Back to Journals » Neuropsychiatric Disease and Treatment » Volume 12
A door-to-door survey to estimate the prevalence of Parkinsonism in Pakistan
Authors Khan S, Nabi G , Naeem M, Ali L, Silburn P, Mellick G
Received 9 April 2015
Accepted for publication 18 May 2015
Published 21 June 2016 Volume 2016:12 Pages 1499—1506
DOI https://doi.org/10.2147/NDT.S86329
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Roger Pinder
Suliman Khan,1 Ghulam Nabi,2 Muhammad Naeem,3 Liaqat Ali,4 Peter A Silburn,1,5 George D Mellick1
1Clinical Neuroscience Group, Eskitis Institute for Drug Discovery, Griffith University, Nathan, QLD, Australia; 2Department of Animal Sciences, 3Department of Biotechnology, Quaid-i-Azam University, Islamabad, 4Saidu Medical College, Saidu Sharif Swat Khyber Pakhtunkhwa, Pakistan; 5The University of Queensland Centre for Clinical Research, Brisbane, QLD, Australia
Introduction: Parkinson’s disease (PD) occurs in all races and cultures, and population-related differences in frequency may provide etiological clues. The present study was designed to explore the prevalence of PD and Parkinsonism in Pakistan, the world’s sixth most populous country, for which no published prevalence data are available.
Methods: We conducted a three-phase door-to-door survey in two districts of the Khyber Pakhtunkhwa province of Pakistan, to assess the prevalence of PD and Parkinsonism in a sample of 4,000 individuals aged 50 years and above.
Results: We identified 14 cases of Parkinsonism, eleven with a diagnosis of idiopathic PD. The overall prevalence estimates were 1.7/100 (95% confidence interval [CI]: 0.9-2.46) for Parkinsonism and 1.28/100 (95% CI: 0.6-1.94) for PD in persons aged 65 years and above. The age-standardized prevalence of PD (aged 65 years and above), normalized to the USA population in 2000, was 1.33/100, which is similar to that observed in other human populations. Of the total 14 cases, five were newly diagnosed and four had a family history of PD.
Conclusion: The estimated prevalence rates in Pakistan are similar to those observed in other human populations. The frequency of familial Parkinsonism is also equivalent to previous estimates.
Keywords: Parkinsonism, Parkinson’s disease, door-to-door survey
Introduction
The reported prevalence of Parkinson’s disease (PD) in different parts of the world varies considerably. Approaches such as door-to-door survey and record-based strategies have been used to estimate the prevalence of Parkinsonism in both Asian and Western populations. Some studies report lower prevalence rates in Asian populations, suggesting differences in environmental conditions and geographic or racial differences.1 Other studies report similar prevalence figures for Asian and Western populations.2 A recent community-based door-to-door survey study from Iran reported the prevalence rate of Parkinsonism closer to that of the European population.3 The probable reasons for these discrepancies are methodological differences, the use of variable diagnostic criteria, and a failure to consider differences in the age-distribution of the studied populations.
Previous literature suggested that multiphase questionnaire-based door-to-door surveys followed by examination of the suspected cases by a neurologist is a cost-effective method to estimate the prevalence of PD and can have a high rate of detection of previously undiagnosed cases. In previous door-to-door survey studies, the proportion of previously undiagnosed cases identified ranged from 24% to 48%.2,4
Consanguineous marriages are still common in developing countries, which may pose serious health implications. Highly consanguineous populations, such as the Pakistani population, may exhibit nearly twice the risk of inherited disorders (autosomal recessive) compared to outbred populations.5 Compared to developing countries such as India and People’s Republic of China, there is no available information of the prevalence of PD or Parkinsonism from Pakistan. Approximately 65% of Pakistan’s population resides in rural areas6 with limited access to large hospitals, which are mainly located in major cities. Furthermore, there is also a lack of hospital-based registers to track patients’ records.
In the present study, we report on the results of a three-phase door-to-door survey to assess the prevalence of idiopathic Parkinson’s disease (iPD) and Parkinsonism in two rural districts of the Khyber Pakhtunkhwa (KPK) province of Pakistan.
Methods
Study population
The study was carried out in the KPK province of Pakistan. The total area of the province is 74,521 km2 and its projected 2011 population was 25,697,000. Lower Dir and Malakand are two of the 25 districts situated in the northern region of the KPK province, with a projected population of 1,124,000 and 703,000, respectively. Almost 90% of the populations of both the districts live in rural areas.7
Ethical approval
Approval for this study was granted by the Institutional Research and Ethics Board of the Lady Reading Hospital (reference number 05/IREB/PGMI/LRH), Quaid-i-Azam University, and Griffith University (ESK/01/15/HREC).
Screening instrument
A standard questionnaire that sensitively assesses the presence of the symptoms of Parkinsonism, based on the previously published screening questionnaire developed by Rocca et al8 was used for the current study. The instrument consisted of 13 questions, which were translated into the national language (Urdu) (Supplementary material). The screening instrument consisted of 12 questions specific to Parkinsonism (Figure 1). Any participant with two positive answers to questions pertaining to resting tremor, bradykinesia, rigidity, and postural instability9 or with ≥6 positive answers to questions in the screening instrument were considered to be eligible for physical examination. Due to low schooling in the target population, one question about small handwriting present in the original questionnaire was excluded from the instrument.
  | Figure 1 The age-specific prevalence of iPD in Pakistan as assessed by our door-to-door survey. |
Study design
The study consisted of three phases. A total of 4,000 individuals aged 50 years and above were interviewed from both districts.
Phase I
In Phase I, conducted between September 2012 and November 2013, interviewers randomly selected houses and people within the region. Participants provided verbal consent to take part in the study. Information related to the address and date of birth was taken from participants’ national identity card. For respondents who were unable to read, the interviewers asked the questions from the screening instrument. Individuals were deemed positive in Phase I on the basis of the criteria defined in the “Screening instrument” section.
Phases II and III
Individuals who screened positive in Phase I were contacted by telephone to arrange a follow-up clinical examination by a trained medical physician. Subjects were asked to bring to the examination any current or previous medical information or prescriptions. The presence of symptoms of Parkinsonism was assessed by clinical examination supported by available previous medical data; this excluded subjects with a formal diagnosis or prescription for conditions other than Parkinsonism that could explain the positive answers to the screening questions. In Phase III, individuals suspected of having signs of Parkinsonism were referred to a specialist neurologist for further detailed examination and diagnosis. The neurologist also took a family history to assess for the presence of additional family members with Parkinsonism (known as “Guzan” in the local language). Families with two or more affected members were considered positive for family history.
Questionnaire validation
The screening questionnaire was validated on 26 hospital-based patients with neurologist-diagnosed PD and 29 randomly selected neurologically normal individuals. The study was conducted at the Lady Reading Hospital, Peshawar, KPK province. PD patients were recruited from the outpatient department and the Neurology ward for the validation of the screening questionnaire.
Statistical analysis
Crude and age-specific prevalence rates were estimated by dividing the number of Parkinsonism cases by the total number of screened individuals. The age-specific prevalence rate was calculated across different age groups with 95% confidence interval (CI). Age-standardized prevalence was calculated by normalizing the data to the standardized USA population in 2000. Extrapolated prevalence for nonrespondents was estimated using Ogura’s method, which assumes that persons who screened positive but did not go to the clinic have a similar prevalence as the individuals who did go to the clinic.10,11 The formula is shown below:
|
where A is the number of individuals by age and sex, B is the number of individuals with positive screening, C is the number of individuals evaluated in a phase, N is the number of individuals affected, and S is the sensitivity (Table 1).
Prevalence ratios were reported per 100. Independent samples t-test or chi-square test, and one way Analysis of Variance (ANOVA) were used to analyze the differences between responders and nonresponders with respect to age, location, and sex.
Results
Phase I
In the first phase of the survey, a total of 4,000 individuals were approached to participate; 285 individuals refused. The overall participation rate in Phase I was 92.8%. The participation rates in Lower Dir and Malakand districts were 90% and 95%, respectively. The mean age of the participants was 61.6 years (standard deviation [SD]: 9.7 years), with a range of 50–97 years. Furthermore, 61.5% of the participants were male (2,288 of 3,715), with mean age of 62.2 years (SD: 9.8 years). The mean age of the female participants was 60.6 years (SD: 9.6 years). Complete demographic characteristics are summarized in Table 1. Of the 3,715 people who completed the screening questionnaire, 303 (8.2%) screened positive for the selected diagnostic criteria.
Phases II and III
Of the 303 participants identified in Phase I, 205 (68%) were clinically examined; 98 (32%) refused or were unavailable for clinical examination. The total response rate in Phase II was 68%. The mean age and sex distribution was not significantly different between respondents and nonrespondents (Table 2). Of the 205 positive individuals, a total of 14 patients were identified with symptoms of Parkinsonism, which were further confirmed in Phase III by a trained neurologist (Table 1). Among the 14 cases, eleven comprised iPD and three cases were identified as exhibiting Parkinsonism (two atypical Parkinsonism and one drug-induced Parkinsonism). Nine of these cases had been previously diagnosed by a neurologist; five individuals were newly diagnosed. Besides iPD and Parkinsonism, other disorders such as self reported tremor, stroke, and epilepsy were also reported by the respondents (Figure 2).
  | Table 2 Number of respondents and nonrespondents in Phase II |
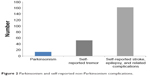  | Figure 2 Parkinsonism and self-reported non-Parkinsonism complications. |
Prevalence
Table 1 summarizes the prevalence data. The crude prevalence of all types of Parkinsonism for ages 50 years and above was 0.55/100 (95% CI: 0.3–0.8) and that for ages 65 years and above was 1.7/100 (95% CI: 0.9–2.46). The crude prevalence of iPD for ages 50 years and above was 0.43/100 (95% CI: 0.22–0.64), and for ages 65 years and above, it was 1.28/100 (95% CI: 0.6–1.94). For both Parkinsonism and iPD, prevalence figures were similar for men and women.
The prevalence figure of iPD in people aged 50 years and above was 0.53/100 (95% CI: 0.27–0.84) for males and 0.3/100 (95% CI: 0.11–0.72) for females, while for participants aged 65 years and above, the data were 1.31% (95% CI: 0.6–2.3) and 1.21% (95% CI: 0.44–2.70) for males and females, respectively (Table 3). After standardization to the USA population in 2000, the prevalence of iPD for ages 50 years and above was 0.66/100 (95% CI: 0.4–0.9); for ages 65 years and above, it was 1.33/100 (95% CI: 0.7–2.1). Prevalence of both Parkinsonism and iPD was not significantly different between the two districts. Age-specific prevalence ratios are described in Table 1. The highest prevalence rate was found in the age group 75–79 years, which was 2.0/100 (95% CI: 0–3.9). In the age group of 80 years and above, the rate was 1.2/100 (95% CI: 0–2.4). A total of four cases were identified with positive family history.
Questionnaire validation
Our screening instrument showed 100% sensitivity and 96.6% specificity using Phase I criteria. Two questions in the first part of the screening tool (arising from chair and poor balance) demonstrated a low degree of specificity (Figure 3). The remaining questions demonstrated excellent specificity.
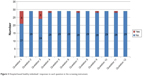  | Figure 3 Hospital-based healthy individuals’ responses to each question in the screening instrument. |
Discussion
This study used a door-to-door survey method to estimate the prevalence of Parkinsonism in provincial Pakistan. This approach had advantages relative to a record-based study design, which may underestimate the prevalence of PD, especially in countries with limited access to centers treating movement disorders. Record-based studies from Asian countries have reported prevalence rates of 0.35–0.68 per 100.12,13 Ratios estimated through the door-to-door methods are generally higher (0.51–1.76 per 100 for all ages),2,14 although one isolated study by Wang et al15 is an exception to this trend.
Our Phase I participation rate (92%) is similar to that obtained in other door-to-door surveys.16 A somewhat lower response rate of 68% was observed in Phase II, although no significant differences were observed between the respondents and nonrespondents in terms of age and sex. The Phase II response rate of a three-phase door-to-door survey conducted in Bidasoa, Spain, was 78.5%,11 which is relatively similar to our Phase II response rate. In Asia, most of the door-to-door surveys have been conducted in People’s Republic of China. A large-scale cross-sectional study2 performed in the Chinese population reported a prevalence rate of 1.7/100 in individuals aged 65 years and above, which is close to our estimated rate of 1.23/100.
Our estimated prevalence figure is also similar to that in door-to-door surveys conducted in European countries. A large collaborative door-to-door study16 conducted in different European countries (EUROPARKINSON) reported a prevalence rate for PD of 1.6/100, which is close to the PD rate of 1.23/100 found in our study. It is generally accepted that the incidence of PD is higher in men. Several studies observe a higher prevalence of PD in men,17 while others report no difference.16 In our study, we observed similar rates of Parkinsonism and iPD in men and women. Our result is similar to that reported in the Chinese population.2
A screening instrument with high sensitivity and specificity can be a good tool for early detection of the symptoms of Parkinsonism. Our screening questionnaire had high sensitivity (100%) and specificity (96.2%) in hospital-based samples of patients and controls. A similar sensitivity and specificity was reported by Duarte et al18 using a nine-item screening instrument in 50 hospital-based PD patients and 100 ophthalmological patients.
In the present study, we identified five individuals with PD. None of these people had a previous diagnosis of the disease. This rate of previously undiagnosed PD cases is similar to that obtained in earlier door-to-door studies.2,4 PD is more frequent in older populations. A similar trend of increasing prevalence with age was found in our study. A peak prevalence rate was observed at the age of 75–79 years, followed by a decrease at 80 years and above (Figure S1). Similar relationships between prevalence and age are reported elsewhere.19 It has been speculated that the observed drop-off at very old ages is in part due to the reduced sample size in this tier of the population and the reduced participation rate of very elderly members of the society. It has also been suggested that common conditions, such as osteoarthritis, stroke, peripheral neuropathy, or dementia, in older individuals may confound the assessment of Parkinsonism.20
High consanguinity may lead to increased risk of segregating inherited disorders. Consanguineous marriages are commonly practiced in Pakistan. It is estimated that the prevalence of consanguineous marriages in Pakistan is almost 60%, with 80% between first cousins.21 We were therefore interested to know whether the frequency of familial Parkinsonism was different in our target population relative to other studies worldwide. In our surveyed samples, we identified four cases with a positive family history. This is similar to the ratio (10%–25%) reported in the literature22 and does not support the idea that there is an obvious increased prevalence of familial Parkinsonism in this group.
Limitations of study
The main limitation of this study is the low response rate in Phase II, which may generate uncertainty in estimating the prevalence ratio. However, the issue was resolved by calculating the extrapolated prevalence ratio, as there was no significant difference between responders and nonresponders.
Conclusion
In conclusion, we identified a prevalence of Parkinsonism and iPD in different regions of Pakistan similar to that reported in other parts of the world. A significant number of individuals with PD were newly diagnosed as part of the study. The ratio of familial PD was similar to those previously reported.
Acknowledgments
The authors thank the subjects for their participation. We thank Dr Rasif Salam for helping in diagnosing patients in Phase II, Professor Dr Intikhab Alam and Professor Dr Adnan Khan, Lady Reading Hospital, Peshawar, Pakistan, for assisting with questionnaire validation, and Mr Tariq Aziz for help with data collection.
Disclosure
Suliman Khan is supported by an Australian Postgraduate Award and Griffith University Postgraduate Research Scholarship. The authors report no other conflicts of interest in this work.
References
Pringsheim T, Jette N, Frolkis A, Steeves TD. The prevalence of Parkinson’s disease: a systematic review and meta-analysis. Mov Disord. 2014;29(13):1583–1590. | ||
Zhang ZX, Roman GC, Hong Z, et al. Parkinson’s disease in China: prevalence in Beijing, Xian, and Shanghai. Lancet. 2005;3(9459):595–597. | ||
Fereshtehnejad S, Shafieesabet M, Rahmani A, Delbari A, Lokk J. Medium-to-high prevalence of screening-detected Parkinsonism in the urban area of Tehran, Iran: data from a community-based door-to-door study. Neuropsychiatr Dis Treat. 2015;11:321–332. | ||
Benito-Leon J, Bermejo-Pareja F, Rodriguez J, Molina J, Gabriel R, Morales J. Prevalence of PD and other types of Parkinsonism in three elderly populations of central Spain. Mov Disord. 2003;18(3):267–274. | ||
Hoodfar E, Teebi A. Genetic referrals of Middle Eastern origin in a western city: inbreeding and disease profile. J Med Genet. 1996;33(3):212–215. | ||
National Institute of Population Studies (NIPS). Pakistan Demographic and Health Survey 2006–2007. Islamabad, Pakistan: Pakistan and Macro International Inc; 2008. | ||
Bureau of Statistic, Government of Khyber Pakhtunkhwa, Demographic Data 2014. Government of Khyber Pakhtunkhwa Pakistan Report; 2014:267–271. Available from: http://kpbos.gov.pk/ | ||
Rocca WA, Maraganore DM, McDonnell SK, Schaid DJ. Validation of a telephone questionnaire for Parkinson’s disease. J Clin Epidemiol. 1998;51(6):517–523. | ||
Melcon MO, Anderson DW, Vergara RH, Rocca WA. Prevalence of Parkinson’s disease in Junin, Buenos Aires Province, Argentina. Mov Disord. 1997;12(2):197–205. | ||
Ogura C, Nakamoto H, Uema T, Yamamoto K, Yonemori T, Yoshimura T. Prevalence of senile dementia in Okinawa, Japan. Int J Epidemiol. 1995;24(2):373–380. | ||
Bergareche A, De La Puente E, López de Munain A, et al. Prevalence of Parkinson’s disease and other types of Parkinsonism. A door-to-door survey in Bidasoa, Spain. J Neurol. 2004;251(3):340–345. | ||
Kimura H, Kurimura M, Wada M, et al. Female preponderance of Parkinson’s disease in Japan. Neuroepidemiology. 2002;21(6):292–296. | ||
Harada H, Nishikawa S, Takahashi K. Epidemiology of Parkinson’s disease in a Japanese city. Arch Neurol. 1983;40(3):151–154. | ||
Li SC, Schoenberg BS, Wang CC, et al. A prevalence survey of Parkinson’s disease and other movement disorders in the People’s Republic of China. Arch Neurol. 1985;42(7):655–657. | ||
Wang YS, Shi YM, Wu ZY, He YX, Zhang BZ. Parkinson’s disease in China. Coordinational group of neuroepidemiology, PLA. Chin Med J (Engl). 1991;104(11):960–964. | ||
de Rijk MC, Tzourio C, Breteler MM, et al. Prevalence of Parkinsonism and Parkinson’s disease in Europe: the EUROPARKINSON collaborative study. European community concerted action on the epidemiology of Parkinson’s disease. J Neurol Neurosurg Psychiatry. 1997;62(1):10–15. | ||
Hofman A, Collette H, Bartelds A. Incidence and risk factors of Parkinson’s disease in the Netherlands. Neuroepidemiology. 1989;8(6):296–299. | ||
Duarte J, Claveria L, De Pedro-Cuesta J, Sempere A, Coria F, Calne D. Screening Parkinson’s disease: a validated questionnaire of high specificity and sensitivity. Mov Disord. 1995;10(5):643–649. | ||
Kis B, Schrag A, Ben-Shlomo Y, et al. Novel three-stage ascertainment method prevalence of PD and Parkinsonism in South Tyrol, Italy. Neurology. 2002;58(12):1820–1825. | ||
Bower J, Maraganore D, McDonnell S, Rocca W. Influence of strict, intermediate, and broad diagnostic criteria on the age- and sex-specific incidence of Parkinson’s disease. Mov Disord. 2000;15(5):819–825. | ||
Hussain R, Bittles A. The prevalence and demographic characteristics of consanguineous marriages in Pakistan. J Biosoc Sci. 1998;30(02):261–275. | ||
Sellbach AN, Boyle RS, Silburn PA, Mellick GD. Parkinson’s disease and family history. Parkinsonism Relat Disord. 2006;12(7):399–409. |
Supplementary material
  | Figure S1 English and Urdu versions of the 12-item screening instrument questionnaire. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.