AF in men than in women (84.7% in men vs. 66.7% in women, p=0.008) (Table 2). With increasing age, there was a significantly reduced proportion of RHD-related AF (p=0.041) and a reduced proportion of other valvular diseases-related AF without reaching significance (p=0.176). However, significantly increased risk factors related to non-valvular AF were observed with increasing age (p=0.003) (Table 2).
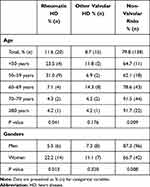 |
Table 2 Atrial Fibrillation Risk Factors in Different Age and Genders |
Pericardial Fat Volume Comparing Non-Valvular AF and Patients without AF
In order to determine the association of AF and non-valvular cardiometabolic risk factors, we excluded those patients with known risk factors for AF including RHD, other valvular diseases and those with EF well below normal (EF < 35%) or with NT-proBNP >900 pg/mL according to the Cleveland Clinic’s Reference Range, and the data comparing patients with non-valvular AF and patients without AF were shown in Table 3 and Supplementary Table 1 stratified by gender.
 |
Table 3 Pericardial Fat and Cardiometabolic Parameters in Non-Valvular AF and Patients without AF |
Patients with non-valvular AF had elevated CAC scores (203.5 vs. 240.0, p=0.269) and significantly increased obstructive coronary artery disease (CAD) evaluated with coronary artery stenosis by the plaques in CCTA (p=0.046) compared with the patients without AF. Further analysis of the risk factors for CAD showed that AF patients had significantly higher BMI (24.3 kg/m2 vs 25.2 kg/m2, p=0.012) and EFV (73.6 cm3 vs 84.0 cm3, p<0.001) with more pronounced elevation of EFV than BMI, and no significant change of PaCFV was observed (p=0.141) (Table 3). A gender difference was observed with a more significant difference in both BMI (23.9 kg/m2 vs 25.4 kg/m2, p=0.006) and EFV (72.7 cm3 vs 86.2 cm3, p=0.003) in women, while men had significant elevation of EFV (74.3 cm3 vs 84.0 cm3, p=0.012) although no significant difference of BMI (p=0.132) (Supplement Table 1).
AF patients exhibited reduced high-density lipoprotein cholesterol (HDL-C) and total cholesterol (TC) overall and the reduction of TC, possibly due to statin use, was significant overall (p=0.012) and separately with men (p=0.045) and women (p=0.073). There was no significant difference in SBP overall, but there was increased use of antihypertensive medication in men, and significantly higher SBP with no difference of antihypertensive medication use in women with AF. Also, no difference of plasma glucose was observed between patients with and without AF, with men having even less usage of hypoglycemic medication (p=0.032) (Table 3 and Supplementary Table 1).
Association of Epicardial Fat Volume and AF
The incidence of AF was higher in men than in women (Table 1) and the incidence of non-valvular AF increased significantly with increasing age (Table 2), thus we used multivariate logistic regression models to evaluate the association of EFV with the presence of AF (Table 4). A significant association of EFV with AF incidence was found in model 1 after adjustment with age and gender (p<0.001) and this was still significant in model 2 after adjustment with age, gender, BMI, hypertension, diabetes, smoking, TC and HDL-C, and use of lipid-lowering medication statins (p=0.048). However, this was not significant in model 3 after additional adjustment with obstructive CAD and LAD and EF by echocardiography examination (p=0.234) (Table 4).
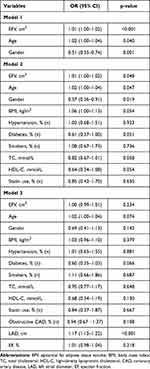 |
Table 4 Multivariate Logistic Regression Analysis of Epicardial Fat Volume and Atrial Fibrillation |
Furthermore, we used EFV quartile categorical multivariate logistic regression models to look at the association of AF and EFV in detail. The mean EFV overall was 74.3 cm3 (Table 1) and the 25th, 50th and 75th percentile values were 53.2, 70.0 and 91.5 cm3 respectively (Table 5). The incidence of AF in the four EFV quartiles was 19, 26, 36 and 57 patients, respectively, and the results of EFV quartile categorical multivariate logistic regression models indicated a significant association of the upper 2 quartiles of EFV with an increased incidence of AF in model 1 and model 2, and the highest quartile in model 2 and model 3 (Table 5). These results suggested that there was an association of EFV with an increased incidence of AF that was independent of traditional cardiometabolic risk factors and general obesity markers such as BMI, SBP, as well as obstructive CAD and left atrial size in EFV patients of the highest EFV quartile.
 |
Table 5 CT-Derived Epicardial Fat Volume and Non-Valvular AF in Chinese Patients |
Discussion
The incidence of AF is increasing in both Western and Asian populations,25,26 and several demographic and clinical risk factors were independently associated with AF including ageing, male sex, obesity, hypertension, coronary artery disease. Obesity is a major global public health issue, implicated in a vast array of conditions affecting multiple body systems and data from epidemiological cohorts support the role of obesity as an independent risk factor for AF.7,27 Although BMI and other clinical measures are useful indicators of general adiposity, recent interest has focused on epicardial fat, a distinct adipose tissue depot that can be readily assessed using non-invasive imaging techniques. Growing evidence from epidemiological and clinical studies has demonstrated that epicardial fat is consistently associated with the presence, severity, and recurrence of AF.15,28
In this study, we found that AF was present in 8.5% of patients undergoing CCTA and the incidence of AF without rheumatic and other valvular diseases significantly increased with increasing age. Furthermore, CT measurement of the epicardial fat showed that patients with non-valvular AF had significantly higher pericardial fat volume with more pronounced elevation of EFV, and a significant association of EFV with AF was observed even after adjustment for BMI and clinical cardiovascular risk factors, as well as for the left atrial size and obstructive CAD in the highest quartile of EFV.
AF is the most common arrhythmia in the general population and is associated with increased risk of ischemic stroke/systemic embolism, heart failure (HF), and cardiac mortality. The prevalence and incidence of new-onset AF is increasing worldwide, with an estimated rate of 9.03 per 1000 patient-years.26 In general, Asian populations have a lower risk of developing AF compared with non-Asian populations.26,29 The prevalence of AF in China was 0.41% in men and 0.43% in women in participants aged less than 60 years, and 1.83% in both men and women aged 60 years and over, and about 19.0% of males and 30.9% of females with AF were diagnosed with valvular disease.30 An epidemiological study including 13 cohorts with 29,079 Chinese patients31 and another large cohort study (n = 471,446) in South-Western China32 found that RHD was the most important risk factor for new-onset AF. We found that the incidence of AF in patients attending Shanghai East Hospital for CCTA during 2012 and 2014 was 8.5% (9.8% in men and 6.9% in women) with 11.6% of AF patients having RHD and 8.7% having other valvular diseases. With increasing age, the proportion of RHD was significantly reduced but the risk factors for non-valvular AF increased.
Apart from RHD and valvular diseases, many other clinical risk factors, including ageing, male sex, obesity, hypertension, diabetes, coronary artery disease, renal dysfunction and tobacco consumption are associated with AF. The worldwide epidemic of overweight and obesity has driven the increasing impact of adiposity on cardiovascular disease risks, and obesity has been reported to account for almost one-fifth of AF cases, and its population attributable-risk is likely to continue rising as developing countries undergo epidemiologic transition.33 Recent studies demonstrated that the obesity-driven expansion of EFV was associated with paroxysmal and/or persistent AF,12–15,28 and with ethnic differences.16 Our study in Chinese patients confirmed the significant association of EFV with AF even after adjustment for BMI and clinical risk factors. EFV quartile analysis showed that the 25th, 50th and 75th percentile of EFV were 53.2, 70.1 and 91.6 cm3, which are lower than in the Dutch prospective population-based Rotterdam cohort study, and moreover, we found a significant association of EFV with AF in the patients in the highest quartile of EFV, which was independent of the left atrial size and obstructive CAD.
Of note, pericardial fat is often referred to interchangeably with EAT, but strictly speaking paracardial fat (PaCFV) refers to the layer of fat external to the parietal pericardium and pericardial fat (PeCFV) is the combination of both EAT and paracardial fat. Most studies evaluated the association of EFV and AF. In this study, we measured EFV, PaCFV and total PeCFV and found a significant elevation of EFV with no significant difference of PaCFV between patients with non-valvular AF and patients without AF. The mechanisms involved in the association between EAT and AF might include direct and indirect effects.10 Histologically, there is no fascia boundary separating the EAT from the myocardium, hence, a direct infiltration of cells from adipose tissue into the atrial myocardium could contribute to the remodeled atria and increased AF risk. Also, EAT is an endocrine organ and a source of proinflammatory cytokines and profibrotic factors which act in a paracrine way on the myocardium contributing to the structural remodeling of the atria and increased AF risk.10
In conclusion, we found significantly increased risk factors related to non-valvular AF with increasing age, and EFV was associated with non-valvular AF, which was independent of generalized adiposity and clinical risk factors, as well as the left atrial size and obstructive CAD in AF patients in the highest quartile of EFV. The novelty of this study is that adiposity is an important risk factor for AF and a stronger association of EFV with non-valvular atrial fibrillation than general obesity observed in Chinese patients, however, several limitations deserve comment. The study participants are Chinese hospital patients with a relatively small number of patients with AF and it is an observational cross-sectional study which cannot prove that EFV is a causative risk factor for AF incidence. Also, we used the thoracic CT scan to measure the total cardiac EFV and a more accurate magnetic resonance imaging measurement of EFV or more specifically left atrial EFV to look for the association with the presence, severity, and recurrence of AF may be more appropriate.
Funding
This study is supported by the funds from the Science and Technology Commission of Shanghai Municipality (19411965300, 17431906600), Top-level Clinical Discipline Project of Shanghai Pudong District Grant (PWYgf2018-02), the National Natural Science Foundation of China (81770259, 81970232, 81970233, 81970234, 81800253, 81770094, 81870202), the Health System Academic Leader Training Plan of Pudong District Shanghai (PWRd2018-06), and the Fundamental Research Funds for the Central Universities (22120180380).
Disclosure
Dr Brian Tomlinson reports speaker fees from Amgen Inc., Kowa, and Merck Serono, outside the submitted work. The authors report no other conflicts of interest in this work.
References
1. Lavie CJ, Pandey A, Lau DH, Alpert MA, Sanders P. Obesity and atrial fibrillation prevalence, pathogenesis, and prognosis: effects of weight loss and exercise. J Am Coll Cardiol. 2017;70(16):2022–2035. doi:10.1016/j.jacc.2017.09.002
2. Wong CX, Lau DH, Sanders P. Atrial fibrillation epidemic and hospitalizations: how to turn the rising tide? Circulation. 2014;129(23):2361–2363. doi:10.1161/CIRCULATIONAHA.114.010073
3. Vyas V, Lambiase P. Obesity and atrial fibrillation: epidemiology, pathophysiology and novel therapeutic opportunities. Arrhythm Electrophysiol Rev. 2019;8:28–36. doi:10.15420/aer.2018.76.2
4. Wang TJ, Parise H, Levy D, et al. Obesity and the risk of new-onset atrial fibrillation. JAMA. 2004;292:2471–2477. doi:10.1001/jama.292.20.2471
5. Wong CX, Sullivan T, Sun MT, et al. Obesity and the risk of incident, post-operative, and post-ablation atrial fibrillation: a meta-analysis of 626,603 individuals in 51 studies. JACC Clin Electrophysiol. 2015;1:139–152. doi:10.1016/j.jacep.2015.04.004
6. Frost L, Hune LJ, Vestergaard P. Overweight and obesity as risk factors for atrial fibrillation or flutter: the Danish diet, cancer, and health study. Am J Med. 2005;118:489–495. doi:10.1016/j.amjmed.2005.01.031
7. Foy AJ, Mandrola J, Liu G, Naccarelli GV. Relation of obesity to new-onset atrial fibrillation and atrial flutter in adults. Am J Cardiol. 2018;121:1072–1075. doi:10.1016/j.amjcard.2018.01.019
8. Fox CS, Massaro JM, Hoffmann U, et al. Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the framingham heart study. Circulation. 2007;116:39–48. doi:10.1161/CIRCULATIONAHA.106.675355
9. Lau DH, Schotten U, Mahajan R, et al. Novel mechanisms in the pathogenesis of atrial fibrillation: practical applications. Eur Heart J. 2016;37:1573–1581. doi:10.1093/eurheartj/ehv375
10. Wong CX, Ganesan AN, Selvanayagam JB. Epicardial fat and atrial fibrillation: current evidence, potential mechanisms, clinical implications, and future directions. Eur Heart J. 2017;38:1294–1302. doi:10.1093/eurheartj/ehw045
11. Sacks HS, Fain JN. Human epicardial adipose tissue: a review. Am Heart J. 2007;153:907–917. doi:10.1016/j.ahj.2007.03.019
12. Muhib S, Fujino T, Sato N, Hasebe N. Epicardial adipose tissue is associated with prevalent atrial fibrillation in patients with hypertrophic cardiomyopathy. Int Heart J. 2013;54(5):297–303. doi:10.1536/ihj.54.297
13. Al Chekakie MO, Welles CC, Metoyer R, et al. Pericardial fat is independently associated with human atrial fibrillation. J Am Coll Cardiol. 2010;56(10):784–788. doi:10.1016/j.jacc.2010.03.071
14. Nakamori S, Nezafat M, Ngo LH, Manning WJ, Nezafat R. Left atrial epicardial fat volume is associated with atrial fibrillation: a prospective cardiovascular magnetic resonance 3d dixon study. J Am Heart Assoc. 2018;7(6):e008232. doi:10.1161/JAHA.117.008232
15. Wong CX, Sun MT, Odutayo A, et al. Associations of epicardial, abdominal, and overall adiposity with atrial fibrillation. Circ Arrhythm Electrophysiol. 2016;9:e004378. doi:10.1161/CIRCEP.116.004378
16. Heckbert SR, Wiggins KL, Blackshear C, et al. Pericardial fat volume and incident atrial fibrillation in the multi-ethnic study of atherosclerosis and jackson heart study. Obesity. 2017;25(6):1115–1121. doi:10.1002/oby.21835
17. Inoue H, Kodani E, Atarashi H, Okumura K, Yamashita T, Origasa H. Impact of body mass index on the prognosis of Japanese patients with non-valvular atrial fibrillation. Am J Cardiol. 2016;118(2):215–221. doi:10.1016/j.amjcard.2016.04.036
18. Tam W-C, Lin Y-K, Chan W-P, et al. Pericardial fat is associated with the risk of ventricular arrhythmia in Asian patients. Circ J. 2016;80(8):1726–1733. doi:10.1253/circj.CJ-16-0047
19. Peng S, Shen T, Liu J, et al. Uncontrolled hypertension increases with age in an older community-dwelling Chinese population in Shanghai. Aging Dis. 2017;8(5):558–569. doi:10.14336/AD.2016.1220
20. Britton KA, Massaro JM, Murabito JM, Kreger BE, Hoffmann U, Fox CS. Body fat distribution, incident cardiovascular disease, cancer, and all-cause mortality. J Am Coll Cardiol. 2013;62(10):921–925. doi:10.1016/j.jacc.2013.06.027
21. Austen WG, Edwards JE, Frye RL, et al. A reporting system on patients evaluated for coronary artery disease. Report of the ad hoc committee for grading of coronary artery disease, council on cardiovascular surgery, American heart association. Circulation. 1975;51(4):5–40. doi:10.1161/01.CIR.51.4.5
22. Puchner SB, Liu T, Mayrhofer T, et al. High-risk plaque detected on coronary ct angiography predicts acute coronary syndromes independent of significant stenosis in acute chest pain. J Am Coll Cardiol. 2014;64(7):684–692. doi:10.1016/j.jacc.2014.05.039
23. Chen C, Lu FC. The guidelines for prevention and control of overweight and obesity in Chinese adults. Biomed Environ Sci. 2004;17(Suppl):1–36.
24. Cury RC, Abbara S, Achenbach S, et al. Cad-rads(TM) coronary artery disease-reporting and data system. An expert consensus document of the society of cardiovascular computed tomography (SCCT), the American college of radiology (ACR) and the North American society for cardiovascular imaging (NASCI). Endorsed by the American college of cardiology. J Cardiovasc Comput Tomogr. 2016;10:269–281.
25. Krijthe BP, Kunst A, Benjamin EJ, et al. Projections on the number of individuals with atrial fibrillation in the European union, from 2000 to 2060. Eur Heart J. 2013;34:2746–2751. doi:10.1093/eurheartj/eht280
26. Dewland TA, Olgin JE, Vittinghoff E, Marcus GM. Incident atrial fibrillation among Asians, hispanics, blacks, and whites. Circulation. 2013;128:2470–2477. doi:10.1161/CIRCULATIONAHA.113.002449
27. Javed S, Gupta D, Lip GYH. Obesity and atrial fibrillation: making inroads through fat. Eur Heart J Cardiovasc Pharmacother. 2021;7(1):59–67.
28. Gaeta M, Bandera F, Tassinari F, et al. Is epicardial fat depot associated with atrial fibrillation? A systematic review and meta-analysis. Europace. 2017;19:747–752. doi:10.1093/europace/euw398
29. Bai Y, Wang YL, Shantsila A, Lip GYH. The global burden of atrial fibrillation and stroke: a systematic review of the clinical epidemiology of atrial fibrillation in Asia. Chest. 2017;152:810–820. doi:10.1016/j.chest.2017.03.048
30. Li Y, Wu YF, Chen KP, et al. Prevalence of atrial fibrillation in China and its risk factors. Biomed Environ Sci. 2013;26:709–716. doi:10.3967/0895-3988.2013.09.001
31. Zhou Z, Hu D. An epidemiological study on the prevalence of atrial fibrillation in the Chinese population of Mainland China. J Epidemiol. 2008;18:209–216. doi:10.2188/jea.JE2008021
32. Guo Y, Tian Y, Wang H, Si Q, Wang Y, Lip GYH. Prevalence, incidence, and lifetime risk of atrial fibrillation in China: new insights into the global burden of atrial fibrillation. Chest. 2015;147:109–119. doi:10.1378/chest.14-0321
33. Schnabel RB, Yin X, Gona P, et al. 50 year trends in atrial fibrillation prevalence, incidence, risk factors, and mortality in the framingham heart study: a cohort study. Lancet. 2015;386:154. doi:10.1016/S0140-6736(14)61774-8
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
