Back to Journals » OncoTargets and Therapy » Volume 13
A Review of Research Progress in Multidrug-Resistance Mechanisms in Gastric Cancer
Authors Ruan T, Liu W, Tao K, Wu C
Received 20 November 2019
Accepted for publication 15 February 2020
Published 28 February 2020 Volume 2020:13 Pages 1797—1807
DOI https://doi.org/10.2147/OTT.S239336
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Takuya Aoki
Tuo Ruan, Weizhen Liu, Kaixiong Tao, Chuanqing Wu
Department of Gastrointestinal Surgery, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China
Correspondence: Kaixiong Tao; Chuanqing Wu
Department of Gastrointestinal Surgery, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China
Email [email protected]; [email protected]
Abstract: Gastric cancer is one of the most common malignant tumors, and it is also one of the leading causes of cancer death worldwide. Because of its insidious symptoms and lack of early dictation screening, many cases of gastric cancer are at late stages which make it more complicated to cure. For these advanced-stage gastric cancers, combination therapy of surgery, chemotherapy, radiotherapy and target therapy would bring more benefit to the patients. However, the drug-resistance to the chemotherapy restricts its effect and might lead to treatment failure. In this review article, we discuss the mechanisms which have been found in recent years of drug resistance in gastric cancer. And we also want to find new approaches to counteract chemotherapy resistance and bring more benefits to the patients.
Keywords: gastric cancer, chemotherapy, resistance
Introduction
Even with the development in medical detection and therapy, the morbidity of gastric cancer still remains to be ranking fourth in all cancers. And it is also the second cause of death due to cancers around the world. East Asian regions, especially China, Japan and Korea, are high-prevalence areas for gastric cancer.1 In China, gastric cancer is the second most common and deadliest cancer just after lung cancer in 2015. It has been estimated that there are over 700,000 new cases every year, and most of them are in rural area. On the other side, because of its insidious symptoms and lack of early dictation screening, many cases in China are at late stages which bring more difficulties to clinical treatments.2,3 For advanced-stage gastric cancers, combination therapy of surgery, chemotherapy, radiotherapy and target therapy would bring more benefit to the patients.4,5 Chemotherapy is one of the main additional methods in treatment, but the rise of drug resistance restricts its effect and might lead to treatment failure.6–8 In order to solve this problem, we need to study the mechanisms of the chemotherapy resistance and find solutions.
The mechanisms of drug resistance to chemotherapy are complex. According to what we have known, we conclude them into seven aspects as followed: (1) Reduce the effective concentration of intracellular drugs; (2) Change of drugs’ targets; (3) Dysfunction of DNA damage repairing; (4) Change of apoptosis and autophagy; (5) Change of tumor micro-environment; (6) Extracellular vesicles and macropinocytosis; (7) MicroRNAs and LncRNAs. Although we have made much progress in studying the molecular mechanisms of the drug resistance, it still lacks applied technique in detecting and controlling the chemotherapy resistance in clinical practice.9,10 In this article, we would like to summarize and analyze the research progress in chemotherapy in gastric cancer, and it might lead us to new solutions to this problem.
Reduce the Effective Concentration of Intracellular Drugs
Drug Efflux
The ATP-binding cassette family is a group of the most well-known proteins in constructing transporters in cells.11–13 These transporters will pump the drugs out of the cells and lead to multi-drug resistance. In these proteins, P-gp, ABCG2 and MRP-1 have been well studied in solid tumors, such as breast cancer, ovarian cancer and gastrointestinal cancers.14–16 It has been proofed that P-gp is overexpressed in gastric cancer, and would be related to the poor prognosis of the patients.17,18 The expression of P-gp is also highly related to acquired drug resistance and the risk of relapse after chemotherapy which makes it important in our studies.16,19
The regulation pathways of P-gp in regulating drug resistance are diverse. MAPK pathway proteins play important roles as down-stream receptors and in regulating intracellular environment when P-gp is up-regulated in cancer cells.20 NF-κB could also enhance the expression of P-gp by targeted combining the promoter of P-gp gene to induce multi-drug resistance.21,22 On the other hand, inhibiting PI3K/AKT pathway could reduce the expression of P-gp and reverse drug resistance.23 The pathways and molecules which we have mentioned above could serve as targets for us to inhibit drug resistance and provide us with new solutions to this problem.24,25
Drug Inactivation
Normal cells could decompose and transform the toxins and their intermediate products to detoxification in order to maintain stable homeostasis. This is also the way that cancer cells decrease the damage from chemotherapeutics.
Glutathione S-transferases (GST) are important in inactivating drugs and induce drug resistance. The main mechanisms are as followed: (1) Catalyze glutathione to combine with electrophilic substrates and prevent reactive oxygen species from causing damage to cell membranes; (2) Up-regulated glutathione and GST will enhance the polarity of drugs to make them inactive; (3) GST could also remove toxic metabolites directly to reduce damage.26,27 In some researches, we could find that the expression of GST is higher in cancer tissues than normal epithelium.28 Specifically, it has a strong connection with the drug resistance to platinum.29,30
Change of Drugs’ Targets
Tumor cells could reduce the level of drugs’ targets or enzymes’ activity in cells to induce drug resistance. DNA topoisomerase is served as drug targets by many commonly used chemotherapeutics, such as doxorubicin and etoposide. DNA topoisomerase is involved in DNA replication, recombination and repairing process of ribozymes. It is an important part in cell cycle regulation. There are two types of DNA topoisomerase isomers: topoisomerase I (Topo I) and topoisomerase II (Topo II). In gastric cancer, the expression of Topo II is up-regulated, and it has an influence on infiltration depth, histological type and lymph node metastasis. Topo II is also a key factor in inducing resistance to adriamycin and mitomycin C in gastric cancer, and it is correlated to prognosis.31–34 Research has noticed that down-regulating the expression of Topo II would strengthen the resistance to adriamycin.35 This would lay the foundation for us to solve this kind of chemotherapy resistance.
With the development of new drugs and therapies, molecule-targeted therapy has become a hotspot in recent years for its higher efficiency and less side-effects.36,37 In gastrointestinal cancers, imatinib has been widely used in curing GIST and has made great progress. It targets at tyrosine kinase to inhibit PDGF receptors, SCF and c-Kit receptors. While in clinical practice, we have found that the mutation of Kit and BRAF gene would lead to drug resistance to imatinib.38,39 This situation demands us to study in the next generation of targeted drugs and make individual treatment more specific.
Dysfunction of DNA Damage Repairing
In normal cells, DNA damage repairing is activated when they have physical, chemical or biological damages in order to stabilize chromatin. However, when the repairing system is dysfunctional, it would lead to oncogenesis.40,41 Chemotherapy utilizes DNA damage and cytotoxicity as an approach to kill cancer cells, especially for platinum drugs. Platinum inhibits DNA transcription and replication, and it might cause DNA cleavage which makes DNA damage repairing activation a key factor in drug resistance to platinum.42,43
Nucleotide excision repair (NER) is an important pathway to repair the damage caused by chemotherapy drugs through identification, resection and reconstruction of damaged DNA. And this progress is composed of dozens of proteins.44,45 While DNA mismatch repair genes (MMR), such as MLH1, also play a role in the development of gastric cancer and inducing drug resistance.46 These pathways could offer us a road to reverse drug resistance to platinum and bring benefits to the patients.
Change of Apoptosis and Autophagy
Dysfunction of Apoptosis Pathways
There are two classical apoptosis pathways in human: (1) Endogenous pathway mediated by mitochondria; (2) Exogenous pathways mediated by tumor necrosis factor (TNF) receptors.47
The Bcl-2 protein family contains a variety of apoptosis-related proteins, such as Bax, Bad and Bid which induce apoptosis, and Bcl-2, Bcl-xl which inhibit the progress. These proteins interact with each other in order to maintain a relatively stable state for cells. When this balance is broken, it would likely lead to carcinogenesis and have resistance effects on chemotherapeutics which aim at inducing cell apoptosis.48,49 In gastric cancer, studies have shown that the over-expression of Bcl-2 is highly related to drug resistance, and it is also an important influence factor in prognosis.50,51 Bax, which is promoting apoptosis, has been confirmed to be used in predicting the patients’ response to chemotherapy.52 Upregulating the expression of Bax could help to promote sensitivity to chemotherapy in cancer cells by releasing cytochrome C from mitochondria.53 In addition, other members in the Bcl-2 family, such as Bcl-xL and Bak, also have been proved to be playing relevant roles in drug resistance.54,55 These make Bcl-2 family critical in gastric cancer development and chemotherapy resistance. They would hopefully be a breakthrough point in our research.
Tumor necrosis factor (TNF) family proteins, such as NFRS-1, Fas, DR4 and DR5, are also important in cell apoptosis regulation. Studies have shown that Fas could induce cell apoptosis to eliminate tumor cells by combining with FasL, while the increase in soluble Fas will inhibit apoptosis.56,57 A recent study has found that the expression of TNF-related apoptosis-inducing ligand (TRAIL) had a negative correlation with multidrug resistance-associated protein P-gp, and P-gp could achieve multi-drug resistance by regulating the expression of TRAIL and its mediated apoptosis pathway.58
P53 is one of the most popular tumor suppressor genes in our studies, and it has multiple functions in cell cycle regulation, DNA repairing and cell apoptosis. Researches have shown that p53 mutation is an important influence factor in gastric cancer development, and high expression of p53 gene is associated with poor prognosis.59,60 At the same time, p53 also takes part in gastric cancer drug resistance. We could find that patients with mutant p53 have a better response to chemotherapy.61 And recent studies suggest that rAd-p53 could induce cell apoptosis in gastric cancer cells, and p53 could reverse cisplatin resistance by regulating the AKT signaling pathway and the expression of Bax.62,63
PI3K/AKT pathway is crucial in cell growth and proliferation. Abnormal activation of the PI3K/AKT pathway has been reported to be one of the important mechanisms of inducing drug resistance in tumor cells.64,65 And further studies have showed that some chemotherapeutics would activate the PI3K/AKT pathway which resulted in acquired drug resistance.66,67 So far, the mechanisms of the PI3K/AKT pathway have not been fully understood. And the abnormal activation of the PI3K/AKT pathway in tumor cells might be via PI3K catalytic subunit of alpha (PIK3CA) mutation or loss of PTEN gene function.68,69 NF-κB could serve as a down-stream targeted protein and take part in inducing drug resistance.70 P-gp, Bcl-2 and Bax could also be regulated by phosphorylated AKT.24 At the same time, how to make use of the PI3K/AKT pathway in clinical treatment is also on the schedule. Now, research has proved that the AKT inhibitor could reverse the drug resistance of gastric cancer cells.71 Further validation and application researches have been carried out and would get promising results for clinical works.72
Change of Autophagy Pathways
Autophagy is a cellular survival mechanism which is a highly conserved cellular process to degrade cytoplasmic materials and recycle to maintain energy homeostasis. And it could be induced by cancer therapy, among other stresses, and frequently contributes to cancer cell survival.73–75 Autophagy can also be classified as macro-autophagy, micro-autophagy, and chaperone-mediated autophagy.
Molecular mechanisms of autophagy could be divided into initiation, nucleation and elongation of autophagosome, fusion of autophagosome with the lysosome, and degradation of sequestered material. Autophagy is constitutively activated in cancer cells through the deregulation of PI3K/Akt/mTOR molecular axis and AMP-activated protein kinase (AMPK) signal transduction, which contributes to the metabolic reprogramming of cancer cells.76–78
A large amount of cancer therapies has been shown to induce autophagy, while many of them indicate that autophagy turns out to promote tumor cell survival and contribute to therapy resistance.79,80 Some studies have also shown that autophagy-related proteins, such as Beclin 1 (BECN1), microtubule-associated protein1 light chain 3 (MAP1-LC3), and p62/sequestosome 1 (SQSTM1) have an important prognostic value in gastric cancer, and act as a protective mechanism for tumor cells in chemotherapy, promoting drug resistance as well.81–83 The therapeutic induction of autophagy is frequently attributed to reduced mTOR activity leading to autophagy de-repression, and this is most obvious with therapies targeted at inhibiting PI3K, AKT or indeed mTOR itself. In addition, autophagy is induced by conventional genotoxic agents, such as radiation or cisplatin, as a result of DNA damage-induced p53 activity. However, the role of p53 in these responses is complicated. Depending on the context, it can also inhibit autophagy.84,85 As there are a growing number of studies in autophagy and its cytoprotective effect, there might be a chance to help cancer therapeutic approaches with agents that inhibit autophagy.86,87
Change of Micro-Environment
There is extracellular matrix, a variety of trace elements, immune cells and other substances in tumor’s micro-environment. These substances have multiple effects on proliferation, invasion, metastasis and multi-drug resistance.88–90
Extracellular Matrix
Extracellular matrix is a complex network containing multifunctional molecules and has an important influence on tumor development. Laminin and collagen IV are important parts of the basement membrane in the gastric cancer’s extracellular matrix.91 And the overexpression of laminin would enhance gastric cancer cells’ resistance to vincristine and adriamycin via regulating MGr1-Ag/37LRP.92 This phenomenon also indicates that cell adhesion ability is associated with the chemo-resistant phenotype of gastric cancer cells, which makes it has a chance to be considered as a reference target to detect drug resistance. The mechanisms of how laminin induced drug resistance in gastric cancer are varied, including regulating the expression of multiple drug resistance-related proteins such as P-gp, and regulating apoptosis-related proteins Bcl-2 and Bax to inhibit drug-mediated apoptosis and multiple signaling pathways (PI3K/AKT and MAPK/ERK).92,93 Besides these, the high expressions of metalloproteinases inducer HMGB1 are also related to drug resistance.94,95 Thus, there are still more details needed to be explored between the extracellular matrix and tumor drug-resistance mechanism which will bring more benefits to clinical treatment.
Cytokines and Growth Factors
The cytokines and growth factors in tumor micro-environment are involved in the activation of a variety of signaling pathways and play a key role in chemoresistance. Many cytokines and growth factors have been confirmed to act as independent factors that influence prognosis in gastric cancer patients under chemotherapy treatment.96 Interleukin family is a major part of cytokines that have been well studied. IL-6 could lead to acquired drug resistance to trastuzumab in gastric cancer cells via activation of the STAT3 signaling pathway.97 IL-33 could cause resistance to platinum through the JNK signaling pathway.98 And IL-24 has also been proved to be involved in regulating multi-drug resistance.99
Anoxia
Anoxia is an important biological characteristic in tumorigenesis and is also one of the mechanisms in drug resistance. HIF-1α is critical in cells’ response to hypoxia, and the over-expression of HIF-1α has been confirmed to be related to poor prognosis in gastric cancer in multiple studies which makes it an independent factor predicting prognosis.100–102 Some studies have showed that HIF-1α could regulate drug resistance by regulating the expression of p53, NF-κB, Bcl-2 and serving as the down-stream target of PI3K/AKT and MAPK/ERK signaling pathways.103–106 HIF-1α has played a significant role in inducing drug resistance in gastric cancer, while the mechanisms are still needed to be explored.103
Extracellular Vesicles and Micropinocytosis
Extracellular vesicles (EVs) are secreted by nearly almost cells and released to the extracellular space. EVs could be divided into three different subgroups according to their size: exosomes (30–100 nm), micro-vesicles (MVs, 100–1000 nm), and apoptotic bodies (1000–5000 nm). While exosomes are small membrane nanovesicles which constituted through the intraluminal budding of the late endosomal membrane and are secreted from the plasma membrane.107–109 Recently, the finding that paclitaxel-resistant gastric cancer cell line (MGC-803R) cell-derived exosomes could be taken up by paclitaxel-sensitive MGC-803 (MGC-803S) cells has been reported. Furthermore, the exosomal miR-155-5p from MGC-803R cells can join in directly inhibiting GATA binding protein 3 (GATA3) and tumor protein p53-inducible nuclear protein 1 (TP53INP1) to induce chemoresistant phenotypes in the sensitive cells which uptake the exosomes.110,111 In addition, exosomal miR-21 can be directly transferred from macrophages to gastric cancer cells to obtain the chemotherapy resistance, and inhibit cell apoptosis and activate the PI3K/AKT pathway by regulating PTEN.112
Macropinocytosis has been known as a primary method for the cellular intake of fluid-phase and membrane-bound bulk cargo. And recent researches show that direct roles for macropinocytosis within tumorigenesis.113 Uptake of nutrients in the tumor microenvironment by macropinocytosis has recently been named as an emerging hallmark of cancer metabolism.114,115 In lung cancer cell line A549, it has been demonstrated the extracellular ATP was internalized by macropinocytosis and induced intracellular ATP increase and drug resistance.116 In addition, there are several researches associated with macropinocytosis and drug resistance in pancreatic cancer and breast cancer.117,118 The mechanisms and effects of micropinocytosis in gastric cancer need further study, and it may lead to a new way to solve the multi-drug resistance in gastric cancer.
MicroRNAs and LncRNAs in Drug Resistance in Gastric Cancer
With the development of research in drug resistance, new research objects are introduced into this study. MicroRNAs are noncoding single-stranded RNA molecules which contain 18 to 22 nucleotides encoded by endogenous genes. They are involved in regulating gene expression and have extensive application prospects in tumor researches,119 while lncRNAs refer to nucleotides long-chain non-coding RNA which have more than 200 nucleotides. And recent studies reveal that they are involved in cell cycle regulation, epigenetic regulation and many other aspects which make them become a focus of current researches. LncRNAs also play an important role in tumorigenesis and might be the breakthrough point.120,121 Now, we will, respectively, introduce the two types of molecules in the research of gastric cancer’s drug-resistance briefly.
MicroRNAs in Drug Resistance of Gastric Cancer
MicroRNAs have been extensively studied in recent years; their important roles in numerous biological behaviors begin to be revealed. A growing number of studies prove that microRNAs are significant in regulating drug transporters, transcription factors and nuclear receptors which may lead us to a new approach in drug-resistant treatment.122 In the study of the drug resistance in gastric cancer, microRNAs are also involved in several classic signaling pathways.
Bcl-2-related apoptosis pathway is important in regulating drug resistance in gastric cancer, and the current researches have shown that a variety of microRNAs are involved in regulating this pathway. MiR-503 and miR-143 can regulate the expression of IGF1R and Bcl-2 to mediate gastric cancer cells’ resistance to cisplatin.123,124 The study of miR-200 BC/429 finds that high expression of miR-200 BC/429 will strengthen sensitivity of SGC7901/vincristine (VCR) cell line to cisplatin (CDDP), etoposide (VP-16) and adriamycin (ADR), while it is not suitable for 5-fluorouracil. And when these cells were transfected with miR-200 BC/429 inhibitor, their resistance to cisplatin, etoposide and adriamycin was enhanced. Through the luciferase assay, the Bcl-2 3ʹ-UTR reporters were detected in resistant SGC7901 cells which suggested that Bcl-2 was a target gene of the miR-200bc/429 cluster.125
PI3K/AKT signaling pathway is another important signaling pathway in drug-resistance mechanisms, while microRNAs are also involved in it. MiR-21 is detected highly expressed in SGC7901/cisplatin (CDDP) resistant cell line. Further experiments confirm that when the cells are induced with over-expression of miR-21, cell apoptosis caused by cisplatin injury would significantly reduce. On the other hand, decreasing the expression of miR-21 increases the anti-proliferative effects and CDDP-related apoptosis. It is also been found that miR-21 regulates cisplatin resistance via down-regulating the expression of PTEN and activation of the PI3K/AKT signaling pathway. This also provides us with a new method in handling the cisplatin resistance in gastric cancer via PI3K/PTEN/AKT pathway.126 Besides, miR-21 could also regulate trastuzumab’s efficiency in addition to chemotherapy in patients with HER2-positive gastric cancer. Over-expressed miR-21 would inhibit gastric cancer cell apoptosis caused by the drugs while inhibiting its expression would help with the therapy.127,128 This result would make microRNAs an important aid in individualized medicine.
For microRNAs involve in a wide variety of drug-resistance mechanism in gastric cancer, further researches are required to conduct.129,130 Although there is a long road for microRNAs from being applied to the treatment, they still introduce us with a new way for our future cancer treatment. New treatments based on microRNAs will come out in the near future promisingly.
LncRNAs in Drug Resistance of Gastric Cancer
In the study of gastric cancer, a variety of abnormal-expressed lncRNAs have been proved to be critical in tumorigenesis, metastasis and drug resistance. PVT1, a lncRNA which is associated with gastric cancer, is closely related with the development and MDR in gastric cancer. Recent researches show that PVT-1 is highly expressed in gastric cancer tissues of cisplatin-resistant patients and SGC7901/DDP cells. In addition, PVT1 is also a risk factor in lymph nodes metastasis. These features make PVT1 have the potential to be the new biomarkers for the evaluation of patients with gastric cancer.131,132 And in the prescription of drug resistance in gastric cancer, up-regulating PVT1’s expression would raise the expression of p-glycoprotein, MRP, mTOR and HIF-1α to induce multidrug-resistance, while inhibiting PVT1’s expression would reverse cell resistance to cisplatin.133,134 Other studies have shown that down-regulating the expression of LEIGC in gastric cancer cells could enhance the resistance to 5-fluorouracil via regulating the EMT process, which suggested LEIGC could be a suppressor lncRNA in gastric cancer.135 Further studies have found that the target site of lncRNA MRUL is close to p-glycoprotein, and the expression of MRUL is higher in drug-resistance cells. For p-glycoprotein is one of the important proteins in inducing multidrug resistance, patients with high expression of MRUL show poor reactivity of chemotherapy drugs in clinical practice. And another study reveals that MRUL induces resistance to adriamycin by regulating the expression of ABCB1 in gastric cancer.136
New lncRNAs are also added to some common molecular mechanism and signaling pathway due to the recent studies, such as Notch 1 could increase lncRNA AK022798 expression to induce resistance to cisplatin in gastric cancer.137 LncRNA HOTAIR could also activate the PI3K/AKT pathway and regulate the expression of p-glycoprotein through miR-216.138 On the other hand, the connection between lncRNAs and microRNAs has been revealed, such as lncRNA UCA1 could enhance drug resistance by down-regulating miR-27b.139 With the deepening of the researches, lncRNAs could play a more active role in gastric cancer drug-resistance treatment.
Discussion
The preferred treatment for advanced-stage gastric cancer is still surgical operation.140,141 However, for patients who have no chance to get surgical treatment, the ultimate goal of comprehensive treatment is to prolong survival and improve the quality of life. The development and progress of novel chemotherapy, targeted drugs, immunotherapy will provide new opportunities for the comprehensive treatment of gastric cancer. We could bring better clinical benefit to patients by carrying out a more comprehensive and personalized diagnosis and treatment strategy. Meanwhile, drug resistance is still one of the major obstacles in gastric cancer treatments. Although there are many mechanisms that have been revealed above, the clinical application is still limited.
The networks of drug-resistance-related pathways are complicated, and many proteins and molecules are involved (Figure 1). It might be hard to distinguish them apart. But the main goal of improving the therapy by enhancing the efficiency of the drugs is similar.
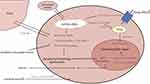 |
Figure 1 The summary of mechanisms of drug resistance in gastric cancer. |
As we have discussed above, the regulation of certain miRNA or lncRNA expressions could participate in improving the response of GC cell lines to chemotherapy and significantly enhance the antitumor properties of specific drugs. It could be helpful in developing personalized therapies, as well as establishing novel therapeutic strategies to reverse the resistance of tumors in combination with chemotherapeutic agents. And macropinocytosis could be used as a new target for therapy and might help with transfer the drug into the cells. Despite these hypotheses, there are many other ways we could try in this field.
In conclusion, there are more studies have been carried out to reveal the mechanisms of drug resistance in gastric cancer, the related pathways and molecules above are likely to help us solve this problem in future. For drug resistance is caused by multiple factors, it makes our research more complicated and brings a challenge in clinical application. However, we should believe that with the development of our technology, the situation will be improved and the patients will get benefit.
Acknowledgments
This work was supported by grants from NSFC to Chuanqing Wu (grant 81600401), Hubei Provincial Natural Science Foundation of China to Chuanqing Wu (grant 2017CFB474), and Health Commission of Hubei Province to Chuanqing Wu (grant WJ2019Q035).
Disclosure
The authors declare no competing interests.
References
1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. doi:10.3322/caac.21262
2. Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66(2):115–132. doi:10.3322/caac.21338
3. Huang H, Yang X-J, Gao R. Research Advances in the Mechanisms of Gastric Cancer Multidrug Resistance. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2016;38(6):739–745. doi:10.3881/j.issn.1000-503X.2016.06.020
4. Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355(1):11–20. doi:10.1056/NEJMoa055531
5. Catalano V, Labianca R, Beretta GD, Gatta G, de Braud F, Van Cutsem E. Gastric cancer. Crit Rev Oncol Hematol. 2009;71(2):127–164. doi:10.1016/j.critrevonc.2009.01.004
6. Lippert TH, Ruoff HJ, Volm M. Intrinsic and acquired drug resistance in malignant tumors. The main reason for therapeutic failure. Arzneimittelforschung. 2008;58(6):261–264. doi:10.1055/s-0031-1296504
7. Wagner AD, Grothe W, Haerting J, Kleber G, Grothey A, Fleig WE. Chemotherapy in advanced gastric cancer: a systematic review and meta-analysis based on aggregate data. J Clin Oncol. 2006;24(18):2903–2909. doi:10.1200/JCO.2005.05.0245
8. Zheng Y, Zhu XQ, Ren XG. Third-line chemotherapy in advanced gastric cancer: a systematic review and meta-analysis. Medicine. 2017;96(24):e6884. doi:10.1097/MD.0000000000006884
9. Szakacs G, Jakab K, Antal F, Sarkadi B. Diagnostics of multidrug resistance in cancer. Pathol Oncol Res. 1998;4(4):251–257. doi:10.1007/BF02905214
10. Volm M, Efferth T. Prediction of cancer drug resistance and implications for personalized medicine. Front Oncol. 2015;5:282. doi:10.3389/fonc.2015.00282
11. Bunting KD. ABC transporters as phenotypic markers and functional regulators of stem cells. Stem Cells. 2002;20(1):11–20. doi:10.1002/stem.200011
12. Choi YH, Yu AM. ABC transporters in multidrug resistance and pharmacokinetics, and strategies for drug development. Curr Pharm Des. 2014;20(5):793–807. doi:10.2174/138161282005140214165212
13. Cui H, Zhang AJ, Chen M, Liu JJ. ABC transporter inhibitors in reversing multidrug resistance to chemotherapy. Curr Drug Targets. 2015;16(12):1356–1371. doi:10.2174/1389450116666150330113506
14. Iangcharoen P, Punfa W, Yodkeeree S, et al. Anti-P-glycoprotein conjugated nanoparticles for targeting drug delivery in cancer treatment. Arch Pharm Res. 2011;34(10):1679–1689. doi:10.1007/s12272-011-1012-4
15. Wang Y, Ma G, Wang Q, et al. Involvement of CUL4A in regulation of multidrug resistance to P-gp substrate drugs in breast cancer cells. Molecules. 2013;19(1):159–176. doi:10.3390/molecules19010159
16. Monden N, Abe S, Hishikawa Y, et al. The role of P-glycoprotein in human gastric cancer xenografts in response to chemotherapy. Int J Surg Investig. 1999;1(1):3–10.
17. Rocco A, Compare D, Liguori E, et al. MDR1-P-glycoprotein behaves as an oncofetal protein that promotes cell survival in gastric cancer cells. Lab Invest. 2012;92(10):1407–1418. doi:10.1038/labinvest.2012.100
18. de Oliveira J, Felipe AV, Neto RA, Oshima CT, de Souza Silva M, Forones NM. Association between ABCB1 immunohistochemical expression and overall survival in gastric cancer patients. Asian Pac J Cancer Prev. 2014;15(16):6935–6938. doi:10.7314/APJCP.2014.15.16.6935
19. Chung HC, Gong SJ, Yoo NC, et al. P-glycoprotein as an intermediate end point of drug resistance to neoadjuvant chemotherapy in locally advanced gastric cancer. Yonsei Med J. 1996;37(6):397–404. doi:10.3349/ymj.1996.37.6.397
20. Katayama K, Yoshioka S, Tsukahara S, Mitsuhashi J, Sugimoto Y. Inhibition of the mitogen-activated protein kinase pathway results in the down-regulation of P-glycoprotein. Mol Cancer Ther. 2007;6(7):2092–2102. doi:10.1158/1535-7163.MCT-07-0148
21. Bentires-Alj M, Barbu V, Fillet M, et al. NF-kappaB transcription factor induces drug resistance through MDR1 expression in cancer cells. Oncogene. 2003;22(1):90–97. doi:10.1038/sj.onc.1206056
22. Zhu ZA, Zhu ZQ, Cai HX, Liu Y. Reversion of multidrug resistance by SKI-II in SGC7901/DDP cells and exploration of underlying mechanisms. Asian Pac J Cancer Prev. 2012;13(2):625–631. doi:10.7314/APJCP.2012.13.2.625
23. Mao Z, Zhou J, Luan J, Sheng W, Shen X, Dong X. Tamoxifen reduces P-gp-mediated multidrug resistance via inhibiting the PI3K/Akt signaling pathway in ER-negative human gastric cancer cells. Biomed Pharmacother. 2014;68(2):179–183. doi:10.1016/j.biopha.2013.10.003
24. Han Z, Hong L, Han Y, et al. Phospho Akt mediates multidrug resistance of gastric cancer cells through regulation of P-gp, Bcl-2 and Bax. J Exp Clin Cancer Res. 2007;26(2):261–268.
25. Xie X, Tang B, Zhou J, Gao Q, Zhang P. Inhibition of the PI3K/Akt pathway increases the chemosensitivity of gastric cancer to vincristine. Oncol Rep. 2013;30(2):773–782. doi:10.3892/or.2013.2520
26. Schecter RL, Alaoui-Jamali MA, Batist G. Glutathione S-transferase in chemotherapy resistance and in carcinogenesis. Biochem Cell Biol. 1992;70(5):349–353. doi:10.1139/o92-054
27. Lo HW, Ali-Osman F. Genetic polymorphism and function of glutathione S-transferases in tumor drug resistance. Curr Opin Pharmacol. 2007;7(4):367–374. doi:10.1016/j.coph.2007.06.009
28. Peters WH, Wormskamp NG, Thies E. Expression of glutathione S-transferases in normal gastric mucosa and in gastric tumors. Carcinogenesis. 1990;11(9):1593–1596. doi:10.1093/carcin/11.9.1593
29. Kodera Y, Isobe K, Yamauchi M, et al. Expression of glutathione-S-transferases alpha and pi in gastric cancer: a correlation with cisplatin resistance. Cancer Chemother Pharmacol. 1994;34(3):203–208. doi:10.1007/BF00685078
30. Schipper DL, Wagenmans MJ, Peters WH, Wils JA, Wagener DJ. Glutathione S-transferases and iododeoxyuridine labelling index during chemotherapy of gastric cancer. Anticancer Res. 2000;20(3a):1705–1710.
31. Kim R, Ohi Y, Inoue H, Toge T. Expression and relationship between topoisomerase I and II alpha genes in tumor and normal tissues in esophageal, gastric and colon cancers. Anticancer Res. 1999;19(6b):5393–5398.
32. Wang G, Huang H, Gao J, et al. Tissue microarray analysis of topoisomerase IIalpha protein in gastric adenocarcinomas: histogenetic and prognostic implications. Cancer Genomics Proteomics. 2011;8(3):127–133.
33. Yu P, Du Y, Yang L, Fan S, Wu J, Zheng S. Significance of multidrug resistance gene-related proteins in the postoperative chemotherapy of gastric cancer. Int J Clin Exp Pathol. 2014;7(11):7945–7950.
34. Geng M, Wang L, Chen X, Cao R, Li P. The association between chemosensitivity and Pgp, GST-pi and Topo II expression in gastric cancer. Diagn Pathol. 2013;8:198. doi:10.1186/1746-1596-8-198
35. Son YS, Suh JM, Ahn SH, et al. Reduced activity of topoisomerase II in an Adriamycin-resistant human stomach-adenocarcinoma cell line. Cancer Chemother Pharmacol. 1998;41(5):353–360. doi:10.1007/s002800050751
36. Zugazagoitia J, Guedes C, Ponce S, Ferrer I, Molina-Pinelo S, Paz-Ares L. Current challenges in cancer treatment. Clin Ther. 2016;38(7):1551–1566. doi:10.1016/j.clinthera.2016.03.026
37. Yang W, Raufi A, Klempner SJ. Targeted therapy for gastric cancer: molecular pathways and ongoing investigations. Biochim Biophys Acta. 2014;1846(1):232–237. doi:10.1016/j.bbcan.2014.05.003
38. Zheng S, Huang KE, Pan YL, et al. KIT and BRAF heterogeneous mutations in gastrointestinal stromal tumors after secondary imatinib resistance. Gastric Cancer. 2015;18(4):796–802. doi:10.1007/s10120-014-0414-7
39. Serrano C, Wang Y, Marino-Enriquez A, et al. KRAS and KIT gatekeeper mutations confer polyclonal primary imatinib resistance in gi stromal tumors: relevance of concomitant phosphatidylinositol 3-kinase/AKT dysregulation. J Clin Oncol. 2015;33(22):e93–e96. doi:10.1200/JCO.2013.48.7488
40. Sanchez-Olea R, Calera MR, Degterev A. Molecular pathways involved in cell death after chemically induced DNA damage. Exs. 2009;99:209–230. doi:10.1007/978-3-7643-8336-7_8
41. Boland CR. Roles of the DNA mismatch repair genes in colorectal tumorigenesis. Int J Cancer. 1996;69(1):47–49. doi:10.1002/(ISSN)1097-0215
42. Lin CT, Lyu YL, Xiao H, Lin WH, Whang-Peng J. Suppression of gene amplification and chromosomal DNA integration by the DNA mismatch repair system. Nucleic Acids Res. 2001;29(16):3304–3310. doi:10.1093/nar/29.16.3304
43. Tanida S, Mizoshita T, Ozeki K, et al. Mechanisms of cisplatin-induced apoptosis and of cisplatin sensitivity: potential of BIN1 to act as a potent predictor of cisplatin sensitivity in gastric cancer treatment. Int J Surg Oncol. 2012;2012:862879. doi:10.1155/2012/862879
44. Marteijn JA, Lans H, Vermeulen W, Hoeijmakers JH. Understanding nucleotide excision repair and its roles in cancer and ageing. Nat Rev Mol Cell Biol. 2014;15(7):465–481. doi:10.1038/nrm3822
45. Liu J, He C, Xing C, Yuan Y. Nucleotide excision repair related gene polymorphisms and genetic susceptibility, chemotherapeutic sensitivity and prognosis of gastric cancer. Mutat Res. 2014;765:11–21. doi:10.1016/j.mrfmmm.2014.04.008
46. Simonelli V, Leuzzi G, Basile G, et al. Crosstalk between mismatch repair and base excision repair in human gastric cancer. Oncotarget. 2017;8(49):84827–84840. doi:10.18632/oncotarget.v8i49
47. Longley DB, Johnston PG. Molecular mechanisms of drug resistance. J Pathol. 2005;205(2):275–292. doi:10.1002/(ISSN)1096-9896
48. Xia H, Hui KM. Mechanism of cancer drug resistance and the involvement of noncoding RNAs. Curr Med Chem. 2014;21(26):3029–3041. doi:10.2174/0929867321666140414101939
49. Geng M, Wang L, Li P. Correlation between chemosensitivity to anticancer drugs and Bcl-2 expression in gastric cancer. Int J Clin Exp Pathol. 2013;6(11):2554–2559.
50. Cheng H, Wang X, Li T, Chen L. Bcl-2 expression and patient survival in gastric cancer: a systematic review of the literature with meta-analysis. Med Oncol. 2015;32(1):389. doi:10.1007/s12032-014-0389-6
51. Yu DF, Wu FR, Liu Y, Liu H, Xia Q. Bcl-2 gene silence enhances the sensitivity toward 5-Fluorouracil in gastric adenocarcinoma cells. Biomed Pharmacother. 2013;67(7):615–619. doi:10.1016/j.biopha.2013.03.007
52. Jeong SH, Han JH, Kim JH, et al. Bax predicts outcome in gastric cancer patients treated with 5-fluorouracil, leucovorin, and oxaliplatin palliative chemotherapy. Dig Dis Sci. 2011;56(1):131–138. doi:10.1007/s10620-010-1280-8
53. Sawa H, Kobayashi T, Mukai K, Zhang W, Shiku H. Bax overexpression enhances cytochrome c release from mitochondria and sensitizes KATOIII gastric cancer cells to chemotherapeutic agent-induced apoptosis. Int J Oncol. 2000;16(4):745–749. doi:10.3892/ijo.16.4.745
54. Tahara M, Ochiai A, Fujimoto J, et al. Expression of thymidylate synthase, thymidine phosphorylase, dihydropyrimidine dehydrogenase, E2F-1, Bak, Bcl-X, and Bcl-2, and clinical outcomes for gastric cancer patients treated with bolus 5-fluorouracil. Oncol Rep. 2004;11(1):9–15.
55. Tsujitani S, Saito H, Wakatsuki T, et al. Relationship between expression of apoptosis-related proteins and the efficacy of postoperative chemotherapy in patients with T3 gastric cancer. Surg Today. 2012;42(3):225–232. doi:10.1007/s00595-011-0062-z
56. Villamizar O, Chambers CB, Riberdy JM, Persons DA, Wilber A. Long noncoding RNA Saf and splicing factor 45 increase soluble Fas and resistance to apoptosis. Oncotarget. 2016;7(12):13810–13826. doi:10.18632/oncotarget.v7i12
57. Rippa E, La Monica G, Allocca R, Romano MF, De Palma M, Arcari P. Overexpression of gastrokine 1 in gastric cancer cells induces Fas-mediated apoptosis. J Cell Physiol. 2011;226(10):2571–2578. doi:10.1002/jcp.v226.10
58. Souza PS, Madigan JP, Gillet JP, et al. Expression of the multidrug transporter P-glycoprotein is inversely related to that of apoptosis-associated endogenous TRAIL. Exp Cell Res. 2015;336(2):318–328. doi:10.1016/j.yexcr.2015.06.005
59. Matozaki T, Sakamoto C, Suzuki T, et al. p53 gene mutations in human gastric cancer: wild-type p53 but not mutant p53 suppresses growth of human gastric cancer cells. Cancer Res. 1992;52(16):4335–4341.
60. Yildirim M, Kaya V, Demirpence O, Gunduz S, Bozcuk H. Prognostic significance of p53 in gastric cancer: a meta- analysis. Asian Pac J Cancer Prev. 2015;16(1):327–332. doi:10.7314/APJCP.2015.16.1.327
61. Xu HY, Xu WL, Wang LQ, Chen MB, Shen HL. Relationship between p53 status and response to chemotherapy in patients with gastric cancer: a meta-analysis. PLoS One. 2014;9(4):e95371. doi:10.1371/journal.pone.0095371
62. Chen GX, Zheng LH, Liu SY, He XH. rAd-p53 enhances the sensitivity of human gastric cancer cells to chemotherapy. World J Gastroenterol. 2011;17(38):4289–4297. doi:10.3748/wjg.v17.i38.4289
63. Kim CW, Lu JN, Go SI, et al. p53 restoration can overcome cisplatin resistance through inhibition of Akt as well as induction of Bax. Int J Oncol. 2013;43(5):1495–1502. doi:10.3892/ijo.2013.2070
64. Tang L, Long Z, Feng G, Guo X, Yu M. NES1/KLK10 promotes trastuzumab resistance via activation of PI3K/AKT signaling pathway in gastric cancer. J Cell Biochem. 2017.
65. Liang J, Ge F, Guo C, et al. Inhibition of PI3K/Akt partially leads to the inhibition of PrP(C)-induced drug resistance in gastric cancer cells. FEBS J. 2009;276(3):685–694. doi:10.1111/j.1742-4658.2008.06816.x
66. Liu SQ, Yu JP, Yu HG, Lv P, Chen HL. Activation of Akt and ERK signalling pathways induced by etoposide confer chemoresistance in gastric cancer cells. Dig Liver Dis. 2006;38(5):310–318. doi:10.1016/j.dld.2006.01.012
67. Yu HG, Ai YW, Yu LL, et al. Phosphoinositide 3-kinase/Akt pathway plays an important role in chemoresistance of gastric cancer cells against etoposide and doxorubicin induced cell death. Int J Cancer. 2008;122(2):433–443. doi:10.1002/ijc.23049
68. Harada K, Baba Y, Shigaki H, et al. Prognostic and clinical impact of PIK3CA mutation in gastric cancer: pyrosequencing technology and literature review. BMC Cancer. 2016;16:400. doi:10.1186/s12885-016-2422-y
69. Oki E, Baba H, Tokunaga E, et al. Akt phosphorylation associates with LOH of PTEN and leads to chemoresistance for gastric cancer. Int J Cancer. 2005;117(3):376–380. doi:10.1002/(ISSN)1097-0215
70. Yu LL, Dai N, Yu HG, Sun LM, Si JM. Akt associates with nuclear factor kappaB and plays an important role in chemoresistance of gastric cancer cells. Oncol Rep. 2010;24(1):113–119. doi:10.3892/or_00000835
71. Almhanna K, Cubitt CL, Zhang S, et al. MK-2206, an Akt inhibitor, enhances carboplatinum/paclitaxel efficacy in gastric cancer cell lines. Cancer Biol Ther. 2013;14(10):932–936. doi:10.4161/cbt.25939
72. Chen X, Dai X, Zou P, et al. Curcuminoid EF24 enhances the anti-tumour activity of Akt inhibitor MK-2206 through ROS-mediated endoplasmic reticulum stress and mitochondrial dysfunction in gastric cancer. Br J Pharmacol. 2017;174(10):1131–1146. doi:10.1111/bph.13765
73. Smith AG, Macleod KF. Autophagy, cancer stem cells and drug resistance. J Pathol. 2019;247(5):708–718. doi:10.1002/path.2019.247.issue-5
74. Li Y-J, Lei Y-H, Yao N, et al. Autophagy and multidrug resistance in cancer. Chin J Cancer. 2017;36(1):52. doi:10.1186/s40880-017-0219-2
75. Levine B, Kroemer G. Autophagy in the pathogenesis of disease. Cell. 2008;132(1):27–42. doi:10.1016/j.cell.2007.12.018
76. Mizushima N, Yoshimori T, Ohsumi Y. The role of Atg proteins in autophagosome formation. Annu Rev Cell Dev Biol. 2011;27:107–132. doi:10.1146/annurev-cellbio-092910-154005
77. Yoshida GJ. Therapeutic strategies of drug repositioning targeting autophagy to induce cancer cell death: from pathophysiology to treatment. J Hematol Oncol. 2017;10(1):67. doi:10.1186/s13045-017-0436-9
78. White E, DiPaola RS. The double-edged sword of autophagy modulation in cancer. Clin Cancer Res. 2009;15(17):5308–5316. doi:10.1158/1078-0432.CCR-07-5023
79. Kondo Y, Kanzawa T, Sawaya R, Kondo S. The role of autophagy in cancer development and response to therapy. Nat Rev Cancer. 2005;5(9):726–734. doi:10.1038/nrc1692
80. Amaravadi RK, Lippincott-Schwartz J, Yin X-M, et al. Principles and current strategies for targeting autophagy for cancer treatment. Clin Cancer Res. 2011;17(4):654–666. doi:10.1158/1078-0432.CCR-10-2634
81. Cao Y, Luo Y, Zou J, et al. Autophagy and its role in gastric cancer. Clin Chim Acta. 2019;489:10–20. doi:10.1016/j.cca.2018.11.028
82. Qian H-R, Yang Y. Functional role of autophagy in gastric cancer. Oncotarget. 2016;7(14):17641–17651. doi:10.18632/oncotarget.v7i14
83. Al-Akra L, Bae D-H, Leck LYW, Richardson DR, Jansson PJ. The biochemical and molecular mechanisms involved in the role of tumor micro-environment stress in development of drug resistance. Biochim Biophys Acta Gen Subj. 2019;1863(9):1390–1397. doi:10.1016/j.bbagen.2019.06.007
84. Crighton D, Wilkinson S, O’Prey J, et al. DRAM, a p53-induced modulator of autophagy, is critical for apoptosis. Cell. 2006;126(1):121–134. doi:10.1016/j.cell.2006.05.034
85. Simon H-U, Friis R, Tait SWG, Ryan KM. Retrograde signaling from autophagy modulates stress responses. Sci Signal. 2017;10(468):eaag2791. doi:10.1126/scisignal.aag2791
86. Levy JMM, Towers CG, Thorburn A. Targeting autophagy in cancer. Nat Rev Cancer. 2017;17(9):528–542. doi:10.1038/nrc.2017.53
87. Pascolo S. Time to use a dose of Chloroquine as an adjuvant to anti-cancer chemotherapies. Eur J Pharmacol. 2016;771:139–144. doi:10.1016/j.ejphar.2015.12.017
88. Elliott T, Sethi T. Integrins and extracellular matrix: a novel mechanism of multidrug resistance. Expert Rev Anticancer Ther. 2002;2(4):449–459. doi:10.1586/14737140.2.4.449
89. Gilkes DM, Semenza GL, Wirtz D. Hypoxia and the extracellular matrix: drivers of tumour metastasis. Nat Rev Cancer. 2014;14(6):430–439. doi:10.1038/nrc3726
90. Pickup MW, Mouw JK, Weaver VM. The extracellular matrix modulates the hallmarks of cancer. EMBO Rep. 2014;15(12):1243–1253. doi:10.15252/embr.201439246
91. Nakamura K, Mori M, Enjoji M. Distribution of basement membrane antigens in clinical gastric adenocarcinomas: an immunohistochemical study. J Clin Pathol. 1987;40(12):1418–1423. doi:10.1136/jcp.40.12.1418
92. Sun L, Liu L, Liu X, et al. Gastric cancer cell adhesion to laminin enhances acquired chemotherapeutic drug resistance mediated by MGr1-Ag/37LRP. Oncol Rep. 2014;32(1):105–114. doi:10.3892/or.2014.3184
93. Sun L, Liu L, Liu X, et al. MGr1-Ag/37LRP induces cell adhesion-mediated drug resistance through FAK/PI3K and MAPK pathway in gastric cancer. Cancer Sci. 2014;105(6):651–659. doi:10.1111/cas.12414
94. Yang JM, Xu Z, Wu H, Zhu H, Wu X, Hait WN. Overexpression of extracellular matrix metalloproteinase inducer in multidrug resistant cancer cells. Mol Cancer Res. 2003;1(6):420–427.
95. Yin Y, Li W, Deng M, et al. Extracellular high mobility group box chromosomal protein 1 promotes drug resistance by increasing the expression of Pglycoprotein expression in gastric adenocarcinoma cells. Mol Med Rep. 2014;9(4):1439–1443. doi:10.3892/mmr.2014.1961
96. Ock CY, Nam AR, Bang JH, et al. Signature of cytokines and angiogenic factors (CAFs) defines a clinically distinct subgroup of gastric cancer. Gastric Cancer. 2017;20(1):164–174. doi:10.1007/s10120-015-0583-z
97. Yang Z, Guo L, Liu D, et al. Acquisition of resistance to trastuzumab in gastric cancer cells is associated with activation of IL-6/STAT3/Jagged-1/Notch positive feedback loop. Oncotarget. 2015;6(7):5072–5087. doi:10.18632/oncotarget.3241
98. Ye XL, Zhao YR, Weng GB, et al. IL-33-induced JNK pathway activation confers gastric cancer chemotherapy resistance. Oncol Rep. 2015;33(6):2746–2752. doi:10.3892/or.2015.3898
99. Mao Z, Bian G, Sheng W, He S, Yang J, Dong X. Adenovirus-mediated IL-24 expression enhances the chemosensitivity of multidrug-resistantgastric cancer cells to cisplatin. Oncol Rep. 2013;30(5):2288–2296. doi:10.3892/or.2013.2695
100. Chen L, Shi Y, Yuan J, et al. HIF-1 alpha overexpression correlates with poor overall survival and disease-free survival in gastric cancer patients post-gastrectomy. PLoS One. 2014;9(3):e90678. doi:10.1371/journal.pone.0090678
101. Isobe T, Aoyagi K, Koufuji K, et al. Clinicopathological significance of hypoxia-inducible factor-1 alpha (HIF-1alpha) expression in gastric cancer. Int J Clin Oncol. 2013;18(2):293–304.
102. Zhu CL, Huang Q, Liu CH, Lin XS, Xie F. Prognostic value of HIF-1alpha expression in patients with gastric cancer. Mol Biol Rep. 2013;40(11):6055–6062. doi:10.1007/s11033-013-2715-z
103. Liu L, Ning X, Sun L, et al. Hypoxia-inducible factor-1 alpha contributes to hypoxia-induced chemoresistance in gastric cancer. Cancer Sci. 2008;99(1):121–128. doi:10.1111/j.1349-7006.2007.00643.x
104. Rohwer N, Dame C, Haugstetter A, et al. Hypoxia-inducible factor 1alpha determines gastric cancer chemosensitivity via modulation of p53 and NF-kappaB. PLoS One. 2010;5(8):e12038. doi:10.1371/journal.pone.0012038
105. Sun XP, Dong X, Lin L, et al. Up-regulation of survivin by AKT and hypoxia-inducible factor 1alpha contributes to cisplatin resistance in gastric cancer. FEBS J. 2014;281(1):115–128. doi:10.1111/febs.12577
106. Liu L, Zhang H, Sun L, et al. ERK/MAPK activation involves hypoxia-induced MGr1-Ag/37LRP expression and contributes to apoptosis resistance in gastric cancer. Int J Cancer. 2010;127(4):820–829. doi:10.1002/ijc.25098
107. Huang T, Song C, Zheng L, Xia L, Li Y, Zhou Y. The roles of extracellular vesicles in gastric cancer development, microenvironment, anti-cancer drug resistance, and therapy. Mol Cancer. 2019;18(1):62. doi:10.1186/s12943-019-0967-5
108. Schorey JS, Bhatnagar S. Exosome function: from tumor immunology to pathogen biology. Traffic. 2008;9(6):871–881. doi:10.1111/j.1600-0854.2008.00734.x
109. Skog J, Wurdinger T, van Rijn S, et al. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat Cell Biol. 2008;10(12):1470–1476. doi:10.1038/ncb1800
110. Wang M, Qiu R, Yu S, et al. Paclitaxelresistant gastric cancer MGC803 cells promote epithelialtomesenchymal transition and chemoresistance in paclitaxelsensitive cells via exosomal delivery of miR1555p. Int J Oncol. 2019;54(1):326–338. doi:10.3892/ijo.2018.4601
111. Ji R, Zhang B, Zhang X, et al. Exosomes derived from human mesenchymal stem cells confer drug resistance in gastric cancer. Cell Cycle. 2015;14(15):2473–2483. doi:10.1080/15384101.2015.1005530
112. Zheng P, Chen L, Yuan X, et al. Exosomal transfer of tumor-associated macrophage-derived miR-21 confers cisplatin resistance in gastric cancer cells. J Exp Clin Cancer Res. 2017;36(1):53. doi:10.1186/s13046-017-0528-y
113. Ha KD, Bidlingmaier SM, Liu B. Macropinocytosis exploitation by cancers and cancer therapeutics. Front Physiol. 2016;7:381.
114. Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science (New York, NY). 2009;324(5930):1029–1033. doi:10.1126/science.1160809
115. Pavlova NN, Thompson CB. The emerging hallmarks of cancer metabolism. Cell Metab. 2016;23(1):27–47. doi:10.1016/j.cmet.2015.12.006
116. Qian Y, Wang X, Liu Y, et al. Extracellular ATP is internalized by macropinocytosis and induces intracellular ATP increase and drug resistance in cancer cells. Cancer Lett. 2014;351(2):242–251. doi:10.1016/j.canlet.2014.06.008
117. Cullis J, Siolas D, Avanzi A, Barui S, Maitra A, Bar-Sagi D. Macropinocytosis of Nab-paclitaxel drives macrophage activation in pancreatic cancer. Cancer Immunol Res. 2017;5(3):182–190. doi:10.1158/2326-6066.CIR-16-0125
118. Werfel TA, Wang S, Jackson MA, et al. Selective mTORC2 inhibitor therapeutically blocks breast cancer cell growth and survival. Cancer Res. 2018;78(7):1845–1858. doi:10.1158/0008-5472.CAN-17-2388
119. Di Leva G, Garofalo M, Croce CM. MicroRNAs in cancer. Annu Rev Pathol. 2014;9:287–314. doi:10.1146/annurev-pathol-012513-104715
120. Li PF, Chen SC, Xia T, et al. Non-coding RNAs and gastric cancer. World J Gastroenterol. 2014;20(18):5411–5419. doi:10.3748/wjg.v20.i18.5411
121. Gu Y, Chen T, Li G, et al. LncRNAs: emerging biomarkers in gastric cancer. Future Oncol. 2015;11(17):2427–2441. doi:10.2217/fon.15.175
122. Zheng T, Wang J, Chen X, Liu L. Role of microRNA in anticancer drug resistance. Int J Cancer. 2010;126(1):2–10. doi:10.1002/ijc.v126:1
123. Wang T, Ge G, Ding Y, et al. MiR-503 regulates cisplatin resistance of human gastric cancer cell lines by targeting IGF1R and BCL2. Chin Med J. 2014;127(12):2357–2362.
124. Zhuang M, Shi Q, Zhang X, et al. Involvement of miR-143 in cisplatin resistance of gastric cancer cells via targeting IGF1R and BCL2. Tumour Biol. 2015;36(4):2737–2745. doi:10.1007/s13277-014-2898-5
125. Zhu W, Xu H, Zhu D, et al. miR-200bc/429 cluster modulates multidrug resistance of human cancer cell lines by targeting BCL2 and XIAP. Cancer Chemother Pharmacol. 2012;69(3):723–731. doi:10.1007/s00280-011-1752-3
126. Yang SM, Huang C, Li XF, Yu MZ, He Y, Li J. miR-21 confers cisplatin resistance in gastric cancer cells by regulating PTEN. Toxicology. 2013;306:162–168. doi:10.1016/j.tox.2013.02.014
127. Eto K, Iwatsuki M, Watanabe M, et al. The microRNA-21/PTEN pathway regulates the sensitivity of HER2-positive gastric cancer cells to trastuzumab. Ann Surg Oncol. 2014;21(1):343–350. doi:10.1245/s10434-013-3325-7
128. Hu N, Yin JF, Ji Z, et al. Strengthening gastric cancer therapy by trastuzumab-conjugated nanoparticles with simultaneous encapsulation of anti-MiR-21 and 5-fluorouridine. Cell Physiol Biochem. 2017;44(6):2158–2173. doi:10.1159/000485955
129. Wu XM, Shao XQ, Meng XX, et al. Genome-wide analysis of microRNA and mRNA expression signatures in hydroxycamptothecin-resistant gastric cancer cells. Acta Pharmacol Sin. 2011;32(2):259–269. doi:10.1038/aps.2010.204
130. Kim CH, Kim HK, Rettig RL, et al. miRNA signature associated with outcome of gastric cancer patients following chemotherapy. BMC Med Genomics. 2011;4:79. doi:10.1186/1755-8794-4-79
131. Ding J, Li D, Gong M, et al. Expression and clinical significance of the long non-coding RNA PVT1 in human gastric cancer. Onco Targets Ther. 2014;7:1625–1630. doi:10.2147/OTT
132. Zhou DD, Liu XF, Lu CW, Pant OP, Liu XD. Long non-coding RNA PVT1: emerging biomarker in digestive system cancer. Cell Prolif. 2017;50(6):e12398. doi:10.1111/cpr.12398
133. Zhang XW, Bu P, Liu L, Zhang XZ, Li J. Overexpression of long non-coding RNA PVT1 in gastric cancer cells promotes the development of multidrug resistance. Biochem Biophys Res Commun. 2015;462(3):227–232. doi:10.1016/j.bbrc.2015.04.121
134. Zhang XW, Liu L, Zhang XZ, Bo P. Kanglaite inhibits the expression of drug resistance genes through suppressing PVT1 in cisplatin-resistant gastric cancer cells. Exp Ther Med. 2017;14(2):1789–1794. doi:10.3892/etm.2017.4650
135. Han Y, Ye J, Wu D, et al. LEIGC long non-coding RNA acts as a tumor suppressor in gastric carcinoma by inhibiting the epithelial-to-mesenchymal transition. BMC Cancer. 2014;14:932. doi:10.1186/1471-2407-14-932
136. Wang Y, Zhang D, Wu K, Zhao Q, Nie Y, Fan D. Long noncoding RNA MRUL promotes ABCB1 expression in multidrug-resistant gastric cancer cell sublines. Mol Cell Biol. 2014;34(17):3182–3193. doi:10.1128/MCB.01580-13
137. Hang Q, Sun R, Jiang C, Li Y. Notch 1 promotes cisplatin-resistant gastric cancer formation by upregulating lncRNA AK022798 expression. Anticancer Drugs. 2015;26(6):632–640. doi:10.1097/CAD.0000000000000227
138. Yan J, Dang Y, Liu S, Zhang Y, Zhang G. LncRNA HOTAIR promotes cisplatin resistance in gastric cancer by targeting miR-126 to activate the PI3K/AKT/MRP1 genes. Tumour Biol. 2016;37:16345–16355. doi:10.1007/s13277-016-5448-5
139. Fang Q, Chen X, Zhi X. Long non-coding RNA (LncRNA) urothelial carcinoma associated 1 (UCA1) increases multi-drug resistance of gastric cancer via downregulating miR-27b. Med Sci Monit. 2016;22:3506–3513. doi:10.12659/MSM.900688
140. Tang HN, Hu JH. A comparison of surgical procedures and postoperative cares for minimally invasive laparoscopic gastrectomy and open gastrectomy in gastric cancer. Int J Clin Exp Med. 2015;8(7):10321–10329.
141. Song Z, Wu Y, Yang J, Yang D, Fang X. Progress in the treatment of advanced gastric cancer. Tumour Biol. 2017;39(7):1010428317714626. doi:10.1177/1010428317714626
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
