Back to Journals » Infection and Drug Resistance » Volume 7
Staphylococcus aureus – antimicrobial resistance and the immunocompromised child
Authors McNeil JC
Received 16 January 2014
Accepted for publication 19 March 2014
Published 7 May 2014 Volume 2014:7 Pages 117—127
DOI https://doi.org/10.2147/IDR.S39639
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
J Chase McNeil
Department of Pediatrics, Section of Infectious Diseases, Baylor College of Medicine, Houston, TX, USA
Abstract: Children with immunocompromising conditions represent a unique group for the acquisition of antimicrobial resistant infections due to their frequent encounters with the health care system, need for empiric antimicrobials, and immune dysfunction. These infections are further complicated in that there is a relative paucity of literature on the clinical features and management of Staphylococcus aureus infections in immunocompromised children. The available literature on the clinical features, antimicrobial susceptibility, and management of S. aureus infections in immunocompromised children is reviewed. S. aureus infections in children with human immunodeficiency virus (HIV) are associated with higher HIV viral loads and a greater degree of CD4 T-cell suppression. In addition, staphylococcal infections in children with HIV often exhibit a multidrug resistant phenotype. Children with cancer have a high rate of S. aureus bacteremia and associated complications. Increased tolerance to antiseptics among staphylococcal isolates from pediatric oncology patients is an emerging area of research. The incidence of S. aureus infections among pediatric solid organ transplant recipients varies considerably by the organ transplanted; in general however, staphylococci figure prominently among infections in the early posttransplant period. Staphylococcal infections are also prominent pathogens among children with a number of immunodeficiencies, notably chronic granulomatous disease. Significant gaps in knowledge exist regarding the epidemiology and management of S. aureus infection in these vulnerable children.
Keywords: pediatric, HIV, cancer, transplant
Staphylococcus aureus – antimicrobial resistance and the immunocompromised child
Staphylococcus aureus is among the most common pathogens encountered in pediatric practice and is the most common cause of bacterial skin-and-soft tissue infection (SSTI).1 While SSTIs are the most common manifestation, S. aureus also is responsible for a wide spectrum of invasive infections including musculoskeletal infections, complicated pneumonia, and endocarditis. The emergence of community-acquired methicillin-resistant S. aureus (CA-MRSA) led to an evolution of the severity of disease with an increase in invasive infections in some series.2–5 Studies of adults have shown that immunosuppression is associated with increased risk of S. aureus colonization, putting these fragile patients at risk for infection and further morbidity.6
Children with immunocompromising conditions represent a unique group for the acquisition of antimicrobial resistant infections due to their frequent encounters with the health care system, need for empiric antimicrobials, and immune dysfunction. While most pediatricians are familiar with the treatment of staphylococcal infections, these vulnerable children pose clinical challenges as a result of poor immune function, frequent antibiotic resistance, and the presence of medical devices and catheters that circumvent normal anatomic barriers. These infections are further complicated in that there is a relative paucity of literature on the clinical features and management of S. aureus infections in immunocompromised children, and clinical decisions must be drawn from the very limited available pediatric data or extrapolated from studies of adults. This review seeks to provide an overview of the published literature on the epidemiology, clinical features, antimicrobial susceptibility, and treatment of S. aureus infections in immunocompromised children.
Overall epidemiology of pediatric S. aureus infections
The past two decades have seen an enormous increase in the incidence of CA-MRSA infections.7 Studies from the 1990s and early 2000s revealed that CA-MRSA infections differed from typical health care-associated MRSA (HA-MRSA) in that CA-MRSA infections more often occurred in otherwise healthy patients without typical risk factors for antibiotic resistant infections, including children.8,9 In addition, CA-MRSA was more often susceptible to non-β-lactam antimicrobials, carried genes for Panton–Valentine leukocidin (PVL), were staphylococcal chromosome cassette mec type IV, and associated with soft-tissue rather than invasive infections compared to HA-MRSA.8 In the 2000s, the USA300 pulsed field gel type emerged as the predominant genotype of CA-MRSA in North America, accounting for anywhere from 50%–97% of isolates.1,10–12 During this time, USA300 also emerged as an important cause of HA-MRSA in both children and adults in some centers,13–15 thus blurring the distinction between typical CA- and HA-MRSA. Furthermore, some centers have noted an increase in the proportion of methicillin-susceptible S. aureus (MSSA) isolates possessing the USA300 genetic background.16 This is of further import in that the increase in the USA300 clone has been temporally associated with an increase in invasive infections in children, including complicated pneumonia, osteoarticular infections, and pyomyositis.2–5
S. aureus and immune evasion
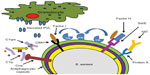
S. aureus possesses a number of mechanisms to evade immune destruction in the healthy host (Figure 1). Among the most notable of these mechanisms include capsule, staphylococcal surface proteins and PVL. The majority of S. aureus possesses a carbohydrate capsule of one of eleven serotypes, the most common and best studied of which are capsular serotypes 5 and 8. Capsule plays a role in inhibiting phagocytosis by granulocytes as well as promoting adherence to surfaces.17 In large part because of the presence of capsule, opsonization is required for the enhanced phagocytosis of staphylococci; other means exist for S. aureus to avoid opsonization. Staphylococcal protein A, expressed on the cell surface, provides a defense mechanism to staphylococci through binding to the Fc region of immunoglobulin G. This results in an inappropriate orientation of the immunoglobulin for opsonophagocytosis and complement activation.18 Collagen adhesin (Cna) is a S. aureus protein involved in adhesion to extracellular collagen; however, it also has a role in immune evasion. Cna binds to C1q preventing the formation of the C1qrs complex and creating a blockade of the classical complement pathway.18 Staphylococcal clumping factor A (ClfA), in addition to binding fibrinogen, facilitates the destruction of the complement component C3b. ClfA interacts with complement regulatory factor I, promoting the conversion of C3 to the inactivated iC3b, thus decreasing complement-mediated phagocytosis.19,20 An additional S. aureus surface protein, SdrE, binds complement factor H and promotes factor I-mediated breakdown of C3b, resulting in less C3b deposition on the organism.21 This is particularly notable in that C3 is a common mediator of the classical, alternative, and lectin-mediated complement pathways.
While S. aureus elaborates a wide array of extracellular toxins, PVL is the only known toxin that specifically targets leukocytes.22 PVL is a bicomponent toxin encoded by the genes luks-PV and lukf-PV and produces pores in leukocytes, resulting in cell death.23 There is a clinical association between infection with PVL-positive S. aureus and severity of disease;24 however, the exact role that PVL plays in pathogenesis has been conflicting in animal models. Taken together, S. aureus exhibits a number of tools for evading the immune system, which can only be accentuated in the immunocompromised host.
Children with human immunodeficiency virus/acquired immunodeficiency syndrome
Research among adults with human immunodeficiency virus (HIV) has revealed higher rates of colonization with S. aureus than the general population, reaching as high as 81% during a 1-year study period.25 In addition, HIV-positive adults are known to more often have invasive S. aureus infections, notably bacteremia, than HIV-negative controls.26
Furthermore, HIV-positive adults and children often are infected with S. aureus isolates exhibiting a multidrug-resistant (MDR) phenotype.27 The MDR-MRSA isolates often exhibit resistance to clindamycin, macrolides, ciprofloxacin, tetracyclines, and mupirocin. Two of the resistance genes in these MDR isolates, ermC and mupA, are carried on a large conjugative plasmid known as pUSA03.28 S. aureus infections in HIV-positive children have high rates of antimicrobial resistance, with over 82% and 40% of isolates resistant to methicillin and clindamycin, respectively, in one small single center study of 28 cases.29 Furthermore, the isolates were also resistant to ciprofloxacin in 47.6% of cases consistent with a MDR phenotype. Resistance to trimethoprim-sulfamethoxazole (TMP-SMX) in these staphylococcal isolates is rather uncommon in the United States (3%), even in the presence of chronic TMP-SMX prophylaxis. In a series from Houston, TX, 19 of 21 (90.5%) isolates available from children with HIV were of the USA300 pulsed field gel electrophoresis type, and 18 (85.7%) isolates possessed the genes for PVL.29
A number of studies in adults have examined the risk factors within the HIV population for S. aureus infection. Within HIV-positive adults, described risk factors for S. aureus infection include elevated HIV viral load, antiretroviral medication noncompliance, high-risk behaviors, and injection drug use.30 A single center study in children found that recent antibiotic exposure, a log10 HIV viral load ≥3, a CD4 T-cell count ≤350 cells/mm3, and a higher Centers for Disease Control and Prevention (CDC) category of disease were associated with S. aureus infections in univariate analyses.29 Another large multicenter study with over 1,800 subjects attempted to determine associations specifically with MRSA infection in pediatric HIV patients.31 This work found that a log10 viral load ≥3, African-American race, and residence in a high MRSA prevalence area were all independently associated with MRSA infection. Notably, behavioral acquisition of HIV (ie, high-risk sexual behavior or injection drug use) was not associated with MRSA infection in this population. Among HIV-positive children, SSTIs are the predominant manifestation of S. aureus disease (over 80%) although other invasive infections do occur.29,31
Studies among African children with HIV have differed somewhat from those among HIV-positive children in the developed world. Up to 24% of 203 HIV-positive children in a study at Tygerberg Children’s Hospital in Cape Town were colonized with S. aureus; more importantly, colonization was associated with a greater degree of immune suppression.32 Other work from Johannesburg has shown that the incidence of S. aureus bacteremia in South African children is higher than that described in the developed world and is particularly associated with HIV infection.33 Furthermore, S. aureus was more commonly a cause of pneumonia in HIV-positive (15% of cases) compared to HIV-negative (3%) patients in a study of 150 South African children.34 Over 90% of the S. aureus isolates in a Cape Town study were resistant to TMP-SMX, and approximately 80% were MRSA.32 In Gaborone, Botswana,35 higher rates of antimicrobial resistance were seen in MRSA compared to MSSA to numerous antibiotics including erythromycin, chloramphenicol, tetracycline, and gentamicin, suggesting the existence of an MDR-MRSA phenotype in this population as well.
While there does appear to be a correlation between viral load, CD4 T-cell count, and S. aureus infection, the interaction of S. aureus and HIV is complex and incompletely understood. It is possible that depletion of CD4 T-cells during HIV infection affects granulocyte function and predisposes to S. aureus infection. Investigations into the pathogenesis of simian immunodeficiency virus in nonhuman primates has revealed that disease progression is associated with depletion of T helper 17 cells.36 Studies in mice deficient in interleukin (IL)-17 producing T-cells have been shown to have impaired neutrophil recruitment and increased severity of cutaneous staphylococcal infections37 as well as surgical site infections.38 Mouse models have also shown S. aureus nasal colonization to be a T-cell dependent process,39 suggesting that HIV-mediated T-cell suppression may predispose to S. aureus colonization and infection. Alternatively, one may hypothesize that at least some of the clinical findings of CD4 suppression are the result of temporary fluctuations in control of HIV disease due to an intercurrent infection, as has been well described for other pathogens such as Treponema pallidum.40
The management of bacterial infections in children with HIV should take into account both the type of infection and the degree of immune suppression. Published guidelines state that children without serious immune compromise (CDC category I) and who are not neutropenic should be treated similarly to HIV-uninfected children.41 In the Houston series, over 50% of patients were successfully managed as outpatients with incision and drainage and oral antimicrobials.29 Most experts, however, recommend hospitalization and broad spectrum empiric antimicrobials, including the use of bactericidal agents, for those children who are neutropenic or severely immunocompromised.
Children with malignancy
Patients suffering from malignancy are at risk for invasive bacterial infection as a result of receipt of antineoplastic chemotherapy, the need for invasive procedures and central venous catheters, and the intrinsic immune dysfunction of their underlying disease (as is the case with hematologic malignancies). S. aureus accounts for 9%–10% of total cases of bacteremia in a series of adults with malignancy. The mortality rates reported in adults, however, range from 15% to as high as 24%;42,43 the mortality rate for S. aureus pneumonia in adult cancer patients is particularly high at 49.5%.44 Furthermore, in a series of 52 nonneutropenic adults with malignancy and S. aureus bacteremia, 33% developed metastatic infectious complications.43
As S. aureus colonization has previously been associated with an increased risk of staphylococcal infection,45 much interest has developed in identifying colonized high-risk patients. The rates of MRSA colonization among adults with malignancy reported in the literature varies widely from 9%–60%.46,47 It is known, however, that MRSA colonization has increased overtime among pediatric cancer patients; in one center, MRSA colonization increased from <1% to 3.8% in a seven year period among 1,735 subjects studied.48
Bloodstream infections are the most common health care-associated infections (HAI) in pediatric cancer patients with Gram-positive cocci, in particular S. aureus, being the most common agents.49 In children with malignancy, S. aureus accounts for up to 9%–13% of neutropenic bloodstream infections and febrile neutropenia.50,51 Moreover, S. aureus has been the most commonly reported cause of infectious death in some studies of pediatric febrile neutropenia.52 In contrast to healthy children and even HIV-positive children, the most common manifestation of S. aureus infection in children with malignancy is bacteremia with central line-associated bloodstream infection (CLA-BSI) accounting for 84.7% of cases in one study.53 A study of 52 cases of S. aureus bacteremia at the St Jude’s Children’s Research Hospital noted that less than a third of patients with S. aureus bacteremia were in remission at the time of infection and that 81% of infections were a result of MSSA.54 While the majority of bacteremic events were due to MSSA, MRSA infections were associated with a longer duration of bacteremia and a greater need for catheter removal.54 This study also revealed that 46% of pediatric cancer patients with S. aureus bacteremia had an absolute neutrophil count less than 500 cells/mm3. Notably MRSA infections are associated with a larger relative proportion of SSTIs,53,55 likely a reflection of the dissemination of the USA300 clone of MRSA in the community, with its predilection for causing SSTI.56 In Houston, TX, the USA300 clone accounted for a much lower percentage of S. aureus infections among pediatric cancer than HIV patients (63 of 171 [36.8%] isolates).29,53 Notably, non-USA300 pulsotypes were more often associated with bacteremia than SSTI. Similarly, Srinivasan et al reported that among 88 MRSA infections in pediatric cancer patients, PVL-positive isolates accounted for only 40% of all cases with the majority being USA300.55
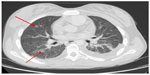
Complications of S. aureus infections have been described to occur in pediatric cancer patients in 10%–18% of all infections and up to 33% of cases of bacteremia.53–55 A study at Texas Children’s Hospital (TCH) of 213 S. aureus infections in children with cancer has shown that complicated S. aureus bacteremia is associated with thrombocytopenia and an absolute monocyte count <100 cells/mm3.53 There was additionally a tendency for complications to occur more frequently in hematopoietic stem cell transplant recipients. Described complications of S. aureus bacteremia in this population include pulmonary nodules, pneumonia, pleural effusion, lung abscess, thromboses, skin nodules, musculoskeletal infection, central nervous system abscess, and endocarditis.53,54 In the TCH series, six patients with pulmonary nodules underwent biopsy and 5 of 6 cultures revealed S. aureus alone; the sixth patient showed evidence of metastatic malignancy (Figure 2). The overall survival from S. aureus infection is much better among pediatric than adult oncology patients. Mortality from S. aureus in this population is described at 2.3% and is associated with a diagnosis of acute myelogenous leukemia and a platelet count <50,000/mm3 at the time of infection.53
The need for alteration of antistaphylococcal therapy in the setting of malignancy is unclear. Some experts recommend a longer duration of therapy than in immunocompetent hosts and/or the use of a bactericidal agent. In the series of S. aureus bacteremia from St Jude’s, patients were treated for a median of 10 days with either vancomycin or oxacillin; notably, no patient had a recurrence of bacteremia within a year of the treatment.54 In the TCH series, 6.8% of neutropenic patients developed relapse of infection following discontinuation of therapy compared to 3.2% of nonneutropenic patients; however, this did not achieve statistical significance.53 Regardless, many experts would recommend use of bactericidal agents and/or a prolonged duration of therapy for neutropenic hosts with a S. aureus infection. Furthermore, for patients who have severe or deep-seated infection, some would recommend continuing therapy beyond the usual duration until granulocyte counts have recovered. For CLA-BSI due to S. aureus, catheter removal is strongly recommended, particularly in this population.57
One of the modalities that has been adopted to decrease the incidence of invasive infections in hospitalized patients, in particular those with hematologic malignancies, is the use of antiseptics such as chlorhexidine gluconate. The use of these agents for decolonization, central line care, and oral rinses has been shown to decrease the incidence of HAIs in adult populations.58–64 While there is the potential for tremendous benefit, there are a number of genes in S. aureus, notably qacA/B, smr, and norA, which confer higher minimum inhibitory concentration/minimum bactericidal concentration to these agents.65–67 These plasmid-borne genes encode efflux pumps which not only confer tolerance to antiseptics but also resistance to a number of other systemic antimicrobials including fluoroquinolones. A recent pediatric study revealed an increase in the prevalence of S. aureus isolates from oncology patients harboring biocide tolerance genes following an escalation of chlorhexidine use at their institution.53 The isolates with antiseptic tolerance were also more likely to have resistance to ciprofloxacin. Notably, however, there was a decrease in the total number of S. aureus infections following implementation of antiseptic prophylaxis, attesting to the potential benefits of their use. In addition, recent laboratory studies have demonstrated that MDR efflux pumps may contribute to S. aureus’ ability to colonize skin, adding further import to these clinical findings.68 While these findings are concerning, the full clinical implications are unclear and are under investigation.
Solid organ transplantation
Patients who have received a solid-organ transplant (SOT) represent a unique population for the acquisition of HAIs given their preoperative organ failure, need for surgery, and often prolonged hospitalization and immunosuppression. The need for anti-T-cell immunosuppressive therapies may predispose to S. aureus infection in a manner similar to that described for HIV infection above. In fact, SOT is a known risk factor for S. aureus bacteremia in a study of Canadian adults.69 Data on S. aureus infections in pediatric SOT recipients is extremely limited, and much of what is known was gleaned from larger studies of infection in pediatric SOT in general or from studies in adult populations. Overall, infections among SOT patients can be divided temporally based on the time since transplantation in the early (<1 month), intermediate (1–6 months), and late (>6 months) periods. The early period is typically characterized by infections that are health care associated in nature, including surgical site infections, ventilator-associated pneumonia, and CLA-BSI, of which S. aureus is among the principal pathogens.70 In fact, among adult liver transplant recipients, recent surgery was associated with an increased risk of S. aureus infection in multivariate analyses.71
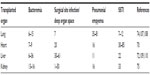
The frequency, clinical manifestations, and outcomes of S. aureus infection vary considerably with the organ transplanted (Table 1). A single center 11-year review of 345 patients revealed that bacterial infection occurred in 45% of pediatric liver transplant recipients within the first 90 days, with S. aureus accounting for 20.4% of infections.72 Colonization has further been shown to be a risk factor for MRSA infection in liver recipients.73 Among 596 adult lung transplant recipients, S. aureus infection occurred in 18% in the first 90 days posttransplant and was associated with an increased length of hospital stay, risk of subsequent cytomegalovirus disease, graft rejection, and mortality.74 By comparison, S. aureus is a much less common complication of renal transplant in adults, complicating only 1.3% of cases in one series of 116 subjects.75
Specific studies of pediatric SOT recipients with S. aureus infection are ongoing, with preliminary data suggesting a rate of clindamycin resistance that is higher than in the general population (McNeil et al, unpublished data). Furthermore, there has been demonstrated a temporal increase in the proportion of MRSA infections among lung transplant recipients.74
Given that these patients often have numerous comorbidities, by-and-large treatment must be individualized to the unique circumstances of the patient. Aggressive treatment of any and all infections is essential, particularly in the early posttransplant period, with a goal towards identifying an organism. Patients who are ill-appearing or recently transplanted should be admitted and receive broad spectrum intravenous antimicrobial therapy for both Gram-positive and Gram-negative agents including coverage for MRSA. Once patients are clinically stable and a pathogen identified, therapy can be deescalated to oral agents in many cases. Preliminary data from ongoing pediatric studies suggests that clindamycin can be used successfully to treat nonlife-threatening infection in SOT patients in the late posttransplant period (McNeil et al, unpublished data).
Due to the evidence of increasing frequency of MRSA infection among SOT recipients,74 prevention strategies are needed. Numerous centers have implemented surveillance and decolonization measures to curtail the spread of antibiotic-resistant organisms. In one single-center study of 144 adult liver recipients, a protocol was instituted of active surveillance for nasal and rectal colonization, cohorting, and decolonization with intranasal mupirocin.76 These measures were associated with a decrease in the rate of newly colonized patients as well as S. aureus infections, suggesting a benefit from these measures in this high risk population.
Primary immunodeficiency
Severe or recurrent infections, including skin infections, are among the warning signs of a primary immunodeficiency (PI). While healthy children frequently experience recurrent infections with S. aureus,77,78 the clinician should consider the possibility of PI particularly when patients are exhibiting other warning signs of immune dysfunction such as failure to thrive, pneumonia, chronic diarrhea, other infections, or generalized lymphadenopathy. Phagocyte defects in particular predispose to staphylococcal infections. The most common primary phagocyte disorder encountered in clinical practice is chronic granulomatous disease (CGD).79,80 CGD is caused by any number of mutations in the genes encoding components of the respiratory burst pathway of phagocytes, resulting in impaired production of reactive oxygen species and bacterial killing. Patients with CGD are predisposed to suppurative infections, notably skin abscesses, lymphadenitis, pneumonia/lung abscesses, and osteomyelitis.81 S. aureus is the most common bacterial pathogen isolated in children with CGD, accounting for up to 31% of all positive cultures;82,83 other major pathogens in CGD include Serratia, Nocardia, and Aspergillus spp. Long-term prophylaxis with TMP-SMX in addition to itraconazole is helpful at decreasing the frequency of serious infections in patients with CGD.84,85 Interferon-gamma injections have also been used successfully to decrease the incidence of major infections in children with CGD. In addition, hematopoietic stem cell transplant has successfully provided cure in patients with CGD.86
Leukocyte adhesion defect (LAD) is a phagocyte disorder characterized by failure of neutrophils to extravasate from the peripheral circulation into infected tissues. Patients with LAD experience recurrent infections of the skin, mucosal surfaces, and gastrointestinal tract. The most common infectious agents in these patients are staphylococci as well as Pseudomonas spp.; infections in LAD are characterized by extreme leukocytosis but the absence of purulence at the site of infection.87 Similar to CGD, TMP-SMX prophylaxis is recommended for children with LAD.
Hyperimmunoglobulin E syndrome (HIES), also known as Job’s syndrome, is an additional increasingly recognized PI. HIES exists in both autosomal dominant and recessive forms with coarse facial features, severe eczema, recurrent cold skin abscesses, recurrent pneumonias, and pneumatocele formation among its many features.87,88 Infections in HIES are primarily due to S. aureus although other commonly implicated bacterial pathogens include Streptococcus pneumoniae and Haemophilus influenzae; chronic mucocutaneous candidiasis is an additional common infectious complication of HIES. The molecular basis behind HIES is incompletely understood but involves mutations within STAT3, a transcription factor involved in the expression of a number of cytokines including IL-6, IL-10, and IL-17.89 Moreover, STAT3 mutations have been shown to impair the differentiation of T helper 17 cells and thus may impair neutrophil recruitment leading to a predisposition to staphylococcal infections.90
Disorders of humoral immunity are also associated with an increased incidence of S. aureus infection. S. aureus, along with S. pneumoniae and H. influenzae, are among the most common bacterial pathogens isolated in both X-linked agammaglobulinemia and common variable immunodeficiency.87,91 Individuals with common variable immunodeficiency often develop bronchiectasis and chronic lung disease, which may be complicated by staphylococcal infection.
In general all infections in children with PIs should be managed aggressively with a goal towards identifying the pathogen and providing specific therapy. There is a paucity of published data on the antimicrobial susceptibility profiles of staphylococcal isolates in patients with PI. One could reasonably hypothesize, however, that this population would have a high rate of infection with drug-resistant organisms given the frequent need for antimicrobials. Furthermore, once clinical improvement is observed, practitioners should be cautious for relapse of symptoms on discontinuation of therapy. Consultation with an infectious diseases specialist or clinical immunologist is recommended in the management of these patients.
Specific antimicrobial therapy for drug resistant S. aureus in children
As a basic principle, empiric use of antimicrobials should be guided by local epidemiology and antimicrobial susceptibility as well as the clinical state of the patient, with final therapy determined by culture and sensitivity data. A brief overview of specific antimicrobial therapy commonly used for pediatric staphylococcal infections is provided with data from immunocompromised hosts reviewed when applicable.
There is a great deal of strong in vitro as well as clinical evidence from adult patients supporting the use of nafcillin or cefazolin over vancomycin for the treatment of MSSA infections.92 Thus an antistaphylococcal β-lactam is recommended for both healthy and immunocompromised hosts for disease as a result of MSSA. Moreover, for severely ill patients with suspected staphylococcal disease, empiric combination of an antistaphylococcal β-lactam along with vancomycin pending culture data is recommended by some experts.
Vancomycin remains the drug of choice for the treatment of severe MRSA infections in children, with typical doses ranging from 10–15 mg/kg/dose every 8 hours.93 For critically ill, bacteremic, or patients with central nervous system disease, it is recommended to increase dosing to every 6 hours. The Infectious Disease Society of America (IDSA) recommends early addition of vancomycin if there is suspicion of staphylococcal infection in febrile neutropenic patients.94 The need for therapeutic monitoring of vancomycin levels in children is unclear. From studies in adults, primarily with S. aureus bacteremia, there is evidence to suggest improved clinical outcomes with vancomycin troughs of 15–20 μg/mL.95 Many experts recommend similar goal levels in pediatric patients, yet the evidence to support this is limited. Serum vancomycin troughs >15 μg/mL are often difficult to achieve in young children; in an Australian series only 43% of children 1–12 years old achieved goal troughs after 8 days of therapy.96
Clindamycin is the most commonly used antimicrobial for the management of mild–moderate CA-MRSA infections.97 Previous retrospective series in immunocompetent hosts have shown it to be noninferior to vancomycin in children.98 Clindamycin was the most commonly used antimicrobial for the treatment of S. aureus infections in a small series of children with HIV, albeit, these were primarily SSTIs and not invasive infections.29 Thus clindamycin could be considered for select patients who are not bacteremic, not severely immune suppressed, and have clindamycin susceptible isolates. Nearly all S. aureus isolates exhibit in vitro susceptibility to TMP-SMX, and this agent is endorsed by the IDSA for the treatment of staphylococcal SSTI.93 Pediatric retrospective studies have been conflicting in the efficacy of TMP-SMX for SSTI, with some studies showing equivalence while others showed increased treatment failure compared to clindamycin.99,100 Increasing rates of clindamycin resistance have resulted in increased use of TMP-SMX in some centers. There is very limited data for TMP-SMX treatment of invasive infections, particularly in immunocompromised hosts.
Linezolid, a bacteriostatic oxazolidinone, has been shown to be noninferior to vancomycin for Gram-positive bacteremia and hospital-acquired pneumonia in pediatric clinical trials.101 The IDSA also recommends that physicians consider the use of linezolid for empiric treatment of febrile neutropenic adults when Gram-positive organisms are a concern.94 A potential concern with linezolid is the occurrence of medication-induced bone marrow suppression; this could be potentially worrisome in the already immunocompromised host. A series of 17 pediatric cancer patients from Athens, Greece sought to evaluate the safety and efficacy of linezolid for Gram-positive infections in this population.102 All patients achieved microbiologic cure in this small series; 23.5% experienced adverse events; however, chemotherapy induced myelosuppression did not appear to be exacerbated by linezolid.
Other newer antistaphylococcal agents have thus far seen limited use in children. Daptomycin, a cyclic lipopeptide antimicrobial, is approved by the US Food and Drug Administration for the treatment of SSTI and bacteremia in adults. Daptomycin is currently undergoing investigation to establish the optimal dosing as well as its safety in children. A handful of case reports/series have described daptomycin use in children with doses ranging from 4–6 mg/kg/day with good efficacy and few adverse events.103,104 Ceftaroline, a new anti-MRSA cephalosporin, is approved in adults for treating SSTI as well as community-acquired pneumonia.105,106 Notably, this agent had a similar side effect profile to other cephalosporins and was better tolerated than vancomycin in adults. Clinical trials in pediatric patients are ongoing.
Conclusion
Children with immunocompromising conditions represent a unique group for the development of S. aureus infections. While many of the manifestations of staphylococcal disease are similar to that seen in healthy pediatric populations, there is the potential for the development of complications, particularly in those children with malignancy. Furthermore, infections in this population often exhibit high rates of resistance to commonly used antimicrobials. These infections should be managed aggressively, letting local epidemiology, culture, and susceptibility data guide antibiotic choice. There remain significant gaps in knowledge regarding the epidemiology and management of these infections that warrant further investigation.
Disclosure
The author reports no conflicts of interest in this work.
References
Moran GJ, Krishnadasan A, Gorwitz RJ, et al; EMERGEncy ID Net Study Group. Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med. 2006;355(7):666–674. | |
Gonzalez BE, Teruya J, Mahoney DH Jr, et al. Venous thrombosis associated with staphylococcal osteomyelitis in children. Pediatrics. 2006;117(5):1673–1679. | |
Gonzalez BE, Hulten KG, Dishop MK, et al. Pulmonary manifestations in children with invasive community-acquired Staphylococcus aureus infection. Clin Infect Dis. 2005;41(5):583–590. | |
Martínez-Aguilar G, Avalos-Mishaan A, Hulten K, Hammerman W, Mason EO Jr, Kaplan SL. Community-acquired, methicillin-resistant and methicillin-susceptible Staphylococcus aureus musculoskeletal infections in children. Pediatr Infect Dis J. 2004;23(8):701–706. | |
Pannaraj PS, Hulten KG, Gonzalez BE, Mason EO Jr, Kaplan SL. Infective pyomyositis and myositis in children in the era of community-acquired, methicillin-resistant Staphylococcus aureus infection. Clin Infect Dis. 2006;43(8):953–960. | |
McKinnell JA, Miller LG, Eells SJ, Cui E, Huang SS. A systematic literature review and meta-analysis of factors associated with methicillin-resistant Staphylococcus aureus colonization at time of hospital or intensive care unit admission. Infect Control Hosp Epidemiol. 2013;34(10):1077–1086. | |
Dukic VM, Lauderdale DS, Wilder J, Daum RS, David MZ. Epidemics of community-associated methicillin-resistant Staphylococcus aureus in the United States: a meta-analysis. PLoS One. 2013;8(1):e52722. | |
Naimi TS, LeDell KH, Como-Sabetti K, et al. Comparison of community- and health care-associated methicillin-resistant Staphylococcus aureus infection. JAMA. 2003;290(22):2976–2984. | |
Herold BC, Immergluck LC, Maranan MC, et al. Community-acquired methicillin-resistant Staphylococcus aureus in children with no identified predisposing risk. JAMA. 1998;279(8):593–598. | |
David MZ, Glikman D, Crawford SE, et al. What is community-associated methicillin-resistant Staphylococcus aureus? J Infect Dis. 2008;197(9):1235–1243. | |
Liu C, Graber CJ, Karr M, et al. A population-based study of the incidence and molecular epidemiology of methicillin-resistant Staphylococcus aureus disease in San Francisco, 2004-2005. Clin Infect Dis. 2008;46(11):1637–1646. | |
Al-Rawahi GN, Reynolds S, Porter SD, et al. Community-associated CMRSA-10 (USA-300) is the predominant strain among methicillin-resistant Staphylococcus aureus strains causing skin and soft tissue infections in patients presenting to the emergency department of a Canadian tertiary care hospital. J Emerg Med. 2010;38(1):6–11. | |
Gonzalez BE, Rueda AM, Shelburne SA, Musher DM, Hamill RJ, Hulten KG. Community-associated strains of methicillin-resistant Staphylococccus aureus as the cause of healthcare-associated infection. Infect Control Hosp Epidemiol. 2006;27(10):1051–1056. | |
Hultén KG, Kaplan SL, Lamberth LB, et al. Hospital-acquired Staphylococcus aureus infections at Texas Children’s Hospital, 2001–2007. Infect Control Hosp Epidemiol. 2010;31(2):183–190. | |
Pasquale TR, Jabrocki B, Salstrom SJ, et al; IMPACT-HAP Study Group. Emergence of methicillin-resistant Staphylococcus aureus USA300 genotype as a major cause of late-onset nosocomial pneumonia in intensive care patients in the USA. Int J Infect Dis. 2013;17(6):e398–e403. | |
McCaskill ML, Mason EO, Kaplan SL, Hammerman W, Lamberth LB, Hultén KG. Increase of the USA300 clone among community-acquired methicillin-susceptible Staphylococcus aureus causing invasive infections. Pediatr Infect Dis J. 2007;26(12):1122–1127. | |
Kaplan SL, Hulten KG, Mason EO. Staphylococcus aureus infections (coagulase-positive staphylococci). In: Cherry JD, Demmler-Harrison GJ, Kaplan SL, Steinbach WJ, Hotez PJ, editors. Textbook of Pediatric Infectious Diseases. Philadelphia, PA: Elsevier; 2013;1:1113–1130. | |
Foster TJ, Geoghegan JA, Ganesh VK, Höök M. Adhesion, invasion and evasion: the many functions of the surface proteins of Staphylococcus aureus. Nat Rev Microbiol. 2014;12(1):49–62. | |
Hair PS, Ward MD, Semmes OJ, Foster TJ, Cunnion KM. Staphylococcus aureus clumping factor A binds to complement regulator factor I and increases factor I cleavage of C3b. J Infect Dis. 2008;198(1):125–133. | |
Hair PS, Echague CG, Sholl AM, et al. Clumping factor A interaction with complement factor I increases C3b cleavage on the bacterial surface of Staphylococcus aureus and decreases complement-mediated phagocytosis. Infect Immun. 2010;78(4):1717–1727. | |
Sharp JA, Echague CG, Hair PS, et al. Staphylococcus aureus surface protein SdrE binds complement regulator factor H as an immune evasion tactic. PLoS One. 2012;7(5):e38407. | |
Panton PN, Valentine FCO. Staphylococcal toxins. Lancet. 1932;219(5662):506–508. | |
Boyle-Vavra S, Daum RS. Community-acquired methicillin-resistant Staphylococcus aureus: the role of Panton-Valentine leukocidin. Lab Invest. 2007;87(1):3–9. | |
Bocchini CE, Hulten KG, Mason EO, Gonzalez BE, Hammerman WA, Kaplan SL. Panton-Valentine leukocidin genes are associated with enhanced inflammatory response and local disease in acute hematogenous Staphylococcus aureus osteomyelitis in children. Pediatrics. 2006;117(2):433–440. | |
Gordon RJ, Chez N, Jia H, et al. The NOSE study (nasal ointment for Staphylococcus aureus eradication): a randomized controlled trial of monthly mupirocin in HIV-infected individuals. J Acquir Immune Defic Syndr. 2010;55(4):466–472. | |
Senthilkumar A, Kumar S, Sheagren JN. Increased incidence of Staphylococcus aureus bacteremia in hospitalized patients with acquired immunodeficiency syndrome. Clin Infect Dis. 2001;33(8):1412–1416. | |
Diep BA, Chambers HF, Graber CJ, et al. Emergence of multidrug-resistant, community-associated, methicillin-resistant Staphylococcus aureus clone USA300 in men who have sex with men. Ann Intern Med. 2008;148(4):249–257. | |
Diep BA, Gill SR, Chang RF, et al. Complete genome sequence of USA300, an epidemic clone of community-acquired methicillin-resistant Staphylococcus aureus. Lancet. 2006;367(9512):731–739. | |
McNeil JC, Hulten KG, Kaplan SL, Schwarzwald HL, Mason EO. Staphylococcus aureus infections in HIV-positive children and adolescents. Pediatr Infect Dis J. 2012;31(3):284–286. | |
Lee NE, Taylor MM, Bancroft E, et al. Risk factors for community-associated methicillin-resistant Staphylococcus aureus skin infections among HIV-positive men who have sex with men. Clin Infect Dis. 2005;40(10):1529–1534. | |
Siberry GK, Frederick T, Emmanuel P, et al. Methicillin-resistant Staphylococcus aureus infections in human immunodeficiency virus-infected children and adolescents. AIDS Res Treat. 2012;2012:627974. | |
Cotton MF, Wasserman E, Smit J, Whitelaw A, Zar HJ. High incidence of antimicrobial resistant organisms including extended spectrum beta-lactamase producing Enterobacteriaceae and methicillin-resistant Staphylococcus aureus in nasopharyngeal and blood isolates of HIV-infected children from Cape Town, South Africa. BMC Infect Dis. 2008;8:40. | |
Groome MJ, Albrich WC, Wadula J, Khoosal M, Madhi SA. Community-onset Staphylococcus aureus bacteraemia in hospitalised African children: high incidence in HIV-infected children and high prevalence of multidrug resistance. Paediatr Int Child Health. 2012;32(3):140–146. | |
Zar HJ, Hanslo D, Hussey G. The impact of HIV infection and trimethoprim-sulphamethoxazole prophylaxis on bacterial isolates from children with community-acquired pneumonia in South Africa. J Trop Pediatr. 2003;49(2):78–83. | |
Wood SM, Shah SS, Bafana M, et al. Epidemiology of methicillin-resistant Staphylococcus aureus bacteremia in Gaborone, Botswana. Infect Control Hosp Epidemiol. 2009;30(8):782–785. | |
Favre D, Lederer S, Kanwar B, et al. Critical loss of the balance between Th17 and T regulatory cell populations in pathogenic SIV infection. PLoS Pathog. 2009;5(2):e1000295. | |
Cho JS, Pietras EM, Garcia NC, et al. IL-17 is essential for host defense against cutaneous Staphylococcus aureus infection in mice. J Clin Invest. 2010;120(5):1762–1773. | |
Maher BM, Mulcahy ME, Murphy AG, et al. Nlrp-3-driven interleukin 17 production by γδT cells controls infection outcomes during Staphylococcus aureus surgical site infection. Infect Immun. 2013;81(12):4478–4489. | |
Archer NK, Harro JM, Shirtliff ME. Clearance of Staphylococcus aureus nasal carriage is T cell dependent and mediated through interleukin-17A expression and neutrophil influx. Infect Immun. 2013;81(6):2070–2075. | |
Buchacz K, Patel P, Taylor M, et al. Syphilis increases HIV viral load and decreases CD4 cell counts in HIV-infected patients with new syphilis infections. AIDS. 2004;18(15):2075–2079. | |
aidsinfo.nih.gov [homepage on the Internet]. Guidelines for the prevention and treatment of opportunistic infections In HIV-exposed and HIV-infected Children; 2013. AidsInfo. US Department of Human Health and Services. Available from: http://aidsinfo.nih.gov/guidelines. Accessed March 12, 2014. | |
Klastersky J, Ameye L, Maertens J, et al. Bacteraemia in febrile neutropenic cancer patients. Int J Antimicrob Agents. 2007;30(Suppl 1):S51–S59. | |
Gopal AK, Fowler VG Jr, Shah M, et al. Prospective analysis of Staphylococcus aureus bacteremia in nonneutropenic adults with malignancy. J Clin Oncol. 2000;18(5):1110–1115. | |
Kang CI, Song JH, Chung DR, et al; Korean Network for Study on Infectious Diseases (KONSID). Bloodstream infections in adult patients with cancer: clinical features and pathogenic significance of Staphylococcus aureus bacteremia. Support Care Cancer. 2012;20(10):2371–2378. | |
von Eiff C, Becker K, Machka K, Stammer H, Peters G. Nasal carriage as a source of Staphylococcus aureus bacteremia. Study Group. N Engl J Med. 2001;344(1):11–16. | |
Hagiwara S, Miwa A, Yoshida M, et al. Methicillin-resistant Staphylococcus aureus: colonization and development of infection in patients with haematological disorders. Eur J Haematol. 1995;55(4):267–271. | |
Ghanem HM, Abou-Alia AM, Alsirafy SA. Prevalence of methicillin-resistant Staphylococcus aureus colonization and infection in hospitalized palliative care patients with cancer. Am J Hosp Palliat Care. 2013;30(4):377–379. | |
Srinivasan A, Seifried SE, Zhu L, et al. Increasing prevalence of nasal and rectal colonization with methicillin-resistant Staphylococcus aureus in children with cancer. Pediatr Blood Cancer. 2010;55(7):1317–1322. | |
McCullers JA, Williams BF, Wu S, et al. Healthcare-Associated Infections at a Children’s Cancer Hospital, 1983–2008. J Pediatric Infect Dis Soc. 2012;1(1):26–34. | |
Bakhshi S, Padmanjali KS, Arya LS. Infections in childhood acute lymphoblastic leukemia: an analysis of 222 febrile neutropenic episodes. Pediatr Hematol Oncol. 2008;25(5):385–392. | |
Castagnola E, Fontana V, Caviglia I, et al. A prospective study on the epidemiology of febrile episodes during chemotherapy-induced neutropenia in children with cancer or after hemopoietic stem cell transplantation. Clin Infect Dis. 2007;45(10):1296–1304. | |
Santolaya ME, Alvarez AM, Avilés CL, et al. Admission clinical and laboratory factors associated with death in children with cancer during a febrile neutropenic episode. Pediatr Infect Dis J. 2007;26(9):794–798. | |
McNeil JC, Hulten KG, Kaplan SL, Mahoney DH, Mason EO. Staphylococcus aureus infections in pediatric oncology patients: high rates of antimicrobial resistance, antiseptic tolerance and complications. Pediatr Infect Dis J. 2013;32(2):124–128. | |
Srinivasan A, Seifried S, Zhu L, et al. Staphylococcus aureus bacteremia in pediatric patients with cancer. Pediatr Infect Dis J. 2010;29(2):172–174. | |
Srinivasan A, Seifried S, Zhu L, et al. Panton-Valentine leukocidin-positive methicillin-resistant Staphylococcus aureus infections in children with cancer. Pediatr Blood Cancer. 2009;53(7):1216–1220. | |
Talan DA, Krishnadasan A, Gorwitz RJ, et al; EMERGEncy ID Net Study Group. Comparison of Staphylococcus aureus from skin and soft-tissue infections in US emergency department patients, 2004 and 2008. Clin Infect Dis. 2011;53(2):144–149. | |
Mermel LA, Allon M, Bouza E, et al. Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection: 2009 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2009;49(1):1–45. | |
Ferretti GA, Hansen IA, Whittenburg K, Brown AT, Lillich TT, Ash RC. Therapeutic use of chlorhexidine in bone marrow transplant patients: case studies. Oral Surg Oral Med Oral Pathol. 1987;63(6):683–687. | |
DeRiso AJ 2nd, Ladowski JS, Dillon TA, Justice JW, Peterson AC. Chlorhexidine gluconate 0.12% oral rinse reduces the incidence of total nosocomial respiratory infection and nonprophylactic systemic antibiotic use in patients undergoing heart surgery. Chest. 1996;109(6):1556–1561. | |
Bleasdale SC, Trick WE, Gonzalez IM, Lyles RD, Hayden MK, Weinstein RA. Effectiveness of chlorhexidine bathing to reduce catheter-associated bloodstream infections in medical intensive care unit patients. Arch Intern Med. 2007;167(19):2073–2079. | |
Kassakian SZ, Mermel LA, Jefferson JA, Parenteau SL, Machan JT. Impact of chlorhexidine bathing on hospital-acquired infections among general medical patients. Infect Control Hosp Epidemiol. 2011;32(3):238–243. | |
Miller MR, Niedner MF, Huskins WC, et al; National Association of Children’s Hospitals and Related Institutions Pediatric Intensive Care Unit Central Line-Associated Bloodstream Infection Quality Transformation Teams. Reducing PICU central line-associated bloodstream infections: 3-year results. Pediatrics. 2011;128(5):e1077–e1083. | |
Pereira Pinto L, de Souza LB, Gordón-Núñez MA, et al. Prevention of oral lesions in children with acute lymphoblastic leukemia. Int J Pediatr Otorhinolaryngol. 2006;70(11):1847–1851. | |
Popovich KJ, Hota B, Hayes R, Weinstein RA, Hayden MK. Effectiveness of routine patient cleansing with chlorhexidine gluconate for infection prevention in the medical intensive care unit. Infect Control Hosp Epidemiol. 2009;30(10):959–963. | |
Grinius LL, Goldberg EB. Bacterial multidrug resistance is due to a single membrane protein which functions as a drug pump. J Biol Chem. 1994;269(47):29998–30004. | |
Rouch DA, Cram DS, DiBerardino D, Littlejohn TG, Skurray RA. Efflux-mediated antiseptic resistance gene qacA from Staphylococcus aureus: common ancestry with tetracycline- and sugar-transport proteins. Mol Microbiol. 1990;4(12):2051–2062. | |
Smith K, Gemmell CG, Hunter IS. The association between biocide tolerance and the presence or absence of qac genes among hospital-acquired and community-acquired MRSA isolates. J Antimicrob Chemother. 2008;61(1):78–84. | |
Truong-Bolduc QC, Villet RA, Estabrooks ZA, Hooper DC. Native efflux pumps contribute resistance to antimicrobials of skin and the ability of Staphylococcus aureus to colonize skin. J Infect Dis. Epub December 23, 2013. | |
Laupland KB, Ross T, Gregson DB. Staphylococcus aureus bloodstream infections: risk factors, outcomes, and the influence of methicillin resistance in Calgary, Canada, 2000–2006. J Infect Dis. 2008;198(3):336–343. | |
Richards MJ, Edwards JR, Culver DH, Gaynes RP. Nosocomial infections in pediatric intensive care units in the United States. National Nosocomial Infections Surveillance System. Pediatrics. 1999; 103(4):e39. | |
Florescu DF, McCartney AM, Qiu F, et al. Staphylococcus aureus infections after liver transplantation. Infection. 2012;40(3):263–269. | |
Nafady-Hego H, Elgendy H, Moghazy WE, Fukuda K, Uemoto S. Pattern of bacterial and fungal infections in the first 3 months after pediatric living donor liver transplantation: an 11-year single-center experience. Liver Transpl. 2011;17(8):976–984. | |
Desai D, Desai N, Nightingale P, Elliott T, Neuberger J. Carriage of methicillin-resistant Staphylococcus aureus is associated with an increased risk of infection after liver transplantation. Liver Transpl. 2003;9(7):754–759. | |
Shields RK, Clancy CJ, Minces LR, et al. Staphylococcus aureus infections in the early period after lung transplantation: epidemiology, risk factors, and outcomes. J Heart Lung Transplant. 2012;31(11):1199–1206. | |
Oliveira-Cunha M, Bowman V, di Benedetto G, et al. Outcomes of methicillin-resistant Staphylococcus aureus infection after kidney and/or pancreas transplantation. Transplant Proc. 2013;45(6):2207–2210. | |
Singh N, Squier C, Wannstedt C, Keyes L, Wagener MM, Cacciarelli TV. Impact of an aggressive infection control strategy on endemic Staphylococcus aureus infection in liver transplant recipients. Infect Control Hosp Epidemiol. 2006;27(2):122–126. | |
Bocchini CE, Mason EO, Hulten KG, Hammerman WA, Kaplan SL. Recurrent community-associated Staphylococcus aureus infections in children presenting to Texas Children’s Hospital in Houston, Texas. Pediatr Infect Dis J. 2013;32(11):1189–1193. | |
Fritz SA, Hogan PG, Hayek G, et al. Household versus individual approaches to eradication of community-associated Staphylococcus aureus in children: a randomized trial. Clin Infect Dis. 2012;54(6):743–751. | |
Wolach B, Gavrieli R, Roos D, Berger-Achituv S. Lessons learned from phagocytic function studies in a large cohort of patients with recurrent infections. J Clin Immunol. 2012;32(3):454–466. | |
van den Berg JM, Kuijpers TW. Educational paper: Defects in number and function of neutrophilic granulocytes causing primary immunodeficiency. Eur J Pediatr. 2011;170(11):1369–1376. | |
Song E, Jaishankar GB, Saleh H, Jithpratuck W, Sahni R, Krishnaswamy G. Chronic granulomatous disease: a review of the infectious and inflammatory complications. Clin Mol Allergy. 2011; 9(1):10. | |
Mouy R, Fischer A, Vilmer E, Seger R, Griscelli C. Incidence, severity, and prevention of infections in chronic granulomatous disease. J Pediatr. 1989;114(4 Pt 1):555–560. | |
van den Berg JM, van Koppen E, Ahlin A, et al. Chronic granulomatous disease: the European experience. PLoS One. 2009;4(4):e5234. | |
Margolis DM, Melnick DA, Alling DW, Gallin JI. Trimethoprim-sulfamethoxazole prophylaxis in the management of chronic granulomatous disease. J Infect Dis. 1990;162(3):723–726. | |
Gallin JI, Alling DW, Malech HL, et al. Itraconazole to prevent fungal infections in chronic granulomatous disease. N Engl J Med. 2003;348(24):2416–2422. | |
Leung T, Chik K, Li C, Shing M, Yuen P. Bone marrow transplantation for chronic granulomatous disease: long-term follow-up and review of literature. Bone Marrow Transplant. 1999;24(5):567–570. | |
Chinen J, Shearer WT. Primary immunodeficiency. In: Cherry JD, Demmler-Harrison GJ, Kaplan SL, Steinbach WJ, Hotez PJ, editors. Textbook of Pediatric Infectious Diseases. Vol 1. Philadelphia, PA: Elsevier; 2014:903–920. | |
Grimbacher B, Holland SM, Puck JM. Hyper-IgE syndromes. Immunol Rev. 2005;203:244–250. | |
Mogensen TH. STAT3 and the Hyper-IgE syndrome: Clinical presentation, genetic origin, pathogenesis, novel findings and remaining uncertainties. JAKSTAT. 2013;2(2):e23435. | |
Milner JD, Brenchley JM, Laurence A, et al. Impaired T(H)17 cell differentiation in subjects with autosomal dominant hyper-IgE syndrome. Nature. 2008;452(7188):773–776. | |
Cunningham-Rundles C, Bodian C. Common variable immunodeficiency: clinical and immunological features of 248 patients. Clin Immunol. 1999;92(1):34–48. | |
Schweizer ML, Furuno JP, Harris AD, et al. Comparative effectiveness of nafcillin or cefazolin versus vancomycin in methicillin-susceptible Staphylococcus aureus bacteremia. BMC Infect Dis. 2011;11:279. | |
Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children: executive summary. Clin Infect Dis. 2011;52(3):285–292. | |
Freifeld AG, Bow EJ, Sepkowitz KA, et al. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the infectious diseases society of america. Clin Infect Dis. 2011;52(4):e56–e93. | |
Rybak M, Lomaestro B, Rotschafer JC, et al. Therapeutic monitoring of vancomycin in adult patients: a consensus review of the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, and the Society of Infectious Diseases Pharmacists. Am J Health Syst Pharm. 2009;66(1):82–98. | |
Gordon CL, Thompson C, Carapetis JR, Turnidge J, Kilburn C, Currie BJ. Trough concentrations of vancomycin: adult therapeutic targets are not appropriate for children. Pediatr Infect Dis J. 2012; 31(12):1269–1271. | |
Herigon JC, Hersh AL, Gerber JS, Zaoutis TE, Newland JG. Antibiotic management of Staphylococcus aureus infections in US children’s hospitals, 1999–2008. Pediatrics. 2010;125(6):e1294–e1300. | |
Martinez-Aguilar G, Hammerman WA, Mason EO Jr, Kaplan SL. Clindamycin treatment of invasive infections caused by community-acquired, methicillin-resistant and methicillin-susceptible Staphylococcus aureus in children. Pediatr Infect Dis J. 2003;22(7):593–598. | |
Hyun DY, Mason EO, Forbes A, Kaplan SL. Trimethoprim-sulfamethoxazole or clindamycin for treatment of community-acquired methicillin-resistant Staphylococcus aureus skin and soft tissue infections. Pediatr Infect Dis J. 2009;28(1):57–59. | |
Williams DJ, Cooper WO, Kaltenbach LA, et al. Comparative effectiveness of antibiotic treatment strategies for pediatric skin and soft-tissue infections. Pediatrics. 2011;128(3):e479–e487. | |
Jantausch BA, Deville J, Adler S, et al. Linezolid for the treatment of children with bacteremia or nosocomial pneumonia caused by resistant gram-positive bacterial pathogens. Pediatr Infect Dis J. 2003;22(Suppl 9):S164–S171. | |
Moschovi M, Trimis G, Tsotra M, et al. Efficacy and safety of linezolid in immunocompromised children with cancer. Pediatr Int. 2010; 52(5):694–698. | |
Ardura MI, Mejías A, Katz KS, Revell P, McCracken GH Jr, Sánchez PJ. Daptomycin therapy for invasive Gram-positive bacterial infections in children. Pediatr Infect Dis J. 2007;26(12):1128–1132. | |
Palma DM, Giordano S, Neville Cracchiolo A, Zangara V, Coffaro G, Tetamo R. Daptomycin in the treatment of invasive Gram-positive bacterial infections in children: personal experience. Minerva Pediatr. 2013;65(2):173–178. | |
File TM, Low DE, Eckburg PB, et al. Integrated analysis of FOCUS 1 and FOCUS 2: randomized, doubled-blinded, multicenter phase 3 trials of the efficacy and safety of ceftaroline fosamil versus ceftriaxone in patients with community-acquired pneumonia. Clin Infect Dis. 2010;51(12):1395–1405. | |
Corey GR, Wilcox MH, Talbot GH, Thye D, Friedland D, Baculik T; CANVAS 1 investigators. CANVAS 1: the first Phase III, randomized, double-blind study evaluating ceftaroline fosamil for the treatment of patients with complicated skin and skin structure infections. J Antimicrob Chemother. 2010;65(Suppl 4):iv41–iv51. | |
Manuel O, Lien D, Weinkauf J, Humar A, Cobos I, Kumar D. Methicillin-resistant Staphylococcus aureus infection after lung transplantation: 5-year review of clinical and molecular epidemiology. J Heart Lung Transplant. 2009;28(11):1231–1236. | |
Gupta MR, Valentine VG, Walker JE Jr, et al. Clinical spectrum of gram-positive infections in lung transplantation. Transpl Infect Dis. 2009;11(5):424–431. | |
Bouchut JC, Stamm D, Boillot O, Lepape A, Floret D. Postoperative infectious complications in paediatric liver transplantation: a study of 48 transplants. Paediatr Anaesth. 2001;11(1):93–98. | |
George DL, Arnow PM, Fox AS, et al. Bacterial infection as a complication of liver transplantation: epidemiology and risk factors. Rev Infect Dis. 1991;13(3):387–396. | |
Chen M, Kallenberg CG. ANCA-associated vasculitides – advances in pathogenesis and treatment. Nat Rev Rheumatol. 2010;6(11):653–664. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.