Back to Journals » Clinical Epidemiology » Volume 10
A cohort study on the risk of lymphoma and skin cancer in users of topical tacrolimus, pimecrolimus, and corticosteroids (Joint European Longitudinal Lymphoma and Skin Cancer Evaluation – JOELLE study)
Authors Castellsague J , Kuiper JG , Pottegård A , Anveden Berglind I, Dedman D , Gutierrez L , Calingaert B , van Herk-Sukel MP, Hallas J , Sundström A, Gallagher AM, Kaye JA , Pardo C, Rothman KJ, Perez-Gutthann S
Received 19 July 2017
Accepted for publication 27 January 2018
Published 13 March 2018 Volume 2018:10 Pages 299—310
DOI https://doi.org/10.2147/CLEP.S146442
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Vera Ehrenstein
Jordi Castellsague,1 Josephina G Kuiper,2 Anton Pottegård,3 Ingegärd Anveden Berglind,4 Daniel Dedman,5 Lia Gutierrez,1 Brian Calingaert,6 Myrthe PP van Herk-Sukel,2 Jesper Hallas,3 Anders Sundström,4 Arlene M Gallagher,5 James A Kaye,7 Carolina Pardo,8 Kenneth J Rothman,7 Susana Perez-Gutthann1
1Department of Epidemiology, RTI Health Solutions, Barcelona, Spain; 2Department Research, PHARMO Institute for Drug Outcomes Research, Utrecht, the Netherlands; 3Clinical Pharmacology and Pharmacy, Department of Public Health, University of Southern Denmark, Odense, Denmark; 4Centre for Pharmacoepidemiology, Unit of Clinical Epidemiology, Department of Medicine, Karolinska Institutet, Stockholm, Sweden; 5Clinical Practice Research Datalink, Medicines and Healthcare products Regulatory Agency, London, UK; 6Department Epidemiology, RTI Health Solutions, Research Triangle Park, NC, 7Department of Epidemiology, RTI Health Solutions, Waltham, MA, USA; 8Pharmacovigilance Department, Astellas Pharma Europe B.V., Leiden, the Netherlands
Background: There is a concern that topical tacrolimus and pimecrolimus, indicated for second-line treatment of atopic dermatitis, may increase the risk of lymphoma and skin cancer, particularly in children.
Objective: The aim of this study was to compare incidence rates (IRs) of lymphoma and skin cancer between new users of topical tacrolimus or pimecrolimus and users of moderate- to high-potency topical corticosteroids (TCSs) and untreated subjects.
Methods: This is a multicenter cohort study with frequency matching by strata of propensity scores in population databases in the Netherlands, Denmark, Sweden, and the UK. IR ratios (IRRs) were estimated using Mantel–Haenszel methods for stratified analysis.
Results: We included 19,948 children and 66,127 adults initiating tacrolimus, 23,840 children and 37,417 adults initiating pimecrolimus, 584,121 users of TCSs, and 257,074 untreated subjects. IRs of lymphoma per 100,000 person-years were 10.4 events in children and 41.0 events in adults using tacrolimus and 3.0 events in children and 27.0 events in adults using pimecrolimus. The IRR (95% confidence interval [CI]) for lymphoma, tacrolimus versus TCSs, was 3.74 (1.00–14.06) in children and 1.27 (0.94–1.71) in adults. By lymphoma type, the highest IRR was 3.17 (0.58–17.23) for Hodgkin lymphoma in children and 1.76 (95% CI, 0.81–3.79) for cutaneous T-cell lymphoma (CTCL) in adults. For pimecrolimus versus TCSs, the highest IRR was 1.31 (95% CI, 0.33–5.14) for CTCL in adults. Compared with untreated subjects, adults using TCSs had a higher incidence of CTCL (IRR, 10.66; 95% CI, 2.60–43.75). Smaller associations were found between tacrolimus and pimecrolimus use and the risk of malignant melanoma or nonmelanoma skin cancer.
Conclusion: Use of topical tacrolimus and pimecrolimus was associated with an increased risk of lymphoma. The low IRs imply that even if the increased risk is causal, it represents a small excess risk for individual patients. Residual confounding by severity of atopic dermatitis, increased monitoring of severe patients, and reverse causation could have affected the results.
Keywords: topical calcineurin inhibitors, cutaneous T-cell lymphoma, malignant melanoma skin cancer, database study
Introduction
Topical tacrolimus and pimecrolimus are topical calcineurin inhibitors indicated for second-line treatment of atopic dermatitis in children aged 2 years and older. Tacrolimus is indicated for moderate-to-severe atopic dermatitis, and pimecrolimus is indicated for mild-to-moderate atopic dermatitis.
The long-term safety of these agents is not well established. Safety data from animal studies, systemic use in patients with organ transplants, and case reports have raised concerns about a potential increase in the risk of lymphoma and skin cancer.1–4 Data from observational studies are scarce and inconclusive, particularly in children. Some studies reported an increased risk for lymphoma, particularly for cutaneous T-cell lymphoma (CTCL), among users of these medications, but other studies did not find any association.5–8 Some of these studies had a small study size and short follow-up.5,6 The causal effect of tacrolimus and pimecrolimus is difficult to elucidate as atopic dermatitis itself and severity of atopic dermatitis have been associated with an increased risk of lymphoma.9,10
To further evaluate the safety of topical tacrolimus and pimecrolimus, we conducted a postauthorization study of children and adults in four European population-based databases. Using data from launch until the end of 2011, we compared the incidence rates (IRs) of nonmelanoma skin cancer, malignant melanoma, and lymphoma between patients initiating treatment with these agents and users of moderate- to high-potency topical corticosteroids (TCSs). The study has been extended 4 years to assess long-term effects.
Methods
Source population
We conducted a multicenter database cohort study in populations from the PHARMO Database Network in the Netherlands (PHARMO), the Danish and the Swedish national health registers, and the Clinical Practice Research Datalink (CPRD) in the UK.
The study population comprised children (aged <18 years) and adults (aged ≥18 years) initiating treatment with topical tacrolimus or pimecrolimus or treated with TCSs, from 2002 to 2011, who had at least 12 months of continuous enrollment in the study databases. Initiation of tacrolimus and pimecrolimus was defined as not having any prescription for either medication at any time before the date of cohort entry. Study TCSs were those classified as moderately potent, potent, or very potent in the Anatomical Therapeutic Chemical (ATC) classification.11 Users of TCSs were required to have a recorded diagnosis of atopic dermatitis or at least one additional prescription for TCSs in the 12 months before cohort entry. Because the study focused on incident events, we excluded all users with a history of any study outcome before cohort entry from the study population.
Study cohorts
Users of TCSs were frequency matched by twentiles (20 quantiles) of propensity scores to initiators of tacrolimus or pimecrolimus (Figure 1). Propensity scores were estimated in each database as the predicted probability of receiving treatment with tacrolimus or pimecrolimus instead of TCSs given a set of baseline covariates. Variables included in the estimation of propensity scores were those showing an association (odds ratio >1.25 or <0.80) in logistic regression models fitted for each study outcome as the independent variable. To account for patients with nonoverlapping propensity scores, patients below the 2.5th percentile and above the 97.5th percentile of the distribution of propensity scores were excluded.12 Within each stratum, frequency matching was conducted by randomly selecting users of TCSs from the eligible pool until reaching the matching ratio (4:1, TCSs to study drug). Variables evaluated in the estimation of propensity scores were age, sex, calendar year of cohort entry, health practice or region, medical history, and use of medications and health care resources (Table 1). Medical diagnoses were identified by Read codes and International Statistical Classification of Diseases and Related Health Problems, 10th revision (ICD-10) codes in CPRD, ICD-9 codes in PHARMO, and ICD-10 codes in Denmark and Sweden. Medications were identified by ATC codes. In CPRD, the National Health Service Dictionary of Medicines and Devices codes were mapped to ATC codes using SafeScript (www.safescript.com).
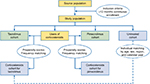  | Figure 1 Study cohorts. Note: “Corticosteroids” refer to moderate- to high-potency topical corticosteroids. |
From the general population, we identified a secondary cohort of subjects untreated with tacrolimus, pimecrolimus, or TCSs. Untreated subjects were matched in a 1:1 ratio to the cohort of users of TCSs identified as the comparative cohort for tacrolimus. Matching was done on year of birth, sex, health practice/region (except in Denmark), and calendar year of cohort entry. Patients from each study cohort were followed from the date of cohort entry to the earliest occurrence of first study outcome, death, disenrollment from the study database, or December 31, 2011.
Exposure definition
Patterns of use of tacrolimus and pimecrolimus in the study populations have been described elsewhere.13 We assumed that the time at risk associated with the use of tacrolimus and pimecrolimus started 6 months after cohort entry and continued until the end of follow-up (induction period). Person-time of follow-up was classified as ever use of tacrolimus or pimecrolimus, ignoring any potential switching between the two agents. In addition, we defined single use of tacrolimus and single use of pimecrolimus as the person-time starting 6 months after the date of cohort entry to the earliest of 6 months after the date of switching to either tacrolimus or pimecrolimus or end of follow-up.
We calculated the cumulative dose of active substance of tacrolimus and pimecrolimus that patients received during follow-up. Cumulative dose was defined as low, medium, or high according to break points established prior to the analysis based on the distribution of cumulative dose in each database. Cutoff values for tacrolimus were ≤0.05 g for low dose, >0.05–0.10 g for medium dose, and >0.10 g for high dose. Cutoff values for pimecrolimus were ≤0.5 g for low dose, >0.5–1.0 g for medium dose, and >1.0 g for high dose. For each level of cumulative dose, the time at risk started 6 months after reaching the corresponding cutoff value.
Study outcomes
The study outcomes were malignant melanoma, nonmelanoma skin cancer, any lymphoma, non-Hodgkin lymphoma (excluding CTCL), Hodgkin lymphoma, and CTCL. In situ tumors were included in the definition of malignant melanoma and nonmelanoma skin cancer.
Outcomes were identified in 1) cancer registries in Denmark, Sweden, and CPRD (linked practices), 2) the Dutch National Pathology Registry (PALGA) in PHARMO, and 3) primary care records in CPRD for practices not linked to the cancer registry. Outcomes were identified using International Classification of Diseases for Oncology, third edition (ICD-O-3) codes in Denmark and Sweden, PALGA codes mapped to ICD-O-3 codes in PHARMO, and Read and ICD-10 codes in CPRD. Outcomes identified in the cancer registries were considered confirmed.14–17 In PHARMO, an independent pathologist reviewed the pathology excerpts of all pediatric outcomes, all CTCLs, and a random sample of adult outcomes. Overall, 98.0% of outcomes were confirmed. In CPRD, we performed a clinical review of patient profiles and free-text fields for all pediatric outcomes, all CTCLs, all outcomes identified in the primary care record, and all outcomes with inconsistent information between the primary care record and the cancer registry. A total of 864 outcomes were reviewed, and 762 (88.2%) were confirmed.
Analysis
In each database, person-years of follow-up and number of outcomes were aggregated across deciles of propensity scores (after aggregating neighboring pairs of strata from the initial twentiles used to match the study cohorts), age categories, and sex. We applied Mantel–Haenszel methods for stratified analyses to estimate IRs, adjusted IR ratios (IRRs) and IR differences (IRD), and 95% confidence intervals (CIs) for each study outcome and type of exposure, maintaining database as a stratification variable. Because recorded information on the severity of atopic dermatitis was scarce in all databases, we further adjusted IRRs by the type of prescriber of the first prescription (dermatologist and nondermatologist), as a marker of atopic dermatitis severity, in those databases where this information was available (PHARMO and Sweden). To account for the effect of type of prescriber in Denmark and CPRD, we used information from PHARMO and Sweden to correct adjusted IRRs using quantitative probabilistic bias analysis for unmeasured confounders (“Methods” section in the Supplementary materials).18 These IRRs are presented as corrected IRRs (IRRc). To evaluate latency of the effect of exposure and reverse causation, we estimated IRRs by time since first exposure and for additional lag-times.
We obtained ethical and scientific reviews from the RTI International Institutional Review Board, the Regional Ethical Review Board of Stockholm, the CPRD Independent Scientific Advisory Committee, and the UK National Cancer Intelligence Network. Ethical approval was not required in PHARMO and Denmark. In PHARMO, the study fulfilled the requirements of the PHARMO Compliance Commission, and permission for the use of data from PALGA was obtained. In Denmark, the study was approved by the Danish Data Protection Agency via Statistics Denmark. The study was registered in the European Union Electronic Register of Post-Authorisation Studies (EU PAS Register) study number 4357.19
Results
The study included 19,948 children and 66,127 adults initiating treatment with tacrolimus and 23,840 children and 37,417 adults initiating treatment with pimecrolimus (Table 2). Among users of TCSs, 79,700 children and 264,482 adults were matched to new users of tacrolimus and 90,268 children and 149,671 adults were matched to new users of pimecrolimus. The untreated cohort comprised 79,040 children and 257,074 adults. In general, the matching on propensity scores achieved a good balance between the study cohorts (Tables S1 and S2).
The median follow-up ranged from 2.2 years (Sweden) to 4.2 years (CPRD) in children and from 2.2 years (Sweden) to 3.6 years (Denmark and CPRD) in adults treated with tacrolimus and from 3.2 years (Sweden) to 6.5 years (Denmark) in children and from 2.8 years (Sweden) to 5.6 years (Denmark) in adults treated with pimecrolimus.
Among children treated with tacrolimus, there were no events of malignant melanoma or nonmelanoma skin cancer and five events of any lymphoma (Table 3). The IR of any lymphoma was higher in children treated with tacrolimus than in children treated with TCSs (IRRc, 3.74; 95% CI, 1.00–14.1) (Table 3). Among adults, there was a negative association between the use of tacrolimus and malignant melanoma and no association with nonmelanoma skin cancer. For any lymphoma, the IRRc comparing adults treated with tacrolimus and TCSs was 1.27 (95% CI, 0.94–1.71). By type of lymphoma, the highest IRR was for CTCL (IRRc, 1.76; 95% CI, 0.81–3.79).
Among children treated with pimecrolimus, there were no events of malignant melanoma, one event of nonmelanoma skin cancer, and three events of lymphoma (Table 4). The IR (95% CI) per 100,000 person-years of nonmelanoma skin cancer was 0.9 (0–5.3) in children treated with pimecrolimus and 0.3 (0–1.6) in children treated with TCSs.
No marked increased rates of any lymphoma were found in children treated with pimecrolimus. In adults treated with pimecrolimus, the IR (95% CI) per 100,000 person-years was 41.1 (32.1–51.9) for malignant melanoma, 360.8 (333.1–390.3) for nonmelanoma skin cancer, and 1.7 (0.4–5.1) for CTCL compared with the IR (95% CI) per 100,000 person-years for adults treated with TCSs: 33.9 (29.7–38.6), 269.1 (256.9–281.7), and 1.6 (0.8–2.9), respectively.
Compared to untreated subjects, in general, users of TCSs had increased IRs of skin cancer and lymphoma (Table 5). The highest IRRc was 10.7 (95% CI, 2.6–44) for CTCL in adults. The actual IR for cohorts of TCSs matched to pimecrolimus or tacrolimus differed (refer “Results” section in the Supplementary materials).
IRRs for any lymphoma and CTCL increased with increasing cumulative dose of tacrolimus and pimecrolimus (Table 6). Analysis by time since first exposure in children showed no events in the first 6 months after the start of tacrolimus or pimecrolimus (Table S3). In adults, IRRs of skin cancer and CTCL were higher in the first 6 months after the start of tacrolimus or pimecrolimus than in the subsequent months (Table S4).
IRRs for any lymphoma in children treated with tacrolimus or pimecrolimus were higher for lag times 0 and 6 months than for lag times 12 and 24 months (Tables S5 and S6). In adults, IRRs for CTCL increased with longer lag times in users of tacrolimus and decreased with longer lag times in users of pimecrolimus.
Discussion
In this European multicenter study, we found an approximately fourfold increase in the IR of lymphoma in children initiating treatment with tacrolimus and a less than twofold increase in the IR of CTCL in adults initiating treatment with tacrolimus or pimecrolimus compared with users of TCSs. We also found a >10-fold increase in the rate of CTCL in adults treated with TCSs compared with untreated subjects, implying that the contrast in rates between users of tacrolimus and an untreated population would be considerably high. We found smaller associations for malignant melanoma or nonmelanoma skin cancer. CIs around effect estimates tended to be wide, especially for children, indicating low precision.
This is the first observational study reporting results on the risk of lymphoma and skin cancer for children treated only with tacrolimus and the largest study, with the longest follow-up, conducted in adults. The study has been extended for four additional years to study the effects at a later time and with greater precision. The IR of lymphoma observed in the untreated population of this study, 16.6 per 100,000 person-years, is comparable to the IR of lymphoma per 100,000 person-years in the THIN database (1720) and in the UK general population (17 in females and 19 in males21).
Results are consistent with the findings of some observational studies5,6 but not others.7 The only published study conducted in children was restricted to users of pimecrolimus and reported a 2.6-fold increase in the standardized incidence ratio for lymphoma.22 In a cohort study of patients diagnosed with atopic dermatitis, T-cell lymphoma, primarily CTCL, was the only type of cancer associated with an increased risk in users of tacrolimus and pimecrolimus compared with untreated patients.5 In another cohort study, the IR of cutaneous forms of lymphoma was higher in new users of tacrolimus and pimecrolimus than in untreated subjects.6 However, in that study, there was an increased risk for the use of high-potency TCSs versus medium-potency TCSs, suggesting reverse causation, with early manifestations of undiagnosed cutaneous lymphoma triggering the initiation of treatment, and an increased surveillance of patients with more severe forms of cutaneous diseases. Other studies have not detected any association between lymphoma and the use of topical calcineurin inhibitors7 or between nonmelanoma skin cancer and the use of pimecrolimus.8
An increased risk of cancer among solid organ transplant recipients has been well documented, particularly for infection-related skin malignancies.23–26 The risk of lymphoma in patients undergoing organ transplantation is closely related to the intensity of immunosuppression (ie, number and dose of immunosuppressive agents used) and ensuing inability of the immune system to control Epstein–Barr virus infection.27 Concerns about a potential increase in the risk of cancer from the use of topical tacrolimus emerged from the increased risk observed in animal studies, organ transplant patients treated with systemic tacrolimus, and a small number of case reports.27–29 Prolonged administration of systemic tacrolimus at sustained high concentrations in transplant patients, alongside other immunosuppressant drugs such as corticosteroids, has been associated with an increased rate of lymphomas, nonmelanoma skin cancer, and melanomas in sun-exposed areas.27–33 The hypothetical mechanisms through which topical immunosuppressants such as calcineurin inhibitors could increase the risk of cancer in patients with atopic dermatitis involve the development of local immunosuppression at the application site and/or systemic immunosuppression due to systemic absorption. Large, severe skin lesions could increase the cutaneous absorption of these topical medications, leading to higher systemic concentrations that may cause immunosuppression, particularly in children.
The increased risk of lymphoma found in our study could be compatible with a causal effect of exposure to tacrolimus and pimecrolimus. However, several limitations should be considered. Atopic dermatitis and severity of atopic dermatitis have in themselves been associated with an increased risk of lymphoma.9 This relation could introduce confounding by indication as patients with more severe atopic dermatitis are more likely to be treated with tacrolimus or pimecrolimus than with TCSs. This confounding could be more relevant for tacrolimus than for pimecrolimus as tacrolimus is indicated for more severe atopic dermatitis. In our study, we underestimated the occurrence of atopic dermatitis, as available information was mainly based on hospital discharge diagnoses. Also, we ascertained atopic dermatitis severity indirectly through the intensity of use of health care resources and type of prescriber of the first prescription. Although we corrected IRRs for the effect of type of prescriber using probabilistic bias analysis, residual confounding can still be present. The dose–response relationship observed with cumulative dose for CTCL could be compatible with a causal effect and also with residual confounding arising from the severity of atopic dermatitis. We should consider that the cutoff values for cumulative dose categories used in this study were relatively low (eg, tacrolimus high dose cutoff value was 0.10 g, which corresponds to 5.6 tubes of 60 g of tacrolimus 0.03% ointment or 1.7 tubes of 60 g of tacrolimus 0.1% ointment). Also, data on the patterns of use of tacrolimus and pimecrolimus in the study populations show that the mean number of prescriptions per patient was low in both children and adults (around two prescriptions in most databases).13 These data are more consistent with a severity bias than with a causal effect of tacrolimus and pimecrolimus. Severity of atopic dermatitis could also lead to increased monitoring and surveillance bias. The stronger associations found in the first 6 months of treatment are consistent with increased surveillance.
Reverse causation could have introduced bias in our results. CTCL can be misdiagnosed as atopic dermatitis and treated as such long term, up to 10 years.34 This possibility is supported by the findings by Hui et al;5 in four of the 16 patients with atopic dermatitis, those researchers found evidence of suspected CTCL recorded in the medical records before the initiation of treatment. Reverse causation would result in the overestimation of the measures of effect. In our study, the analysis of time since first exposure showed that the IRR for CTCL associated with tacrolimus was maintained during follow-up, suggesting the possibility of long-term reverse causation. The increased IRR associated with lag times of 12 and 24 months, although compatible with a long induction period, could also support the hypothesis of reverse causation.
We required users of tacrolimus and pimecrolimus to be first-ever users of these medications. However, to prevent including in the comparator group patients with very early stages of atopic dermatitis, the cohorts of TCSs included new and prevalent users. Inclusion of prevalent users of TCSs could result in survival bias, through the depletion of patients with early events, and subsequent overestimation of the effect of tacrolimus and pimecrolimus. However, the potential bias is probably minimal as there was no strong evidence of increased risk of lymphoma and skin cancer associated with the use of TCSs.
Results from the comparison of TCS users with the untreated population could be explained by residual confounding, as the effect of risk factors such as diagnosis of atopic dermatitis and severity of atopic dermatitis were not taken into account.
Conclusion
We found increased rates of lymphoma in children initiating treatment with topical tacrolimus and of CTCL in adults initiating treatment with topical tacrolimus or pimecrolimus. These findings are compatible with a causal effect and also with residual confounding by severity of atopic dermatitis, increased monitoring of more severe patients, and reverse causation. The low absolute magnitude of risks for lymphoma and CTCL means that even if the increased risk observed is causal, the excess risk for an individual patient would be small.
Acknowledgments
The authors would like to thank the general practitioners contributing information to the Clinical Practice Research Datalink (CPRD) in the UK and the health care providers contributing information to the PHARMO Database Network. The research team chose the acronym JOELLE to honor Dr Joelle Erkens, pharmacoepidemiologist at Astellas and formerly at PHARMO Institute, who was lost to the research team in June 2011 upon her untimely death. Note: data in the study were aggregated. Individual patient-level data from each database remained in the home institutions because of data protection rules. The study was funded by Astellas Pharma. In June 2016, LEO Pharma became the marketing authorization holder of Protopic® and provided partial funding. The contracts provide the research team independent publication rights. The sponsors had no role in the data collection or analysis and were not involved in the interpretation of results; however, in line with the Guideline on Good Pharmacovigilance Practices (GVP): Module VIII – Post-authorisation Safety Studies of the European Medicines Agency, the sponsors had the opportunity to view the results and interpretations included in the article and provide comments prior to submission of the article for publication. Preliminary, partial results of this study were presented at the 32nd International Conference on Pharmacoepidemiology & Therapeutic Risk Management, Dublin, August 25–28, 2016, and were published in Pharmacoepidemiology and Drug Safety, Volume 25, Issue Supplement S3. The article complies with the STROBE statement for the reporting of epidemiological studies (https://www.strobe-statement.org/index.php?id=strobe-home).
Author contributions
The corresponding author, JC, and BC had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. The analysis of the data for this study was conducted by the following researchers (all coauthors): JGK, AP, DD, AS, and BC. All authors contributed toward data analysis, drafting and revising the paper and agree to be accountable for all aspects of the work.
Disclosure
At the time of the study, JC, LG, BC, KJR, JAK, and SP-G were full-time employees of RTI Health Solutions, an independent nonprofit research organization that does work for government agencies and pharmaceutical companies. As employees of RTI Health Solutions, SP-G, KJR, and JAK also participate in scientific advisory boards (for studies and medications) that are funded by pharmaceutical companies. JGK and MPPvH-S are employees of the PHARMO Institute for Drug Outcomes Research. This independent research institute performs financially supported studies for government and related health care authorities and several pharmaceutical companies. JH and AP are employees of the University of Southern Denmark, Clinical Pharmacology and Pharmacy. They have participated in studies funded by pharmaceutical companies (Alcon, Almirall, Astellas, Astra-Zeneca, Boehringer-Ingelheim, Pfizer, Menarini, Servier, and Takeda), with money paid to their employer. DD and AMG are employees of the Clinical Practice Research Datalink (CPRD), which provides contract research services for government and related health care authorities and pharmaceutical companies. IAB and AS were full-time employees of the Centre for Pharmacoepidemiology at the Karolinska Institutet during the conduct of the study. They have both taken part in studies undertaken at the Centre, financed by pharmaceutical companies, but have never received compensation personally from any company. CP is an employee of Astellas Pharma Europe. The authors report no other conflicts of interest in this work.
References
European Medicines Agency. Summary information on referral opinion pursuant to Article 31 of Council Directive 2001/83/EC, as amended, for Elidel and associated names [EMEA/262776/2005]. Committee for Medicinal Products for Human Use (CHMP). 2006. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Referrals_document/Elidel_31/WC500012313.pdf. Accessed July 29, 2016. | ||
European Medicines Agency [webpage on the Internet]. Protopic: EPAR – Scientific Conclusion; 2006. Available from: http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/000374/human_med_001000.jsp&mid=WC0b01ac058001d124. Accessed July 29, 2016. | ||
US Food and Drug Administration. Information for Healthcare Professionals: Pimecrolimus (Marketed as Elidel). FDA Alert [3/2005]; 2005. Available from: https://wayback.archive-it.org/7993/20170406045053/https://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm153525.htm. Accessed February 16, 2018. | ||
US Food and Drug Administration. Information for Healthcare Professionals: Tacrolimus (Marketed as Protopic). FDA Alert [3/2005]; 2005. Available from: https://wayback.archive-it.org/7993/20170406045634/https://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm126497.htm Accessed February 16, 2018. | ||
Hui RL, Lide W, Chan J, Schottinger J, Yoshinaga M, Millares M. Association between exposure to topical tacrolimus or pimecrolimus and cancers. Ann Pharmacother. 2009;43(12):1956–1963. | ||
Schneeweiss S, Doherty M, Zhu S, et al. Topical treatments with pimecrolimus, tacrolimus and medium- to high-potency corticosteroids, and risk of lymphoma. Dermatology. 2009;219(1):7–21. | ||
Arellano FM, Arana A, Wentworth CE, Fernandez-Vidaurre C, Schlienger RG, Conde E. Lymphoma among patients with atopic dermatitis and/or treated with topical immunosuppressants in the United Kingdom. J Allergy Clin Immunol. 2009;123(5):1111–1116, 116.e1–13. | ||
Margolis DJ, Hoffstad O, Bilker W. Lack of association between exposure to topical calcineurin inhibitors and skin cancer in adults. Dermatology. 2007;214(4):289–295. | ||
Tennis P, Gelfand JM, Rothman KJ. Evaluation of cancer risk related to atopic dermatitis and use of topical calcineurin inhibitors. Br J Dermatol. 2011;165(3):465–473. | ||
Legendre L, Barnetche T, Mazereeuw-Hautier J, Meyer N, Murrell D, Paul C. Risk of lymphoma in patients with atopic dermatitis and the role of topical treatment: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;72(6):992–1002. | ||
WHO Collaborating Centre for Drug Statistics Methodology, Guidelines for ATC classification and DDD assignment 2018. Oslo, Norway, 2017. Available at: https://www.whocc.no/filearchive/publications/guidelines.pdf Accesed February 16, 2018. | ||
Glynn RJ, Schneeweiss S, Sturmer T. Indications for propensity scores and review of their use in pharmacoepidemiology. Basic Clin Pharmacol Toxicol. 2006;98(3):253–259. | ||
Kuiper JG, van Herk-Sukel M, Castellsague J, et al. Utilization of topical tacrolimus and topical pimecrolimus in Europe: results from the Joint European Longitudinal Lymphoma and Skin Cancer Evaluation (JOELLE) study [abstract]. Pharmacoepidemiol Drug Saf. 2016;25(S3):632. | ||
Barlow L, Westergren K, Holmberg L, Talback M. The completeness of the Swedish Cancer Register: a sample survey for year 1998. Acta Oncol. 2009;48(1):27–33. | ||
Casparie M, Tiebosch AT, Burger G, et al. Pathology databanking and biobanking in The Netherlands, a central role for PALGA, the nationwide histopathology and cytopathology data network and archive. Cell Oncol. 2007;29(1):19–24. | ||
Danish Health Data Authority & Danish Cancer Society Research Center [webpage on the Internet]. Validation of the Danish Cancer Registry and Selected Clinical Cancer Databases. Copenhagen: Danish Health Authority; 2012. Available from: http://sundhedsdatastyrelsen.dk/da/registre-og-services/om-de-nationale-sundhedsregistre/sygedomme-laegemidler-og-behandlinger/cancerregisteret. Accessed September 14, 2016. | ||
Gjerstorff ML. The Danish Cancer Registry. Scand J Public Health. 2011;39(7 suppl):42–45. | ||
Lash T, Fox MP, Fink AK. Applying Quantitative Bias Analysis to Epidemiologic Data. New York, NY: Springer Science + Business Media, LLC; 2009. | ||
European Network of Centres for Pharmacoepidemiology and Pharmacovigilance [webpage on the Internet]. The European Union Electronic Register of Post-Authorisation Studies (EU PAS Register); 2016. Available from: http://www.encepp.eu/encepp_studies/indexRegister.shtml. Accessed July 15, 2016. | ||
Arana A, Wentworth CE, Fernandez-Vidaurre C, Schlienger RG, Conde E, Arellano FM. Incidence of cancer in the general population and in patients with or without atopic dermatitis in the U.K. Br J Dermatol. 2010;163(5):1036–1043. | ||
Ferlay J, Bray F, Pisani P, Parkin DM. GLOBOCAN 2002. Cancer Incidence, Mortality and Prevalence Worldwide. IARC Cancer Base No. 5, Version 2.0. Lyon: IARC Press; 2004. | ||
Margolis DJ, Abuabara K, Hoffstad OJ, Wan J, Raimondo D, Bilker WB. Association between malignancy and topical use of pimecrolimus. JAMA Dermatol. 2015;151(6):594–599. | ||
Collett D, Mumford L, Banner NR, Neuberger J, Watson C. Comparison of the incidence of malignancy in recipients of different types of organ: a UK Registry audit. Am J Transplant. 2010;10(8):1889–1896. | ||
Engels EA, Pfeiffer RM, Fraumeni JF Jr, et al. Spectrum of cancer risk among US solid organ transplant recipients. JAMA. 2011;306(17):1891–1901. | ||
Euvrard S, Kanitakis J, Claudy A. Skin cancers after organ transplantation. N Engl J Med. 2003;348(17):1681–1691. | ||
Moloney FJ, Comber H, O’Lorcain P, O’Kelly P, Conlon PJ, Murphy GM. A population-based study of skin cancer incidence and prevalence in renal transplant recipients. Br J Dermatol. 2006;154(3):498–504. | ||
Opelz G, Döhler B. Lymphomas after solid organ transplantation: a collaborative transplant study report. Am J Transplant. 2004;4(2):222–230. | ||
Astellas Pharma Ltd [webpage on the Internet]. Prograf 0.5 mg hard capsules (1 mg, 5 mg). Summary of product characteristics. emc; 2015. Available from: https://www.medicines.org.uk/emc/product/6720/smpc. Accessed January 15, 2018. | ||
LEO Pharma A/S. Protopic 0.03% Ointment. Summary of Product Characteristics. European Medicines Agency; 2016. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000374/WC500046824.pdf. Accessed January 15, 2018. | ||
Jonas S, Rayes N, Neumann U, et al. De novo malignancies after liver transplantation using tacrolimus-based protocols or cyclosporine-based quadruple immunosuppression with an interleukin-2 receptor antibody or antithymocyte globulin. Cancer. 1997;80(6):1141–1150. | ||
Ellis D, Jaffe R, Green M, et al. Epstein–Barr virus-related disorders in children undergoing renal transplantation with tacrolimus-based immunosuppression. Transplantation. 1999;68(7):997–1003. | ||
Otley CC, Pittelkow MR. Skin cancer in liver transplant recipients. Liver Transpl. 2000;6(3):253–262. | ||
Shapiro R, Nalesnik M, McCauley J, et al. Posttransplant lymphoproliferative disorders in adult and pediatric renal transplant patients receiving tacrolimus-based immunosuppression. Transplantation. 1999;68(12):1851–1854. | ||
Kalthoff FS, Winiski A, Fichtinger P, et al. Differential inhibition of primary versus preactivated T cells by pimecrolimus but not by tacrolimus in vitro. Int Arch Allergy Immunol. 2007;142(3):255–264. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.






