Back to Journals » Therapeutics and Clinical Risk Management » Volume 14
A preliminary evaluation of serum level of testosterone, LH, and FSH in patients with varicocele after varicocelectomy as a kidney-related disease
Authors Alizadeh M, Nasebakht A, Valizadeh R, Mohammadi Fallah M , Taghizadeh Afshari A, Rahimi MM, Daneshyar C
Received 5 January 2018
Accepted for publication 27 May 2018
Published 4 September 2018 Volume 2018:14 Pages 1585—1590
DOI https://doi.org/10.2147/TCRM.S161641
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Garry Walsh
Mansour Alizadeh,1 Ali Nasebakht,1 Rohollah Valizadeh,2,3 Mohammadreza Mohammadi Fallah,1 Ali Taghizadeh Afshari,1 Mohsen Mohammad Rahimi,4 Chimen Daneshyar5
1Nephrology and Kidney Transplant Research Center, Urmia University of Medical Sciences, Urmia, Iran; 2Student Research Committee, Urmia University of Medical Sciences, Urmia, Iran; 3Student Research Committee, Department of Epidemiology, Iran University of Medical Sciences, Tehran, Iran; 4Department of Kidney Transplantation, Nephrology and Kidney Transplant Research Center, Urmia University of Medical Sciences, Urmia, Iran; 5Faculty of Medicine, Urmia University of Medical Sciences, Urmia, Iran
Introduction: Varicocele is a common problem with a high prevalence in population with primary and secondary infertilities. The adverse effects of varicocele on spermatogenesis and fertility are known, but the association between clinical varicocele and testosterone is not clear. Hence, we decided to evaluate the serum levels of testosterone, luteinizing hormone (LH) and follicle-stimulating hormone (FSH) in patients with varicocele after varicocelectomy.
Methods: In this study, 100 patients with varicocele were divided into two groups: hypogonadal patients with testosterone level <280 ng/dL and eugonadal patients with testosterone level >280 ng/dL. The serum levels of testosterone, FSH, and LH were measured before surgery and 3 months after surgery, and the results were analyzed using the SPSS software. P-value <0.05 was considered statistically significant.
Results: Patients with varicocele after puberty till 50 years were divided into two groups: hypogonadal (testosterone <280 ng/dL) and eugonadal (testosterone >280 ng/dL) patients who required varicocelectomy. The mean testosterone level before surgery in hypogonadal patients was 215.22±83.31 ng/dL, which reached 326.95±35.125 ng/dL after surgery (P<0.0001), which was significant. There was no significant decrease in the mean FSH level, but there was a significant decrease in the mean LH level after varicocelectomy. In eugonadal group, testosterone level before surgery was 471.90±145.71 ng/dL, which reached 469.57±145.61 ng/dL after surgery, which was not significant.
Conclusion: In our study, patients who underwent varicocelectomy had improved testosterone levels, so that this increase was more significant in hypogonadal patients than in eugonadal patients. Decrease in LH and FSH levels in all patients was seen after varicocelectomy, which can be due to increase in testosterone levels.
Keywords: hypogonadal, eugonadal, varicocele, testosterone, FSH, LH
Introduction
Varicocele is the dilation of veins in the pampiniform plexus and its turbulent blood flow.1 It is a common problem with 15% prevalence in general population, 35% prevalence in people with primary infertility, and 81% prevalence in people with secondary infertility.2 The adverse effects of varicocele on spermatogenesis and fertility are known, but the relation between clinical varicocele and impaired testosterone production is not clearly known and some believe that the degree of varicocele is more likely to have a greater negative effect on testosterone production.3,4
Although the effects of varicocele on Leydig cells and usefulness of varicocelectomy in improving the performance of Leydig cells have been imagined for decades, only a few studies have been conducted in this field, and they have often been retrospective, and therefore have limited value compared to other study designs, such as prospective studies, and also have contradictory results.5,6 Some researchers have raised a hypothesis completely inconsistent with the aforementioned content due to vascular vasodilatation effects of testosterone that high serum levels of testosterone may cause dilatation of the spermatic vein and the creation of a varicocele.7
The pathology of varicocele is not clearly known, and the following hypotheses are proposed: renal and suprarenal reflux, oxidative stress and hypoxia, autoimmune hypothesis, hormonal dysfunction, and temperature increase. In addition to the vital importance of testosterone in sexual function and fertility, testosterone is participating in other activities such as message transmission in nerves, smooth muscle relaxation, and neuroplasticity.8,9
Varicocele can lead to a decrease in the level of testosterone through non-natural increase of testicular temperature and disruption of the testicular cooling system. Varicocele can also decrease testosterone levels through oxidative stress and impairment in the synthesis of androgens.10,11
Testosterone deficiency (<280 g/dL) leads to the following symptoms: decrease in sexual function, erection and ejaculation dysfunction, depression, sleep disorder, susceptibility to metabolic syndrome, anemia, increase in leptin, and vasomotor disorder, which cause a lot of harm to patients and require testosterone replacement, which has the following side effects, despite all the benefits: fluid retention, hepatic toxicity and hepatocellular carcinoma, breast and prostate cancer progression, sleep apnea, gynecomastia, and polycythemia.12 Several studies have been carried out in the light of the harmful and negative effects of lowering the level of testosterone on different parts of the human body, but given the severity effect of varicocelectomy, they do not have certain results about varicocelectomy and its effect on the serum levels of testosterone.6,13–16
Also, some studies conducted in Iran have uncertainty, as Jangkhah et al17 in 2014 showed the obvious effects of varicocelectomy on serum testosterone, but these authors have mentioned that there is no significant difference of testosterone level between groups in a study in 2013.18 Regarding the controversial results reported from the severity effects of varicocele on the level of reproductive hormones and the vital importance of this problem, the present study was conducted to evaluate changes in serum levels of testosterone, follicle-stimulating hormone (FSH), and luteinizing hormone (LH) after varicocelectomy. The results of this study may be helpful in the treatment of patients with varicocele and testosterone deficiency syndrome.
Methods
A total of 100 men with varicocele after puberty who need varicocelectomy were involved in the study, based on the similar study done in Iran by Jangkhah et al.19
Grading was done by physical examination, so that if the varicocele veins were visible without Valsalva maneuver, it was considered as grade III, if the varicocele veins were palpable and visible with Valsalva maneuver in normal mode, it was considered as grade II, and if the varicocele veins were palpable only with Valsalva maneuver, it was considered as grade I. All grades of varicocele were included in this study. Exclusion criteria were as follows: history of inguinoscrotal surgery, history of undescended testicle (UDT), trauma, or testicular torsion, endocrine disease (thyroid disorders, pituitary and so on), chronic hepatic disease and chronic renal diseases (changes in tesosterone levels linked to sex hormone-binding globulin [SHBG] changes), recurrent clinical varicocele, azoospermia, and receiving exogenous androgen, clomiphene, or aromatase inhibitors.
All patients in Imam Khomeini Hospital of Urmia, Iran, three blood samples ~2 mL were collected by an experienced technician at intervals 15–20 minutes. Then, the collected serums were mixed and frozen at −20°C. The samples were taken by a qualified technician at the Imam Khomeini Hospital Laboratory.
Testosterone, LH, and FSH were measured for all cases. The measurement of testosterone level was performed at 8–11 am in all cases. The measurement of LH and FSH hormones was done using the Iranian kit (Pishtaz Teb Diagnostics, Tehran, Iran) by means of ELISA method and the measurement of testosterone was done using the American kit (Monobond Plus; Ivoclar Vivadent, Schaan, Liechtenstein) by means of ELISA method. Based on the answers, patients were divided into two groups of 50 persons including hypogonadal patients with testosterone <280 ng/mL and eugonadal patients with testosterone >280 ng/dL.
In the next step, all patients under spinal anesthesia and in supine position underwent microscopic inguinal varicocelectomy surgery by a surgeon. Thus, after inguinal incision, spermatic cord was elevated using microscope and dilated veins of loop cord were closed. After 3 months of surgery, again the levels of testosterone, LH, and FSH were measured and recorded using the same way and same previous kits.
Regarding the consistency of the testicles that were evaluated during physical examination, the normal consistency of the testicle was defined as firm rubbery testicle, the softer consistency of the testicular was defined as a firm testicle, and the harder consistency of the testicular was defined as a hard testicle, based on the Campbell-Walsh Urology definitions.22
This study was approved by the ethics committee of Urmia University of Medical Science (ID code: 1393-04-40-1470), the written informed consent was obtained from patients for their data to be used in the study, the objectives of the study were explained to all participants, and all of them accepted to participate and were assured of the confidentiality of their individual information as well as the voluntary nature of participating in the study.
Results
A total of 100 patients with varicocele after puberty who required varicocelectomy were enrolled in the study and were divided into two groups with 50 patients each. The average age of hypogonadal patients was 28.68±8.37 years and that of eugonadal patients was 28.84±7.89 years. The mean body mass index of hypogonadal patients was 24.54±3.84 kg/m2 and that of eugonadal patients was 24.50±5.42. The mean testosterone level in hypogonadal patients before surgery was 215.22±83.31 ng/dL and after varicocelectomy was 326.95±35.125 ng/dL. Based on paired-samples statistics examination, there was a significant difference between testosterone levels before and after varicocelectomy surgery in hypogonadal patients.
The mean FSH level in hypogonadal patients before varicocelectomy was 4.09±2.12 mIU/mL and after varicocelectomy was 3.91±1.73 mIU/mL. Based on paired-samples statistics examination, there was no significant difference between FSH levels before and after varicocelectomy surgery in hypogonadal patients. The mean LH level in hypogonadal patients before surgery was 4.09±2.47 IU/L and after varicocelectomy was 3.80±2.21 IU/L. Based on paired-samples statistics examination, there was a significant difference between LH level before and after varicocelectomy surgery in hypogonadal patients. The mean testosterone level in eugonadal patients before surgery was 471.90±145.71 ng/dL and after varicocelectomy was 496.57±145.61 ng/dL. Based on paired-samples statistics examination, there was no significant difference between testosterone levels before and after varicocelectomy surgery in eugonadal patients. The average FSH level in eugonal patients before surgery was 4.92±3.14 mIU/mL and after varicocelectomy was 4.36±2.39 mIU/mL. Based on paired-samples statistics examination, there was no significant difference between FSH levels before and after varicocelectomy in eugonal patients (Table 1).
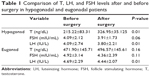  | Table 1 Comparison of T, LH, and FSH levels after and before surgery in hypogonadal and eugonadal patients |
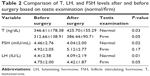
In this study, 91 patients had a normal testis and nine patients had a firm testis during physical examination. The average testosterone level in patients with normal testis before surgery was 346.61±178.38 ng/dL and after surgery was 425.70±155.29 ng/dL. The average testosterone level in patients with firm testis before surgery was 312.66±138.91 ng/dL and after surgery was 386.44±90.71 ng/dL. Based on paired-samples statistics examination, there is significant difference between testosterone levels before and after surgery in patients with firm testis (P=0.051). The average FSH level in patients with normal testis before surgery was 4.46±2.67 mIU/mL, and after surgery it was 4.04±2 mIU/mL. The average FSH level in patients with firm testis before surgery was 4.92±2.05 mIU/mL and after surgery was 5.15±2.27 mIU/mL. The average level of LH in patients with normal testis before surgery was 4.46±2.58 IU/L, which decreased to 4.09±2.19 IU/L after surgery. The mean level of LH in patients with firm testis before surgery was 4.75±2 IU/L and after surgery was 4.42±1.87 IU/L. The average testosterone level in patients with normal testis before surgery was 346.61±178.38 ng/dL, and the level in patients with firm testis was 312.66±138.97 ng/dL. Based on Mann–Whitney Wilcoxon U examination, there is no significant difference in testosterone (P=0.79), FSH (P=0.29), LH (P=0.57), and testosterone (P=0.45) between normal testis and firm testis (Table 2).
The average FSH level after surgery in patients with normal testis was 4.04±2 mIU/mL and in patients with firm testis was 5.12±2.77 mIU/mL. Based on Mann–Whitney Wilcoxon U examination, there is no significant difference in LH level examination (P=0.42). In this study, seven patients had grade I varicocele, 24 patients had grade II varicocele, and 69 patients had grade III varicocele. The average testosterone level in patients with grade I varicocele before surgery was 492.42±227.34 ng/dL and after surgery increased to 574.71±191.70 ng/dL. The average testosterone level in patients with grade II varicocele before surgery was 423.45±161.61 ng/dL, and after surgery increased to 484.29±127.55 ng/dL. The mean FSH level in patients with grade I varicocele before surgery was 3.67±1.78 mIU/mL, and after surgery decreased to 3.21±1.37 mIU/mL. The average FSH level in patients with grade II varicocele before surgery was 4.76±2.19 mIU/mL and after surgery decreased to 4.27±1.84 mIU/mL. Based on paired-samples statistics examination, there is a significant difference between FSH levels before surgery and after surgery in patients with grade II varicocele (P=0.02). The average FSH level in patients with grade III varicocele before surgery was 4.51±2.93 mIU/mL and after surgery decreased to 4.18±2.22 mIU/mL. Based on paired-samples statistics examination, there is a significant difference between LH levels before surgery and after surgery in patients with grade II varicocele (P=0.02). The average LH level in patients with grade III varicocele before surgery was 4.36±2.68 IU/L and after surgery decreased to 4.03±2.23 IU/L. The average difference in the increasing level of testosterone was 119.48±89.26 ng/dL in hypogonadal patients after varicocelectomy and 37.74±74.85 ng/dL in eugonadal patients after varicocelectomy (Table 3).
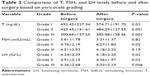  | Table 3 Comparison of T, FSH, and LH levels before and after surgery based on varicocele grading |
The mean difference in the increasing level of testosterone in patients with varicocele grades I, II, and III after surgery was 82.28±81.56, 60.83±15.93, and 84.42±11.68 ng/dL, respectively. Based on ANOVA with the P-value of 0.55, there was no significant difference between the increasing levels of testosterone in patients with varicocele grades I, II, and III. Based on ANOVA examination, there was no significant difference between increased levels of testosterone in patients with varicocele grades I and II. The comparison of increasing level of testosterone in patients with varicocele grade I with that in patients with varicocele grade III also was not significant. According to the comparison, the increase in testosterone level of patient with grade III varicocele was not significant.
The mean level difference of FSH in patients with varicocele grades I, II, and III was 0.39±0.62, 0.49±0.96, and 0.32±0.24 mIU/mL, respectively. Based on ANOVA statistics examination with P-value =0.92, there was no significant difference in the decreasing level of FSH according to patients in varicocele grade in this study. Based on conducted comparison between patients’ FSH level decreasing with varicocele grade I and III, there was no significant difference after surgery in patients with varicocele grades I, II, and III and the differences were 1.12±0.46, 0.64±0.51, and 0.177±0.35 IU/L, respectively. Based on ANOVA examination with P-value 0.52, there was no significant difference between the decreasing level of LH and the grade of varicocele. Based on ANOVA and the comparison of the decreasing level of LH in patients with varicocele grade I versus grade II, the decreasing level of LH was not significant. Based on ANOVA and the comparison of the decreasing level of LH in patients with varicocele grade II versus grade III, there was also no significant difference between the decreasing level of LH in patients with varicocele grades II and III.
The mean difference in the increasing levels of testosterone in patients with normal testis after surgery was 79.08±9.61 ng/dL, and the level in patients with firm testis was 96.48±32.16 ng/dL. The mean decreasing level of FSH in patients with normal testis was 0.42±1.8 mIU/mL, and the level in patients with firm testis was 1.5±0.19 mIU/mL. Based on nonparametric Mann–Whitney U statistics examination, there was no significant difference between variations in the decreasing level of FSH in patients with firm and normal testis. The mean difference in the LH reduction in patients with normal testis was 0.42±0.29 IU/L, and the difference in patients with firm testis was 0.79±0.37 IU/L. Based on nonparametric Mann–Whitney U statistics examination, there is no significant difference between variation in LH level in patients with normal and firm testis.
Discussion
Varicocele is dilation of veins in pampiniform plexus and the veins’ turbulent blood flow.1 The relation between clinical varicocele and impaired testosterone production is not clearly known.2,4 The mean testosterone level in hypogonadal patients before surgery was significantly less than in hypogonadal patients after surgery. The mean FSH level had no significant decrease but the mean LH level had a significant decrease following varicocelectomy. In eugonadal group, the testosterone level before surgery was slightly changed after surgery, which was not significant. A decrease in FSH and LH after surgery was significant in both groups. In grades I, II, and III, an increase in testosterone was significant.
Hypothalamus by depleting GnRH releases LH and FSH from anterior pituitary gland. LH and FSH are the primary hormones of pituitary, which regulate testicular function. Androgen and estrogen regulate LH releasing by negative feedback pass way. LH and FSH are known as only effective gonadal hormones. In testis, LH stimulates steroidogenesis by the induction of mitochondrial conversion of cholesterol to pregnenolone and testosterone in Leydig cells and FSH banded to Sertoli cells and spermatogonia membrane in testis, and it is a main growth stimulator of seminifer tubules beginning in puberty. Varicocele is a common problem and increases internal scrotal temperature, and high temperature can cause reduction of testosterone synthesis by Leydig cells, and germinal cells damage protein metabolism and decrease Sertoli cell function.
The adverse effects of varicocele on spermatogenesis and fertility have been known for a long time, but the relation between clinical varicocele and impaired testosterone production is not clearly identified.2–4 In some hypothesis, harmful effects of varicocele on testis are explained and, in their comments, the effects of ambient temperature are accepted. In normal condition in scrotum, there is a temperature exchange mechanism between the out flow from pampiniform venous plexus and blood in flow to testis as supporting the cooling temperature required for optimal sperm production.20
Thermal effects caused by the varicoocele have negative effect on the production of testosterone.18,21 In this study, TT after varicocelectomy in patients with hypogonadism increased significantly compared to before surgery.
Abdel-Meguid et al studied a group of men, monitored them for 6 months, indicated that the improvement in testosterone level in hypogonadal patients was clearly more than that in eugonadal patients, and finally concluded that varicocele could decrease testosterone level, leading to an improvement in hypogonadal patients.13 Alan and Louis, in a retrospective study, indicated that varicocelectomy improves the testosterone level independent of varicocele grade, which is fully consistent with our results.12 In our study, the mean difference in the increasing level of testosterone in grades I, II, and III showed a significant increasing per level after varicocelectomy. In the comparison of the difference between TT of grades I, II, and III, there was no significant difference suggesting the notifiable effect of varicocele grade, which is similar to our results. In 50 cases of hypogonadal patients after varicocelectomy, there was improvement in 34 cases (68%) (T>200), of whom three (6%) were grade I, six (12%) were grade II, and 25 (50%) were grade III. The serum level of testosterone after varicocelectomy in patients with firm testis and normal testis was increased significantly, but the difference level in patients with firm testis and normal testis before surgery and after surgery was not significant.
Su et al15 showed that the men with firm testis based on physical examination before surgery had more increase in testosterone level after varicocelectomy. A decrease in the serum level of FSH and LH was shown after varicocelectomy and was different in the various subgroups. As FSH reduction in grade III and grade I was not significant, FSH reduction was significant. FSH reduction was also not significant between hypogonadal and eugonadal patients, based on physical examination. LH in grades I, II, and III of hypogonadal and eugonadal patients with varicocele, with firm testis and normal testis, showed a significant decrease, which could show improvement in testosterone level due to varicocelectomy.
Li et al showed the decreasing level of FSH and LH after varicocelectomy which was nonsignificant in almost cases.6 In this study, LH and FSH gonadotropins before varicocelectomy were normal. To date, there have been few studies on LH, DSH, and testosterone, and there is a need for more research in this field. It is suggested that similar study evaluating hormonal changes after varicocelectomy in a time shorter than 3 months be done in the future.
Limitations
The major limitation of this study was small sample size and performing in the single center. Further studies are needed to access additional information about the prediction of serum level of testosterone, LH, and FSH in patients with varicocele for better treatment.
Conclusion
Our study results in comparison with other studies showed that in patients with varicocele due to varicocelectomy, testosterone level improved, so that its increase in hypogonadal patients is more significant than in eugonadal patients. It seems that surgical treatment is useful for the increase in testosterone. The decreasing level of FSH and LH was shown in all patient groups with varicocele after varicocelectomy due to testosterone increase.
Acknowledgment
The authors would like to thank the research committee of Urmia University of Medical Sciences for their support.
Author contributions
MMR and MA contributed to the research idea and study design. AN and MMR contributed to the data acquisition. RV and ATA contributed to the data analysis/interpretation. RV contributed to the statistical analysis. MMF contributed to the supervision or mentorship. CD and RV contributed to the drafting and writing. All authors contributed toward data analysis, drafting and critically revising the paper, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Alan JW, Louis RK. Campbell Walsh Text Book of Urology. 10th ed. Philadelphia: Elsevier; 2011:636. | ||
Meacham RB, Townsend RR, Rademacher D, Drose JA. The incidence of varicoceles in the general population when evaluated by physical examination, gray scale sonography and color Doppler sonography. J Urol. 1994;151(6):1535–1538. | ||
Gorelick JI, Goldstein M. Loss of fertility in men with varicocele. Fertil Steril. 1993;59(3):613–616. | ||
World Health Organization. The influence of varicocele on parameters of fertility in a large group of men presenting to infertility clinics. Fertil Steril. 1992;57(6):1289–1293. | ||
Tanrikut C, Goldstein M. Varicocele repair for treatment of androgen deficiency. Curr Opin Urol. 2010;20(6):500–502. | ||
Li F, Yue H, Yamaguchi K, et al. Effect of surgical repair on testosterone production in infertile men with varicocele: a meta-analysis. Int J Urol. 2012;19(2):149–154. | ||
Irkilata HC, Yildiz O, Yildirim I, et al. The vasodilator effect of testosterone on the human internal spermatic vein and its relation to varicocele grade. J Urol. 2008;180(2):772–776. | ||
Fretz PC, Sandlow JI. Varicocele: current concepts in pathophysiology, diagnosis, and treatment. Urol Clin North Am. 2002;29(4):921–937. | ||
Luo DY, Yang G, Liu JJ, Yang YR, Dong Q. Effects of varicocele on testosterone, apoptosis and expression of StAR mRNA in rat Leydig cells. Asian J Androl. 2011;13(2):287–291. | ||
Hsiao W, Rosoff JS, Pale JR, Greenwood EA, Goldstein M. Older age is associated with similar improvements in semen parameters and testosterone after sub inguinal microsurgical varicocelectomy. J Urol. 2011;185(2):620–625. | ||
Yildiz O, Gul H, Ozgok Y, et al. Increased vasoconstrictor reactivity and decreased endothelial function in high grade varicocele; functional and morphological study. Urol Res. 2003;31(5):323–328. | ||
Alan JW, Louis RK. Campbell Walsh Text Book of Urology. 10th ed. Philadelphia: Elsevier 2011:810–820. | ||
Abdel-Meguid TA, Farsi HM, Al-Sayyad A, Tayib A, Mosli HA, Halawani AH. Effects of varicocele on serum testosterone and changes of testosterone after varicocelectomy: a prospective controlled study. Urology. 2014;84(5):1081–1087. | ||
Hsiao W, Rosoff JS, Pale JR, Powell JL, Goldstein M. Varicocelectomy is associated with increases in serum testosterone independent of clinical grade. Urology. 2013;81(6):1213–1218. | ||
Su LM, Goldstein M, Schlegel PN. The effect of varicocelectomy on serum testosterone levels in infertile men with varicoceles. J Urol. 1995;154(5):1752–1755. | ||
Gat Y, Gornish M, Belenky A, Bachar GN. Elevation of serum testosterone and free testosterone after embolization of the internal spermatic vein for the treatment of varicocele in infertile men. Hum Reprod. 2004;19(10):2303–2306. | ||
Jangkhah M, Farrahi F, Shiravi A, et al. S21 the effect of varicocelectomy on the serum testosterone level. Eur Urol Supplements. 2013;12(4):e1129. | ||
Jangkhah M, Farrahi F, Shiravi A, et al. P-26: comparison of serum testosterone level between men with and without varicocele. Int J Fertil Steril. 2013;7(3):45. | ||
Jangkhah M, Farrahi F, Shiravi A, et al. The effect of varicocelectomy on serum testosterone level in infertile men with varicocele: an interventional study. Tehran Univ Med J. 2015;73(6):420–424. | ||
Zorgniotti AW. Testis temperature, infertility, and the varicocele paradox. Urology. 1980;16(1):7–10. | ||
Sathya Srini V, Belur Veerachari S. Does varicocelectomy improve gonadal function in men with hypogonadism and infertility? Analysis of a prospective study. Int J Endocrinol. 2011;2011:916380. | ||
Wein AJ, Kavoussi LR, Partin AW, Peters CA. Campbell-Walsh Urology, 11th Edition. New York: Elsevier; 2016. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.