Back to Journals » International Journal of Nanomedicine » Volume 9 » Supplement 2
A 90-day study of sub-chronic oral toxicity of 20 nm positively charged zinc oxide nanoparticles in Sprague Dawley rats
Authors Park H, Kim S, Lee T, Kim G, Meang E, Hong J, Kim S, Koh S, Hong S, Sun Y, Kang JS, Kim Y, Kim M, Jeong J, Lee JK, Son W, Park J, An SSA
Received 20 November 2013
Accepted for publication 12 June 2014
Published 15 December 2014 Volume 2014:9(Supplement 2) Pages 93—107
DOI https://doi.org/10.2147/IJN.S57927
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Hark-Soo Park,1 Seon-Ju Kim,1 Taek-Jin Lee,1 Geon-Yong Kim,1 EunHo Meang,1 Jeong-Sup Hong,1 Su-Hyon Kim,1 Sang-Bum Koh,1 Seung-Guk Hong,1 Yle-Shik Sun,1 Jin Seok Kang,2 Yu-Ri Kim,3 Meyoung-Kon Kim,3 Jayoung Jeong,4 Jong-Kwon Lee,4 Woo-Chan Son,5 Jae-Hak Park6
1General Toxicology Team, Korea Testing and Research Institute, Seoul, 2Department of Biomedical Laboratory Science, Namseoul University, Cheonan, 3Department of Biochemistry and Molecular Biology, Korea University Medical School and College, Seoul, 4National Institute of Food and Drug Safety Evaluation, Seoul, 5Department of Pathology, University of Ulsan College of Medicine, Asan Medical Center, Seoul, 6Laboratory Animal Medicine, College of Veterinary Medicine, Seoul National University, Seoul, Korea
Purpose: The study reported here was conducted to determine the systemic oral toxicity and to find the no-observed-adverse-effect level of 20 nm positively charged zinc oxide (ZnOSM,20(+)) nanoparticles in Sprague Dawley rats for 90 days.
Methods: For the 90-day toxicity study, the high dose was set as 500 mg per kg of body weight (mg/kg) and the middle and low dose were set to 250 mg/kg and 125 mg/kg, respectively. The rats were held for a 14-day recovery period after the last administration, to observe for the persistence or reduction of any toxic effects. A distributional study was also carried out for the systemic distribution of ZnOSM,20(+) NPs.
Results: No rats died during the test period. There were no significant clinical changes due to the test article during the experimental period in functional assessment, body weight, food and water consumption, ophthalmological testing, urine analysis, necropsy findings, or organ weights, but salivation was observed immediately after administration in both sexes. The total red blood cell count was increased, and hematocrit, albumin, mean cell volume, mean cell hemoglobin, and mean cell hemoglobin concentration were decreased significantly compared with control in both 500 mg/kg groups. Total protein and albumin levels were decreased significantly in both sexes in the 250 and 500 mg/kg groups. Histopathological studies revealed acinar cell apoptosis in the pancreas, inflammation and edema in stomach mucosa, and retinal atrophy of the eye in the 500 mg/kg group.
Conclusion: There were significant parameter changes in terms of anemia in the hematological and blood chemical analyses in the 250 and 500 mg/kg groups. The significant toxic change was observed to be below 125 mg/kg, so the no-observed-adverse-effect level was not determined, but the lowest-observed-adverse-effect level was considered to be 125 mg/kg in both sexes and the target organs were found to be the pancreas, eye, and stomach.
Keywords: positive charge, oral-toxicity study, no-observed-adverse-effect level, ZnO
Introduction
In recent years, the use of nanotechnology in medicine and health, and in cosmetic, clothing, personal-care, sporting-good, and sunscreen products has been a hot issue.1 Zinc oxide (ZnO) nanoparticles (NPs) are being used in personal-care products, as coatings, and paint, and concerns about the potential risks of NPs to the human body are continuing.2 One popular NP in the modern cosmetics industry is ZnO, which is used in sunscreen products.3 NPs have interesting physical properties that make them ideal for use in skin-care cosmetics.4 ZnO NPs have been also used in fabric, because when NPs are used in a coating agent, they confer UV protection.5 Nanotechnology is also being applied widely in the food industry, for example, in packaging, delivery, formulation, evaluation, and food processing.6 Other potential applications of novel NPs are in medicine, predominantly in detection, diagnostics, monitoring, and therapeutics.7 However, there are potential risks concerning toxicity and environmental impacts associated with nanotechnology. Many studies have shown the toxicity of ZnO NPs in various studies with cell lines, bacteria, nematodes, algae, plants, and fish.8 Twenty-nanometer ZnO was shown to have the greatest cytotoxic effect on human fetal lung fibroblast cells compared with other metallic NPs (ZnO, TiO2, SiO2, and Al2O3).9 In other research, ZnO NPs were found to have cytotoxic effects on human glomerular and tubular renal culture cells that correlate with the physicochemical properties and metal composition of the NPs.10 The initial transient exposure of cells to oxidative stress showed the cytotoxicity of ZnO NPs and initial exposure of BEAS-2B cells to oxidative stress sensitized their subsequent response to cytotoxic challenge with ZnO NPs.8 Recently, the induction of cellular oxidative stress mediated by free radical and reactive oxygen species (ROS) was shown to increase the cytotoxicity of ZnO.11,12 ZnO NPs were also revealed to cause DNA damage and apoptosis in mouse liver cells through the generation of ROS triggered by the mitochondrial pathway, which then induced the cytotoxic and genotoxic effects of the ZnO NPs.13 Other researchers demonstrated that ZnO NPs induces apoptosis in cancer cells via the tumor suppressor p53 pathway.14 Also, ZnO NPs have the potential to induce cell death and apoptosis in human dermal fibroblasts via the mitogen-activated protein kinase p38 and cell-cycle checkpoint protein p53 pathways.15
The cytotoxicity of NPs can be varied by the addition of different NP coatings with different physicochemical properties. It was shown that uncoated ZnO NPs increased ROS accumulation and diminished cell viability, but that all coated ZnO NPs reduced ROS accumulation and cytotoxicity. However, poly(methacrylic acid)-coated ZnO NPs had significant genotoxicity compared with uncoated ZnO NPs and other coated ZnO NPs.16 In in vivo studies, a recent study showed that the oral administration of ZnO NPs for 14 days to Sprague Dawley (SD) rats induced lesions in the liver, pancreas, heart, and stomach.17,18 In addition, a nanoparticulate extract derived from Brassica rapa (turnip) was recently shown to exert anti-hepatofibrogenic effects in the liver.19
For these reasons, we conducted a novel, 90-day, repeated-dose, sub-chronic oral-toxicity investigation of ZnOSM20(+) NPs in SD rats to ascertain their systemic toxicity and no-observed-adverse-effect level or lowest-observed-adverse-effect level in vivo.
Materials and methods
Test and control materials
ZnO NPs (ZnO-310 ultrafine zinc oxide) were purchased from the Sumitomo Osaka Cement Co, Ltd (Tokyo, Japan). In a previous study by our group, the crystalline structure and the size of the ZnO NPs were analyzed by X-ray diffraction and Fourier transform infrared spectroscopy; the average diameter was 29±3 nm in deionized water.20 The vehicle control was 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES)-serine buffer (1M Na2CO3 [MW =105.99], 20 mM HEPES buffer [MW =238.3], and L-Serine). The negative control was distilled water (Daehan New Pharm Co, Ltd, Hwaseong, Korea).
ZnOSM20(+) NP preparation
To obtain a positive charge, surface-charge modification was performed with serine, as previously reported.20 To produce the HEPES-serine buffer, HEPES buffer solution was prepared and pH-adjusted to 6 using 1M Na2CO3. To this, 1 wt/v% L-Serine (Sigma-Aldrich, St Louis, MO, USA) was added. The test article was prepared, with HEPES buffer solution, in high dose (500 mg/mL), middle dose (250 mg/mL), low dose (125 mg/mL) formulations. For the high 500 mg/mL dose, the test material was weighed and HEPES-serine buffer added. The middle (250 mg/mL) and low (125 mg/mL) doses were diluted by suspending the modified ZnO NPs in sterile distilled water. Preparation of the test substance for each group for the treatment period was carried out every day.
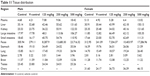
The stability and homogeneity of ZnOSM20(+) were confirmed using the validation and verification of the concentration of the formulation method outlined in Korea Testing and Research Institute (KTR) study number TBH-1367. The concentration of each preparation was measured at 1 and 45 days and 90 days prior to administration; all the preparations were appropriate within 100±15% (Table 1).
 
|
Table 1 Results of dose formulation analysis in the 90-day oral-toxicity study of zinc oxide (ZnOSM20(+)) nanoparticles |
Test animals and experimental system
Five-week-old male and female specific-pathogen-free SD rats were purchased from Orient Bio Co, Ltd (Seongnam, Korea) and acclimated for 7 days before the treatment. During the acclimation and experimental periods, the rats were housed in wire cages (maximum of two rats per cage) in a room with controlled temperature (22°C±3°C) and humidity (50%±20%), and a 12-hour light–dark cycle. The rats were fed a gamma-ray-irradiated rodent diet (Cargill Agri Purina Korea Inc, Seongnam, Korea) and filtered water ad libitum. The rats were divided into five groups (ten rats in each group, an additional five recovery animals in each of the negative, HEPES-serine, and high-dose groups, and two for an organ-distribution study): negative control (distilled water), HEPES-serine buffer vehicle, low dose, middle dose, and high dose. The distilled water, HEPES-serine, and NP solutions were administered to the rats by gavage once a daily.
The dose levels were determined based on the results of the dose-finding studies for 14-day repeated oral-toxicity studies (KTR study number: TBH-1092). The significant adverse effects were observed at dose levels of 2,000 and 1,000 mg/kg; therefore, 500 mg/kg was selected as the high dose for the high-dose group, and then the middle- and low-dose groups were set up by the twofold intervals as 250 mg/kg and 125 mg/kg, respectively.
The control group was treated with HEPES-serine and the negative control group was treated with sterile distilled water. In addition, to assess the persistence and reversibility of toxicity in the negative control, vehicle control, and high-dose groups, a recovery group was added. The recovery period was 2 weeks after the end of the treatment for more observations.
The dosages used in the organ-distribution study were identical to those employed in the primary oral-toxicity study – details of the study design are summarized in Table 2. The studies were conducted according to the OECD Principles of Good Laboratory Practice and approved by the Institutional Animal Care and Use Committee.
Animal observation
General symptoms, presence of toxic symptoms, and death were observed once a day after administration of the solutions during the testing period for all animals. Main studies for all animals, clonic and tonic movements, repetitive behavior (hyperactive grooming, circling) or abnormal behavior, aggression, and motor coordination, gait, and posture and handling changes were observed once per week.
Assessment of sensory reflexes and motor activity
The sensory-response assessment for all animals in the primary oral-toxicity study was completed at the end of the 90-day experimental period by assessing grasp response; touch escape; vocalization; pupil reflex; blink reflex; and response to toe pinch, tail pinch, and finger approach. Grip strength and motor activity were tested in the negative and vehicle control groups and the high-dose group at the end of the experimental period (n=5 animals per group). Grip strength in the front and hind limbs/feet was determined by using a rat grip-strength measurement system (1027 CSX Grip Strength Meter, Columbus Instruments, Columbus, OH, USA). Grip strength was measured three times and averaged. A motor-activity monitoring system (Columbus Instruments) and Truscan 99 data acquisition software (version 2.0; Deymed Diagnostic, Payette, ID, USA) were used to test motor activity.
Measurement of food intake, water consumption, and body weight
Feed and water intake were measured once a week after the initiation of treatment, and the average intake (g/rat/day; n=5 animals per group) was calculated. Body weight was measured at animal acquisition, grouping, and once a week after the initiation of treatment.
Urinalysis
Urine samples were analyzed for nitrite, protein, glucose, ketone, urobilinogen, and bilirubin levels, specific gravity, pH, leukocyte count, and the presence of blood during the last week of the study by using a Clinitek 500 Urine Analyzer with a Multistix 10SG Urine Strip (Bayer, Morristown, NJ, USA) (n=5 animals per group). Fresh urine for the analysis was collected over a 3-hour period using metabolic cages in the negative, vehicle control, and high-dose groups (n=5 animals per group), followed by microscopic examination of the sediment. Urinary volume was measured by collecting the urine over a 24-hour period.
Ophthalmology examination
The visual appearance of the eye was inspected in the vehicle, negative, and high-dose groups before grouping and during the final week of the 90-day oral-toxicity study (n=5 animals per group). In addition, funduscopy was performed through the dilated pupil after dripping mydriatic fluid (Ocu-Tropine, Samil Pharmaceutical Co, Ltd, Seoul, Korea) into the eye with aid of a fundus camera (Genesis, Kowa, Japan).
Hematology and serum biochemistry
The blood was withdrawn from the abdominal artery in animals fasted for 18 hours and anesthetized with isoflurane. The collected blood was divided into three portions: 3 mL for hematological analysis in EDTA (ethylenediaminetetraacetic acid) complete-blood-count tubes (EDTA 3K from BD, Franklin Lakes, NJ, USA), 2.7 mL for blood clotting in sodium citrate-containing Vacutainer tubes (sodium citrate 3.2%; BD), and the rest for serum biochemistry in plain tubes.
Hematology was analyzed with an automated cell counter (ADVIA® 120 Hematology System, Seimens, Munich, Germany) for the following items: total leukocyte count, differential leukocyte count, total erythrocyte count, hemoglobin concentration (HGB), hematocrit (Hct), mean cell volume, mean cell hemoglobin, mean cell hemoglobin concentration, reticulocyte, platelet.
Blood-clotting time was measured in plasma obtained by centrifuge (3,000 rpm, 10 minutes) with an automatic analyzer (ACL 7000, Werfen Group Instrumentation Laboratory, Bedford, MA, USA). Serum biochemistry was measured in serum after clotting in room temperature then separating by centrifuge (3,000 rpm, 10 minutes) using an automatic serum analyzer (Hitachi 7060, Hitachi, Tokyo, Japan) for total protein, albumin (Alb), Alb/globulin ratio, total bilirubin, alkaline phosphatase, aspartate aminotransferase, alanine aminotransferase, creatinine, blood urea nitrogen, total cholesterol, triglycerides, glucose, calcium, inorganic phosphorus, and creatine kinase. We used an electrolyte analyzer (EasyLyte Plus Na/K/Cl Analyzer, Medica, MA, USA) for analysis of sodium, potassium, and chloride in the same serum.
Necroscopy, organ weights, and histopathology
Gross examination of the external body surface, all orifices, cranial cavity, all organs of the chest cavity, and the abdominal cavity was performed.
The liver, kidney, spleen, adrenal gland, testis, ovaries, brain, pituitary gland, lung, heart, thymus, uterus, prostate, epididymis, submaxillary gland were weighed for their wet weight then the relative organ-weight-to-body-weight ratio was calculated.
The liver, kidney, adrenal gland, heart, lung, brain, pituitary gland, seminal vesicle, spleen, testis, ovaries, epididymis, prostate gland, uterus, vagina, tongue, trachea, esophagus, thymus, thyroid gland, stomach, duodenum, urinary bladder, small intestine, large intestine, eyeball, submandibular gland, pancreas, skin, mammary gland, sternum, femur, spinal cord, sciatic nerve, and mesenteric lymph node were removed and fixed in 10% neutral buffered formalin solution; eyes were fixed in Davidson solution; and the testis and epididymis were fixed in Bouin solution. Histopathologic examination of the control and high-dose groups only was undertaken. However, study of the pancreas, stomach, and eyes was performed for all groups.
Distribution of ZnOSM20(+) NPs in plasma, tissue, and feces
Prior to necroscopy, a 1 mL blood sample was obtained from the tail vein and some stool samples were collected. Samples from brain, liver, kidney, testis, ovary, spleen, lung, stomach, small intestine, and large intestine were collected after the removal of fat and the weights of these measured.The distribution of the ZnOSM20(+) NPs was analyzed on the basis of Zn content in each organ using an ULTIMA2 inductively coupled plasma atomic absorption spectrometer (Horiba, Kyoto, Japan).
Statistical analysis
To evaluate the homogeneity of variance, Levene’s tests were performed, and one-way analysis of variance was undertaken to determine significance in terms of the body weight, feed intake, water consumption, organ weight, and hematological and blood biochemical data. If the data had homogeneous variances and significant differences between treatment groups statistically, Scheffé’s test was conducted as the post hoc test. If the data had heterogeneous variance and significant differences were found between groups, Dunnett’s T3 was performed as the post hoc test. All statistical analysis was performed using SPSS software (v 19.0; IBM Corporation, Armonk, NY, USA).
Results
General observations
There were no moribund or dead animals related to the administration of the test article during the experimental period in either sex. In terms of clinical signs, salivation was observed immediately after administration in both sexes. The salivation was noted firstly in the 500 mg/kg group on Day 11 and reported in all NP-treated groups from that time forward. A wound, fur loss, and red tears were observed once each in each NP treatment group.
Sensor reflex and motor activity
Treatment-related abnormal behavior and functionality were not observed when behavior, sensory reflex, grip strength, and motor activity were assessed.
Effect of ZnOSM20(+) NPs on food intake, water consumption, and body weight
Body-weight changes were not shown to be statistically significant in the study groups compared with in the control group (Figure 1). Food consumption was decreased significantly in males in the 500 mg/kg group compared with in the control group. Water consumption was decreased significantly in males in the 500 mg/kg group compared with in the control groups from 5 to 11 weeks after administration (Tables 3 and 4).
Urine and ophthalmology
There were no toxicological changes related to the administration of the test articles in terms of urine and ophthalmology.
Necropsy and organ weight
Malformation of genitalia or atrophy of the seminal vesicle was observed in a single male, and lung discoloration was observed in a female receiving 500 mg/kg. These were considered not to be related to treatment, as they were low in incidence and there was a lack of a dose relationship compared with the background data kept in our facility.
In terms of absolute organ weight, the weight of the heart was decreased significantly in males in the 500 mg/kg group and the weight of submaxillary glands was increased significantly, compared with controls. In terms of relative organ weight, there were no statistical differences between the treated and control groups in either sex. However, the weights of the submaxillary gland, kidney, and epididymis were increased significantly in males in the recovery group (500 mg/kg) compared with control (Tables 5 and 6).
Effects of ZnOSM20(+) NPs on clinical chemistry and hematology
In the case of males, HGB, Hct, mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), and mean corpuscular hemoglobin concentration (MCHC) decreased (P<0.05) in the 500 mg/kg group compared with in the control group. Total leukocyte and total red blood cell (RBC) levels were significantly (P<0.05) increased in the 500 mg/kg group compared with in the control group. HGB, MCV, MCH, and MCHC were significantly (P<0.05 or P<0.01) decreased in females in the 500 mg/kg group. These decreased HGB, MCV, MCH, and MCHC levels were not recovered to negative-control level in the recovery group (500 mg/kg group). Total leukocyte and total RBC levels in the recovery group were significantly (P<0.05) increased in the 500 mg/kg group compared with in the control group for both sexes (Tables 7 and 8).
Total protein and Alb levels decreased (P<0.05) in males and females in the 250 and 500 mg/kg groups compared with in males and females in the control groups. Creatine kinase levels were significantly (P<0.05) increased in males in the 500 mg/kg group. In the case of females, alkaline phosphatase levels were significantly (P<0.05) increased in the 500 mg/kg group (Table 9).
Histopathological examination
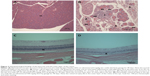
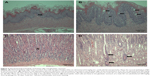
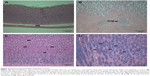
Acinar cell apoptosis and chronic inflammation of the pancreas in all groups were observed. Two cases of retinal atrophy were found in both the male recovery and 500 mg/kg groups, while an additional three cases were also identified in the female recovery group (Figure 2). In all treatment groups, various gastric lesions of different grades were observed. Therefore, we examined these lesions, classifying them as non-glandular stomach and glandular stomach lesions. In all males and females in all groups, squamous cell hyperplasia, squamous cell vacuolation, and subepithelial inflammatory cell infiltration in non-glandular stomach and submucosal inflammatory cells infiltration, superficial epithelial degeneration/regeneration, intracytoplasmic hyaline droplet, mucous cell hyperplasia, and eosinophilic chief cells in glandular stomach were observed (Figures 3 and 4). The lesions observed in the pancreas and stomach disappeared in the recovery group, and we considered that most lesions recovered due to the interruption of test-article administration. Also in the 500mg/kg male and female recovery group, eye retinal atrophy was of the same grade of lesion as those in main study (Table 10).
Distribution of ZnOSM20(+) NPs in the plasma, organs, and feces
Plasma, stools, brain, liver, kidney, testis, ovary, spleen, lung, stomach, small intestine, and large intestine were analyzed with proven methods. There was no differences in organ distribution in terms of sex. ZnO concentration was increased in the liver, kidney, intestine, plasma, and lung in the study groups compared with in the negative control and vehicle control groups. The test article was absorbed by and accumulated in those organs and excreted in the feces. However, NPs were neither detected nor found to be increased in the brain, testis, ovary, spleen and stomach of study-group animals compared with control-group animals (Table 11).
|
Table 11 Tissue distribution |
Discussion
There were no dead animals related to administration of the test article during the experimental period of either sex. There were no significant toxicological changes in the study animals compared with control animals, of either sex, in terms of functional assessment tests, changes in body weight, food and water consumption, urine analysis, ophthalmological tests, necropsy findings, or organ weights. In terms of clinical signs, salivation was observed immediately after administration in both sexes. Salivation was noted firstly in the 500 mg/kg group on Day 11 and reported in all groups from that point forward. This sign occurred due to test-article administration and was related to inhalation of the ZnO.21
Hematological analysis revealed that the total RBC count was increased, and Hct, MCV, MCH, and MCHC were decreased significantly compared with controls in both sexes in the 500 mg/kg group. This concurs with the test-article administration and has been reported to be ZnO-ion related, thus induced iron-deficiency and sideroblastic anemia.22,23 Anemia-related reports have shown that a high Zn concentration resulting from a ZnO NP diet might cause an iron-deficiency anemia.24,25
Retinal atrophy in the eye was observed in males in the 500 mg/kg group of the main study. Two cases in males and three cases in females were observed in the recovery group, therefore, these lesions were considered treatment related. Zn induced retinal damage when it was administrated in high concentrations, but, in another other study, a low concentration of zinc was shown to prevent retinal lesion in ischaemia.26
In all treatment groups, various kinds of gastric inflammatory and degenerative lesions with regeneration were observed. These lesions were stronger than in our former study with negatively charged ZnOSM20(−) NPs. Recently, it was reported that 20 nm and 120 nm ZnO particles caused inflammation of the gastric lamina propria and submucosal layer of the stomach in mice.18,27 When 20 nm ZnO was administrated orally during 14 days, mild inflammatory cell infiltration lesions in the liver and pancreas with severe stomach mucosal damage were observed.18 The smaller size of ZnO NPs produced more toxic lesions in the pancreas.17
The absorption and accumulation of Zn increased with dose increment in liver, kidney, intestine, plasma, and lung, while there was little or no increase in these in the brain, testis, ovary, spleen, and stomach. The difference in accumulation occurred in plasma and lung in our previous study with ZnOSM20(−)NPs.28 Also in our previous study, Zn concentrations dose-dependently increased in the liver, kidney, intestine, and plasma of the experimental compared with control groups. The ZnOSM20(−) NPs were also dose-dependently excreted into the feces, as evidenced by high Zn levels. On the other hand, little or no increase in Zn concentration was found in the brain, testis, ovary, spleen, stomach (except in females in the 500 mg/kg group), or lung.28
In our previous study, ZnOSM20(−) induced degenerative lesions in the pancreas and stomach, irreversible retinal atrophy in the eye, and changes of anemia-related factors.28 Similar results in terms of these pathologic and anemic effects were obtained in the current study. The difference between ZnOSM20(−) and ZnOSM20(+) histopathologic lesions has not been identified yet, so our future studies will look at identifying the difference in toxicity between the two differently charged ZnO NPs.
Conclusion
Based on the results, under the conditions of the study in which doses of 125, 250, and 500 mg/kg of positively charged ZnOSM20(+) NPs were repeatedly administered by gavage for 90 days in SD rats, the adverse effects induced by ZnOSM20(+) NPs were salivation after administration, acinar cell apoptosis, submucosal edema and inflammation in glandular stomach, retinal atrophy of the eye in the 500 mg/kg group, and a significant decreased hematological parameter associated with anemia in the 250 and 500 mg/kg groups.
Among them, the changes in the pancreas, glandular stomach, and eye were considered to be the primary adverse effect. The result of the charge difference on the accumulation of NPs was shown in lung and plasma. Therefore, the no-observed-adverse-effect level was not determined, but the lowest-observed-adverse-effect level was considered to be under 125 mg/kg in both sexes. The target organs were considered to be the stomach, pancreas, and eye.
Acknowledgments
This study was supported by a grant (10182MFDS991) from Ministry of Food and Drug Safety in 2010 and by the Research Institute for Veterinary Science, College of Veterinary Medicine, Seoul National University, Korea.
Disclosure
The authors report no conflicts of interest in this work.
References
Nohynek GJ, Lademann J, Ribaud C, Roberts MS. Grey goo on the skin? Nanotechnology, cosmetic and sunscreen safety. Crit Rev Toxicol. 2007;37(3):251–277. | ||
Klaine SJ, Alvarez PJ, Batley GE, et al. Nanomaterials in the environment: behavior, fate, bioavailability, and effects. Environ Toxicol Chem. 2008;27(9):1825–1851. | ||
Osmond MJ, McCall MJ. Zinc oxide nanoparticles in modern sunscreens: an analysis of potential exposure and hazard. Nanotoxicology.2010;4(1):15–41. | ||
Wiesenthal A, Hunter L, Wang S, Wickliffe J, Wilkerson M. Nanoparticles: small and mighty. Int J Dermatol. 2011;50(3):247–254. | ||
Becheri A, Dürr M, Nostro PL, Baglioni P. Synthesis and characterization of zinc oxide nanoparticles: application to textiles as UV-absorbers. J Nanopart Res. 2008;10(4):679–689. | ||
Rashidi L, Khosravi-Darani K. The applications of nanotechnology in food industry. Crit Rev Food Sci Nutr. 2011;51(8):723–730. | ||
Logothetidis S. Nanotechnology in medicine: the medicine of tomorrow and nanomedicine. Hippokratia. 2006;10(1):7–21. | ||
Heng BC, Zhao X, Xiong S, Ng KW, Boey FY, Loo JS. Toxicity of zinc oxide (ZnO) nanoparticles on human bronchial epithelial cells (BEAS-2B) is accentuated by oxidative stress. Food Chem Toxicol. 2010;48(6):1762–1766. | ||
Zhang XQ, Yin LH, Tang M, Pu YP. ZnO, TiO(2), SiO(2,) and Al(2)O(3) nanoparticles-induced toxic effects on human fetal lung fibroblasts. Biomed Environ Sci. 2011;24(6):661–669. | ||
Pujalté I, Passagne I, Brouillaud B, et al. Cytotoxicity and oxidative stress induced by different metallic nanoparticles on human kidney cells. Part Fibre Toxicol. 2011;8:10. | ||
Fan Z, Lu JG. Zinc oxide nanostructures: synthesis and properties. J Nanosci Nanotechnol. 2005;5(10):1561–1573. | ||
Xia T, Kovochich M, Liong M, et al. Comparison of the mechanism of toxicity of zinc oxide and cerium oxide nanoparticles based on dissolution and oxidative stress properties. ACS Nano. 2008;2(10):2121–2134. | ||
Sharma V, Singh P, Pandey AK, Dhawan A. Induction of oxidative stress, DNA damage and apoptosis in mouse liver after sub-acute oral exposure to zinc oxide nanoparticles. Mutat Res. 2012;745(1–2):84–91. | ||
Akhtar MJ, Ahamed M, Kumar S, Khan MM, Ahmad J, Alrokayan SA. Zinc oxide nanoparticles selectively induce apoptosis in human cancer cells through reactive oxygen species. Int J Nanomedicine. 2012;7:845–857. | ||
Meyer K, Rajanahalli P, Ahamed M, Rowe JJ, Hong Y. ZnO nanoparticles induce apoptosis in human dermal fibroblasts via p53 and p38 pathways. Toxicol In Vitro. 2011;25(8):1721–1726. | ||
Yin H, Casey PS, McCall MJ, Fenech M. Effects of surface chemistry on cytotoxicity, genotoxicity, and the generation of reactive oxygen species induced by ZnO nanoparticles. Langmuir. 2010;26(19):15399–15408. | ||
Pasupuleti S, Alapati S, Ganapathy S, Anumolu G, Pully NR, Prakhya BM. Toxicity of zinc oxide nanoparticles through oral route. Toxicol Ind Health. 2012;28(8):675–686. | ||
Wang B, Feng W, Wang M, et al. Acute toxicological impact of nano- and submicro-scaled zinc oxide powder on healthy adult mice. J Nanopart Res. 2007;10(2):263–276. | ||
Park DH, Li L, Jang HK, et al. Biological safety and anti-hepatofibrogenic effects of Brassica rapa (turnip) nanoparticle. Molecular and Cellular Toxicology. 2009;5(4):317–322. | ||
Kim KM, Kim TH, Kim HM, et al. Colloidal behaviors of ZnO nanoparticles in various aqueous media. Toxicol Environ Health Sci. 2012;4(2):121–131. | ||
Cooper RG. Zinc toxicology following particulate inhalation. Indian J Occup Environ Med. 2008;12(1):10–13. | ||
Plum LM, Rink L, Haase H. The essential toxin: impact of zinc on human health. Int J Environ Res Public Health. 2010;7(4):1342–1365. | ||
Broun ER, Greist A, Tricot G, Hoffman R. Excessive zinc ingestion. A reversible cause of sideroblastic anemia and bone marrow depression. JAMA. 1990;264(11):1441–1443. | ||
Hoffman HN 2nd, Phyliky RL, Fleming CR. Zinc-induced copper deficiency. Gastroenterology. 1988;94(2):508–512. | ||
Latimer KS, Jain AV, Inglesby HB, Clarkson WD, Johnson GB. Zinc-induced hemolytic anemia caused by ingestion of pennies by a pup. J Am Vet Med Assoc. 1989;195(1):77–80. | ||
Ugarte M, Osborne NN. Zinc in the retina. Prog Neurobiol. 2001;64(3):219–249. | ||
Zhao Y, Wang B, Feng W, Bai C. Nanotoxicology: toxicological and biological activities of nanomaterials. In: United Nations Educational, Scientific and Cultural Organization Encyclopedia of Life Support Systems (UNESCO-EOLSS) [online encyclopedia]. Oxford: EOLSS Publishers; 2010. Available from: www.eolss.net/sample-chapters/c05/e6-152-35-00.pdf. Accessed September 22, 2014. | ||
Park HS, Kim SJ, Lee TJ, et al. A 90-day study of subchronic oral toxicity of 20-nm, negatively-charged zinc oxide nanoparticles in Sprague Dawley rats. Int J Nanomedicine. In press. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.